

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: O=c1cc(OCCn2ccnc2)c2ccccc2o1
InChI Key: InChIKey=MXJNKMWQNZHBOE-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aromatase (Homo sapiens (Human)) | BDBM9473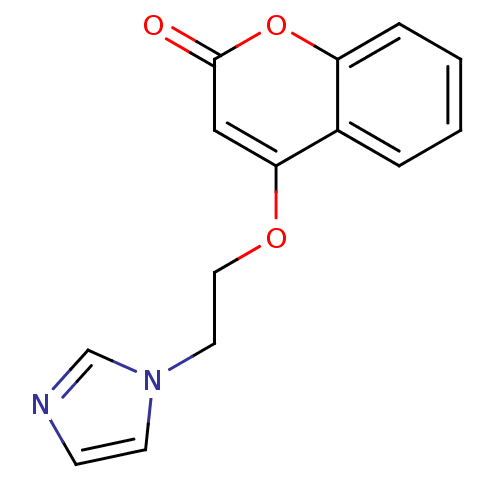 (4-[2-(1H-Imidazol-1-yl)ethoxy]-2H-chromen-2-one | ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 760 | n/a | n/a | n/a | n/a | 7.4 | 30 |
University of Bari | Assay Description The enzyme activity was assayed by measuring the 3H-labeled H2O formed from [1beta, 2beta-3H] testosterone or [1beta-3H] androstenedione during aroma... | J Med Chem 47: 6792-803 (2004) Article DOI: 10.1021/jm049535j BindingDB Entry DOI: 10.7270/Q2MS3R0C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aromatase (Homo sapiens (Human)) | BDBM9473 (4-[2-(1H-Imidazol-1-yl)ethoxy]-2H-chromen-2-one | ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 760 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade de Coimbra Curated by ChEMBL | Assay Description Inhibition of human aromatase-mediated conversion of [1beta3H]androstenedione to estrone by liquid scintillation counting in presence of NADPH | J Med Chem 52: 143-50 (2009) Article DOI: 10.1021/jm800945c BindingDB Entry DOI: 10.7270/Q2SQ9080 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Steroid 17-alpha-hydroxylase/17,20 lyase (Homo sapiens (Human)) | BDBM9473 (4-[2-(1H-Imidazol-1-yl)ethoxy]-2H-chromen-2-one | ...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bari | Assay Description The 17 alpha-hydroxylase activity of CYP 17 was determined by measuring the conversion of progesterone into 17 alpha-hydroxyprogesterone and the bypr... | J Med Chem 47: 6792-803 (2004) Article DOI: 10.1021/jm049535j BindingDB Entry DOI: 10.7270/Q2MS3R0C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||