

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50474424 CHEMBL153051
SMILES: [H][C@@]1(Cc2c[nH]cn2)CCNC1
InChI Key: InChIKey=YGGBVTGACIFVRB-ZETCQYMHSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50474424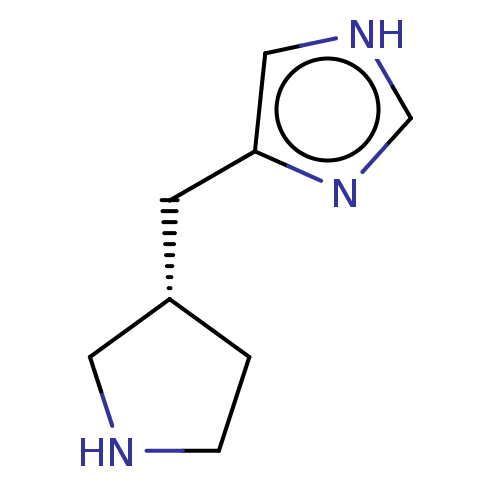 (CHEMBL153051) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Curated by ChEMBL | Assay Description Inhibitory activity measured by [3H]- N alpha- methyl-histamine binding to membranes of SK-N-MC cells expressing the human Histamine H3 receptor | J Med Chem 46: 5445-57 (2003) Article DOI: 10.1021/jm030905y BindingDB Entry DOI: 10.7270/Q2FN18X0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine receptor (H3 and H4) (Homo sapiens (Human)) | BDBM50474424 (CHEMBL153051) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | 1.00E+14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Curated by ChEMBL | Assay Description Inhibitory activity measured by [3H]- N alpha- methyl-histamine binding to membranes of SK-N-MC cells expressing the human Histamine H4 receptor | J Med Chem 46: 5445-57 (2003) Article DOI: 10.1021/jm030905y BindingDB Entry DOI: 10.7270/Q2FN18X0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine receptor (H3 and H4) (Homo sapiens (Human)) | BDBM50474424 (CHEMBL153051) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 1.02E+3 | n/a | n/a | n/a | n/a |
Vrije Universiteit Curated by ChEMBL | Assay Description Inhibitory activity determined by the inhibition of cAMP- stimulated beta-galactosidase transcription in SK-N-MC cells expressing the human Histamine... | J Med Chem 46: 5445-57 (2003) Article DOI: 10.1021/jm030905y BindingDB Entry DOI: 10.7270/Q2FN18X0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50474424 (CHEMBL153051) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.389 | n/a | n/a | n/a | n/a |
Vrije Universiteit Curated by ChEMBL | Assay Description Inhibitory activity determined by the inhibition of cAMP- stimulated beta-galactosidase transcription in SK-N-MC cells expressing the human Histamine... | J Med Chem 46: 5445-57 (2003) Article DOI: 10.1021/jm030905y BindingDB Entry DOI: 10.7270/Q2FN18X0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||