 Found 17 hits for monomerid = 50504104
Found 17 hits for monomerid = 50504104 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Fibroblast growth factor receptor 1
(Homo sapiens (Human)) | BDBM50504104
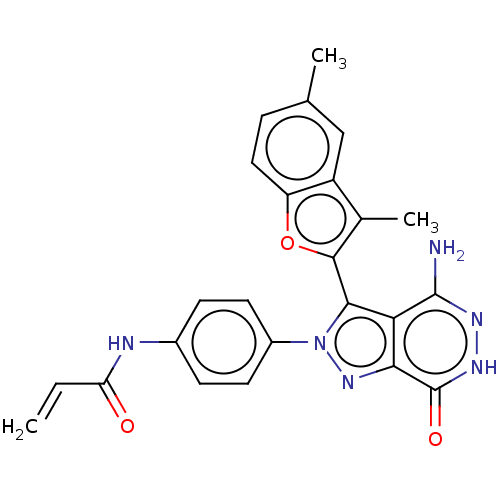
(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Irreversible inhibition of human FGFR1 using poly (Glu, Tyr)4:1 as substrate measured after 60 mins |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human c-MET using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA relative to control |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human EGFR using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA relative to control |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
Receptor tyrosine-protein kinase erbB-4
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human ERbb4 using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA relative to control |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL1
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human ABL using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA relative to control |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 1
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human FGFR1 using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 3
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human FGFR3 using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human PDGFRbeta using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
1,3-beta-glucan synthase component GSC2
(Saccharomyces cerevisiae) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch-clamp method |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human c-Src using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA relative to control |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
ALK tyrosine kinase receptor
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human ALK using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA relative to control |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 1
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human VEGFR1 using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 2
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human FGFR2 using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 4
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human FGFR4 using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Receptor tyrosine-protein kinase erbB-2
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human ERbb2 using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA relative to control |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
Ephrin type-A receptor 2
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human EPHA2 using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA relative to control |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50504104

(CHEMBL4446835)Show SMILES Cc1c(oc2ccc(C)cc12)-c1n(nc2c1c(N)n[nH]c2=O)-c1ccc(NC(=O)C=C)cc1 |(12.65,-38.72,;11.4,-37.83,;9.94,-38.31,;9.03,-37.07,;9.93,-35.82,;9.6,-34.32,;10.73,-33.29,;12.2,-33.76,;13.34,-32.72,;12.53,-35.26,;11.39,-36.29,;9.47,-39.77,;10.38,-41.01,;9.48,-42.26,;8.02,-41.79,;8.01,-40.26,;6.68,-39.48,;6.68,-37.94,;5.35,-40.26,;5.35,-41.8,;6.68,-42.56,;6.68,-44.1,;11.92,-41,;12.69,-42.33,;14.23,-42.32,;15,-40.98,;16.54,-40.97,;17.3,-39.63,;16.52,-38.3,;18.84,-39.62,;19.6,-38.28,;14.21,-39.65,;12.67,-39.66,)| Show InChI InChI=1S/C24H20N6O3/c1-4-18(31)26-14-6-8-15(9-7-14)30-21(19-20(29-30)24(32)28-27-23(19)25)22-13(3)16-11-12(2)5-10-17(16)33-22/h4-11H,1H2,2-3H3,(H2,25,27)(H,26,31)(H,28,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai University
Curated by ChEMBL
| Assay Description
Inhibition of human VEGFR2 using poly (Glu, Tyr)4:1 as substrate measured after 60 mins by ELISA |
J Med Chem 62: 7473-7488 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00510 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data