 Found 278 hits with Last Name = 'abdelazeem' and Initial = 'ah'
Found 278 hits with Last Name = 'abdelazeem' and Initial = 'ah' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50614343
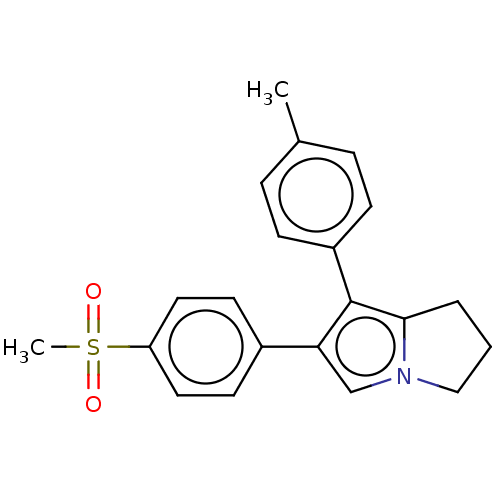
(CHEMBL597733)Show SMILES Cc1ccc(cc1)-c1c2CCCn2cc1-c1ccc(cc1)S(C)(=O)=O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50614341

(CHEMBL5269462)Show SMILES CS(=O)(=O)c1ccc(cc1)-c1cn2CCCc2c1-c1ccc(Cl)cc1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50252033
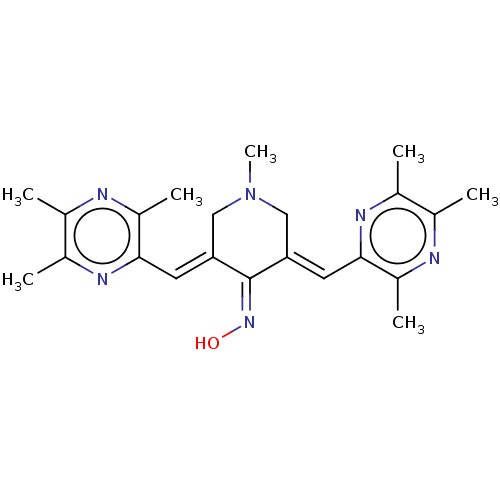
(CHEMBL4078023)Show SMILES [#6]-[#7]1-[#6]\[#6](=[#6]/c2nc(-[#6])c(-[#6])nc2-[#6])\[#6](=[#7]/[#8])\[#6](\[#6]-1)=[#6]\c1nc(-[#6])c(-[#6])nc1-[#6] Show InChI InChI=1S/C22H28N6O/c1-12-14(3)25-20(16(5)23-12)8-18-10-28(7)11-19(22(18)27-29)9-21-17(6)24-13(2)15(4)26-21/h8-9,29H,10-11H2,1-7H3/b18-8+,19-9+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Wuhan University of Technology
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged recombinant EGFR cytoplasmic domain (unknown origin) (645 to 1186 residues) expressed in baculovirus infected Sf-9 cells pr... |
Eur J Med Chem 135: 34-48 (2017)
Article DOI: 10.1016/j.ejmech.2017.04.025
BindingDB Entry DOI: 10.7270/Q2H41TV5 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50252035
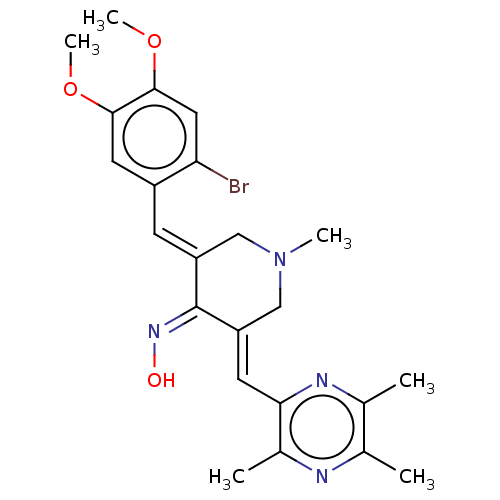
(CHEMBL4064361)Show SMILES COc1cc(Br)c(\C=C2/CN(C)C/C(=C\c3nc(C)c(C)nc3C)/C/2=N\O)cc1OC Show InChI InChI=1S/C23H27BrN4O3/c1-13-14(2)26-20(15(3)25-13)8-18-12-28(4)11-17(23(18)27-29)7-16-9-21(30-5)22(31-6)10-19(16)24/h7-10,29H,11-12H2,1-6H3/b17-7+,18-8+,27-23- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Wuhan University of Technology
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged recombinant EGFR cytoplasmic domain (unknown origin) (645 to 1186 residues) expressed in baculovirus infected Sf-9 cells pr... |
Eur J Med Chem 135: 34-48 (2017)
Article DOI: 10.1016/j.ejmech.2017.04.025
BindingDB Entry DOI: 10.7270/Q2H41TV5 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM50614346
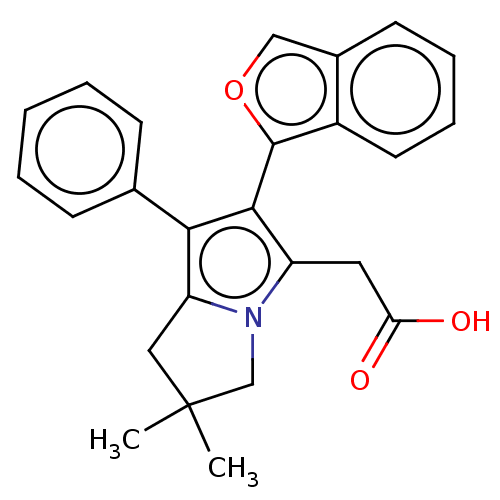
(CHEMBL5271431)Show SMILES CC1(C)Cc2c(c(c(CC(O)=O)n2C1)-c1occ2ccccc12)-c1ccccc1 |(12.77,-20.04,;13.55,-21.39,;12.06,-21.78,;14.79,-20.48,;16.04,-21.39,;17.58,-21.39,;18.06,-22.86,;16.81,-23.76,;16.8,-25.3,;15.47,-26.07,;14.14,-25.3,;15.47,-27.61,;15.56,-22.85,;14.02,-22.85,;19.52,-23.34,;19.99,-24.8,;21.53,-24.79,;22.01,-23.34,;23.42,-22.71,;23.59,-21.18,;22.34,-20.27,;20.93,-20.9,;20.77,-22.43,;18.49,-20.15,;20.02,-20.32,;20.93,-19.08,;20.31,-17.67,;18.77,-17.51,;17.86,-18.75,)| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50614346

(CHEMBL5271431)Show SMILES CC1(C)Cc2c(c(c(CC(O)=O)n2C1)-c1occ2ccccc12)-c1ccccc1 |(12.77,-20.04,;13.55,-21.39,;12.06,-21.78,;14.79,-20.48,;16.04,-21.39,;17.58,-21.39,;18.06,-22.86,;16.81,-23.76,;16.8,-25.3,;15.47,-26.07,;14.14,-25.3,;15.47,-27.61,;15.56,-22.85,;14.02,-22.85,;19.52,-23.34,;19.99,-24.8,;21.53,-24.79,;22.01,-23.34,;23.42,-22.71,;23.59,-21.18,;22.34,-20.27,;20.93,-20.9,;20.77,-22.43,;18.49,-20.15,;20.02,-20.32,;20.93,-19.08,;20.31,-17.67,;18.77,-17.51,;17.86,-18.75,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM50614342
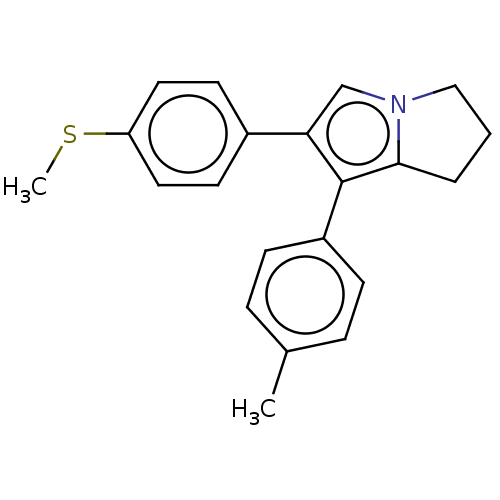
(CHEMBL5279593) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50614358

(CHEMBL5290904) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Ovis aries (Sheep)) | BDBM17638

(2-{1-[(4-chlorophenyl)carbonyl]-5-methoxy-2-methyl...)Show SMILES COc1ccc2n(C(=O)c3ccc(Cl)cc3)c(C)c(CC(O)=O)c2c1 Show InChI InChI=1S/C19H16ClNO4/c1-11-15(10-18(22)23)16-9-14(25-2)7-8-17(16)21(11)19(24)12-3-5-13(20)6-4-12/h3-9H,10H2,1-2H3,(H,22,23) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Umm Al-Qura University, Makkah 21955, Saudi Arabia; Department of Medicinal Chemistry, Faculty of Pharmacy, Beni-Suef University, Beni-Sue
Curated by ChEMBL
| Assay Description
Inhibition of ovine COX1 assessed as reduction in PGH2 formation by measuring PGF2alpha by colorimetric assay |
Bioorg Med Chem 25: 5637-5651 (2017)
Article DOI: 10.1016/j.bmc.2017.08.039
BindingDB Entry DOI: 10.7270/Q2JQ13GQ |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50252089
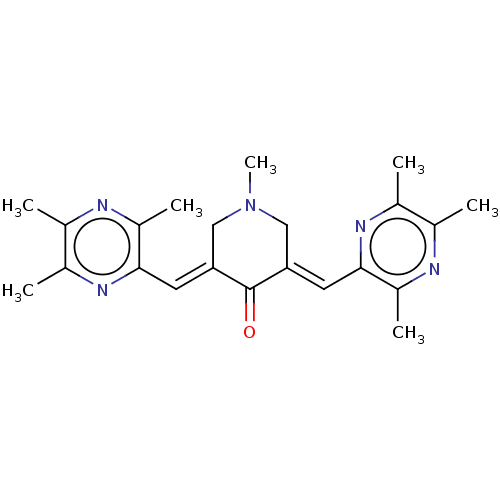
(CHEMBL4083642)Show SMILES CN1C\C(=C/c2nc(C)c(C)nc2C)C(=O)\C(C1)=C\c1nc(C)c(C)nc1C Show InChI InChI=1S/C22H27N5O/c1-12-14(3)25-20(16(5)23-12)8-18-10-27(7)11-19(22(18)28)9-21-17(6)24-13(2)15(4)26-21/h8-9H,10-11H2,1-7H3/b18-8+,19-9+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Wuhan University of Technology
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged recombinant EGFR cytoplasmic domain (unknown origin) (645 to 1186 residues) expressed in baculovirus infected Sf-9 cells pr... |
Eur J Med Chem 135: 34-48 (2017)
Article DOI: 10.1016/j.ejmech.2017.04.025
BindingDB Entry DOI: 10.7270/Q2H41TV5 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50614351
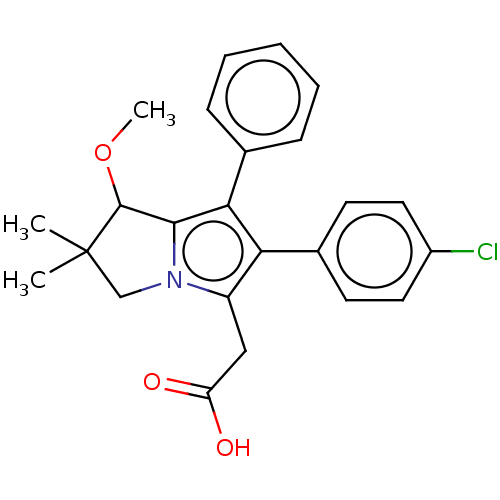
(CHEMBL5268642)Show SMILES COC1c2c(c(c(CC(O)=O)n2CC1(C)C)-c1ccc(Cl)cc1)-c1ccccc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Mus musculus) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Nahda University
Curated by ChEMBL
| Assay Description
Inhibition of mouse full length GST-tagged BRAF V600E mutant using recombinant human His6-tagged MEK1 as substrate preincubated for 1 hr followed by ... |
Eur J Med Chem 156: 774-789 (2018)
Article DOI: 10.1016/j.ejmech.2018.07.024
BindingDB Entry DOI: 10.7270/Q2GF0X69 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM50614344

(CHEMBL5269592)Show SMILES CC1(C)Cn2c(CC(O)=O)c(c(c2C1=O)-c1ccccc1)-c1ccc(Cl)cc1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 46 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM11639

(4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyraz...)Show SMILES Cc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O2S/c1-11-2-4-12(5-3-11)15-10-16(17(18,19)20)22-23(15)13-6-8-14(9-7-13)26(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Umm Al-Qura University, Makkah 21955, Saudi Arabia; Department of Medicinal Chemistry, Faculty of Pharmacy, Beni-Suef University, Beni-Sue
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant COX2 assessed as reduction in PGH2 formation by measuring PGF2alpha by colorimetric assay |
Bioorg Med Chem 25: 5637-5651 (2017)
Article DOI: 10.1016/j.bmc.2017.08.039
BindingDB Entry DOI: 10.7270/Q2JQ13GQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
Beni-Suef University
Curated by ChEMBL
| Assay Description
Inhibition of recombinant full-length GST-tagged human B-RAF V600E mutant (417 to 766 residues) expressed in Baculovirus infected Sf9 cells using N-t... |
Eur J Med Chem 150: 567-578 (2018)
Article DOI: 10.1016/j.ejmech.2018.03.001
BindingDB Entry DOI: 10.7270/Q26D5WNS |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM50614348

(CHEMBL5280353)Show SMILES CC1(C)Cc2c(c(-c3ccsc3)c(CC(O)=O)n2C1)-c1ccccc1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Mus musculus) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
Aljouf University
Curated by ChEMBL
| Assay Description
Inhibition of mouse full-length GST-tagged BRAF V600E mutant using recombinant human full length N-terminal His-tagged MEK1 as substrate preincubated... |
Eur J Med Chem 146: 260-273 (2018)
Article DOI: 10.1016/j.ejmech.2018.01.042
BindingDB Entry DOI: 10.7270/Q25X2CKD |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50614352

(CHEMBL5277274)Show SMILES COC1c2c(c(c(C)n2CC1(C)C)-c1ccc(Cl)cc1)-c1ccccc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM50614351

(CHEMBL5268642)Show SMILES COC1c2c(c(c(CC(O)=O)n2CC1(C)C)-c1ccc(Cl)cc1)-c1ccccc1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50252090
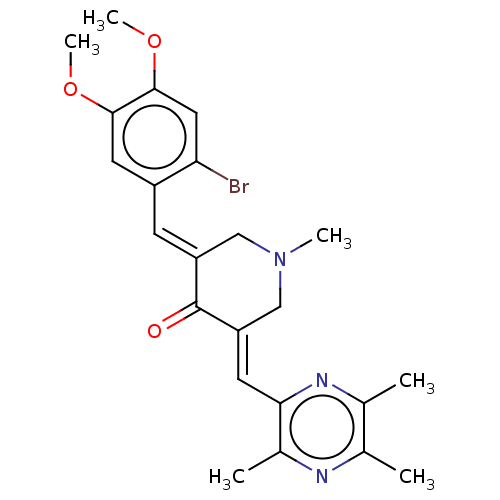
(CHEMBL4063890)Show SMILES COc1cc(Br)c(\C=C2/CN(C)C\C(=C/c3nc(C)c(C)nc3C)C2=O)cc1OC Show InChI InChI=1S/C23H26BrN3O3/c1-13-14(2)26-20(15(3)25-13)8-18-12-27(4)11-17(23(18)28)7-16-9-21(29-5)22(30-6)10-19(16)24/h7-10H,11-12H2,1-6H3/b17-7+,18-8+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
Wuhan University of Technology
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged recombinant EGFR cytoplasmic domain (unknown origin) (645 to 1186 residues) expressed in baculovirus infected Sf-9 cells pr... |
Eur J Med Chem 135: 34-48 (2017)
Article DOI: 10.1016/j.ejmech.2017.04.025
BindingDB Entry DOI: 10.7270/Q2H41TV5 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
Wuhan University of Technology
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged recombinant EGFR cytoplasmic domain (unknown origin) (645 to 1186 residues) expressed in baculovirus infected Sf-9 cells pr... |
Eur J Med Chem 135: 34-48 (2017)
Article DOI: 10.1016/j.ejmech.2017.04.025
BindingDB Entry DOI: 10.7270/Q2H41TV5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
Nahda University
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged recombinant EGFR cytoplasmic domain (645 to 1186 residues) (unknown origin) expressed in baculovirus infected Sf9 insect ce... |
Eur J Med Chem 156: 774-789 (2018)
Article DOI: 10.1016/j.ejmech.2018.07.024
BindingDB Entry DOI: 10.7270/Q2GF0X69 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
Aljouf University
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged EGFR (unknown origin) cytoplasmic domain expressed in baculovirus infected Sf9 insect cells after 10 mins followed by addit... |
Eur J Med Chem 146: 260-273 (2018)
Article DOI: 10.1016/j.ejmech.2018.01.042
BindingDB Entry DOI: 10.7270/Q25X2CKD |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
Beni-Suef University
Curated by ChEMBL
| Assay Description
Inhibition of His-6 tagged recombinant EGFR cytoplasmic domain (645 to 1186 residues) (unknown origin) expressed in Baculovirus infected Sf9 cells by... |
Eur J Med Chem 150: 567-578 (2018)
Article DOI: 10.1016/j.ejmech.2018.03.001
BindingDB Entry DOI: 10.7270/Q26D5WNS |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine/threonine-protein kinase B-raf
(Mus musculus) | BDBM50457924
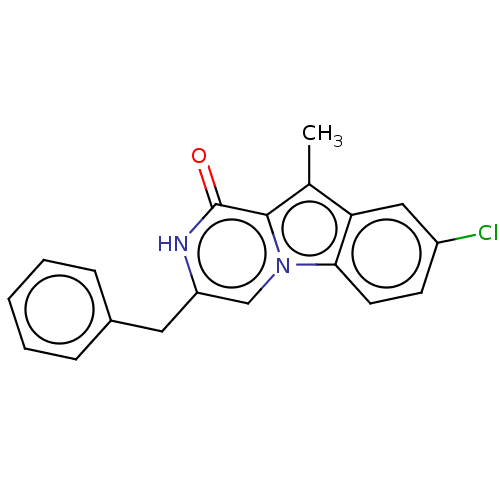
(CHEMBL4208224)Show InChI InChI=1S/C19H15ClN2O/c1-12-16-10-14(20)7-8-17(16)22-11-15(21-19(23)18(12)22)9-13-5-3-2-4-6-13/h2-8,10-11H,9H2,1H3,(H,21,23) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Aljouf University
Curated by ChEMBL
| Assay Description
Inhibition of mouse full-length GST-tagged BRAF V600E mutant using recombinant human full length N-terminal His-tagged MEK1 as substrate preincubated... |
Eur J Med Chem 146: 260-273 (2018)
Article DOI: 10.1016/j.ejmech.2018.01.042
BindingDB Entry DOI: 10.7270/Q25X2CKD |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50261639

(CHEMBL4076689)Show SMILES O=C(Nc1ccccc1)c1c(N=Cc2cccnc2)c(C#N)c2CCCn12 Show InChI InChI=1S/C21H17N5O/c22-12-17-18-9-5-11-26(18)20(21(27)25-16-7-2-1-3-8-16)19(17)24-14-15-6-4-10-23-13-15/h1-4,6-8,10,13-14H,5,9,11H2,(H,25,27)/b24-14+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Umm Al-Qura University, Makkah 21955, Saudi Arabia; Department of Medicinal Chemistry, Faculty of Pharmacy, Beni-Suef University, Beni-Sue
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant COX2 assessed as reduction in PGH2 formation by measuring PGF2alpha by colorimetric assay |
Bioorg Med Chem 25: 5637-5651 (2017)
Article DOI: 10.1016/j.bmc.2017.08.039
BindingDB Entry DOI: 10.7270/Q2JQ13GQ |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50614356

(CHEMBL5270458) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50261633

(CHEMBL4079032)Show SMILES Cc1ccc(NC(=O)c2c(N=Cc3ccc4ccccc4c3)c(C#N)c3CCCn23)cc1 Show InChI InChI=1S/C27H22N4O/c1-18-8-12-22(13-9-18)30-27(32)26-25(23(16-28)24-7-4-14-31(24)26)29-17-19-10-11-20-5-2-3-6-21(20)15-19/h2-3,5-6,8-13,15,17H,4,7,14H2,1H3,(H,30,32)/b29-17+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Umm Al-Qura University, Makkah 21955, Saudi Arabia; Department of Medicinal Chemistry, Faculty of Pharmacy, Beni-Suef University, Beni-Sue
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant COX2 assessed as reduction in PGH2 formation by measuring PGF2alpha by colorimetric assay |
Bioorg Med Chem 25: 5637-5651 (2017)
Article DOI: 10.1016/j.bmc.2017.08.039
BindingDB Entry DOI: 10.7270/Q2JQ13GQ |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50261638
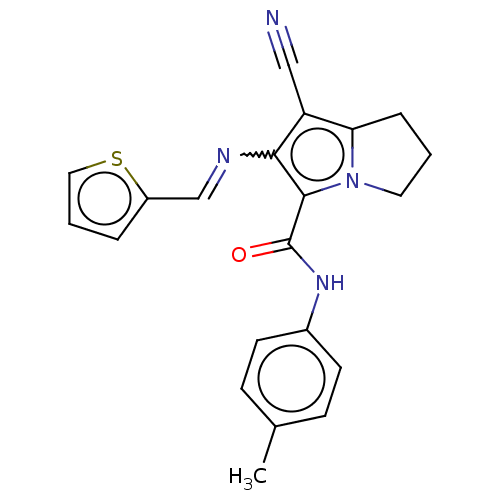
(CHEMBL4073779)Show SMILES Cc1ccc(NC(=O)c2c(N=Cc3cccs3)c(C#N)c3CCCn23)cc1 Show InChI InChI=1S/C21H18N4OS/c1-14-6-8-15(9-7-14)24-21(26)20-19(23-13-16-4-3-11-27-16)17(12-22)18-5-2-10-25(18)20/h3-4,6-9,11,13H,2,5,10H2,1H3,(H,24,26)/b23-13+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Umm Al-Qura University, Makkah 21955, Saudi Arabia; Department of Medicinal Chemistry, Faculty of Pharmacy, Beni-Suef University, Beni-Sue
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant COX2 assessed as reduction in PGH2 formation by measuring PGF2alpha by colorimetric assay |
Bioorg Med Chem 25: 5637-5651 (2017)
Article DOI: 10.1016/j.bmc.2017.08.039
BindingDB Entry DOI: 10.7270/Q2JQ13GQ |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM50614347

(CHEMBL5284424)Show SMILES CC1(C)Cc2c(c(-c3ccco3)c(CC(O)=O)n2C1)-c1ccccc1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50261640

(CHEMBL4096865)Show SMILES Cc1ccc(NC(=O)c2c(N=Cc3cccnc3)c(C#N)c3CCCn23)cc1 Show InChI InChI=1S/C22H19N5O/c1-15-6-8-17(9-7-15)26-22(28)21-20(25-14-16-4-2-10-24-13-16)18(12-23)19-5-3-11-27(19)21/h2,4,6-10,13-14H,3,5,11H2,1H3,(H,26,28)/b25-14+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Umm Al-Qura University, Makkah 21955, Saudi Arabia; Department of Medicinal Chemistry, Faculty of Pharmacy, Beni-Suef University, Beni-Sue
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant COX2 assessed as reduction in PGH2 formation by measuring PGF2alpha by colorimetric assay |
Bioorg Med Chem 25: 5637-5651 (2017)
Article DOI: 10.1016/j.bmc.2017.08.039
BindingDB Entry DOI: 10.7270/Q2JQ13GQ |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50614340
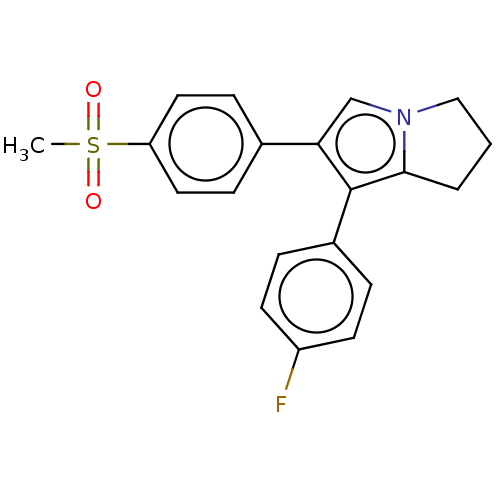
(CHEMBL5286930)Show SMILES CS(=O)(=O)c1ccc(cc1)-c1cn2CCCc2c1-c1ccc(F)cc1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50261634
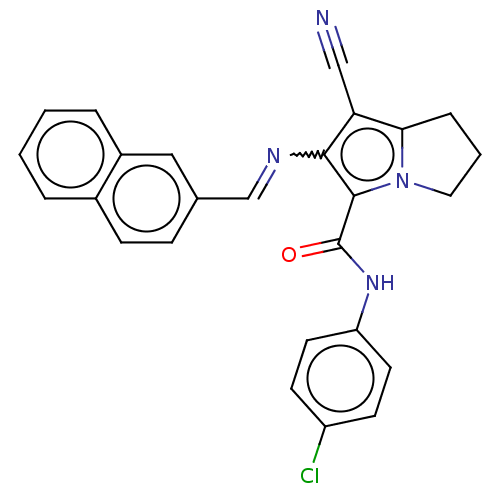
(CHEMBL4097045)Show SMILES Clc1ccc(NC(=O)c2c(N=Cc3ccc4ccccc4c3)c(C#N)c3CCCn23)cc1 Show InChI InChI=1S/C26H19ClN4O/c27-20-9-11-21(12-10-20)30-26(32)25-24(22(15-28)23-6-3-13-31(23)25)29-16-17-7-8-18-4-1-2-5-19(18)14-17/h1-2,4-5,7-12,14,16H,3,6,13H2,(H,30,32)/b29-16+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Umm Al-Qura University, Makkah 21955, Saudi Arabia; Department of Medicinal Chemistry, Faculty of Pharmacy, Beni-Suef University, Beni-Sue
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant COX2 assessed as reduction in PGH2 formation by measuring PGF2alpha by colorimetric assay |
Bioorg Med Chem 25: 5637-5651 (2017)
Article DOI: 10.1016/j.bmc.2017.08.039
BindingDB Entry DOI: 10.7270/Q2JQ13GQ |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50038649
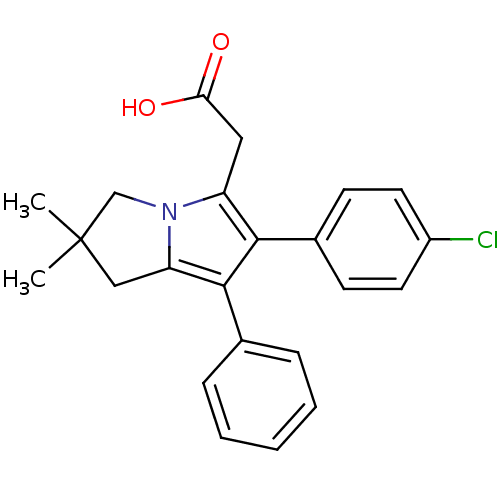
(2-(6-(4-chlorophenyl)-2,2-dimethyl-7-phenyl-2,3-di...)Show SMILES CC1(C)Cc2c(c(c(CC(O)=O)n2C1)-c1ccc(Cl)cc1)-c1ccccc1 Show InChI InChI=1S/C23H22ClNO2/c1-23(2)13-19-22(15-6-4-3-5-7-15)21(16-8-10-17(24)11-9-16)18(12-20(26)27)25(19)14-23/h3-11H,12-14H2,1-2H3,(H,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| | n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50261643

(CHEMBL4071061)Show SMILES Cc1ccc(NC(=O)c2c(N=Cc3ccco3)c(C#N)c3CCCn23)cc1 Show InChI InChI=1S/C21H18N4O2/c1-14-6-8-15(9-7-14)24-21(26)20-19(23-13-16-4-3-11-27-16)17(12-22)18-5-2-10-25(18)20/h3-4,6-9,11,13H,2,5,10H2,1H3,(H,24,26)/b23-13+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Umm Al-Qura University, Makkah 21955, Saudi Arabia; Department of Medicinal Chemistry, Faculty of Pharmacy, Beni-Suef University, Beni-Sue
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant COX2 assessed as reduction in PGH2 formation by measuring PGF2alpha by colorimetric assay |
Bioorg Med Chem 25: 5637-5651 (2017)
Article DOI: 10.1016/j.bmc.2017.08.039
BindingDB Entry DOI: 10.7270/Q2JQ13GQ |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM50614349

(CHEMBL5289423)Show SMILES CC1(C)Cc2c(c(-c3ccc(Cl)s3)c(CC(O)=O)n2C1)-c1ccccc1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM50038649

(2-(6-(4-chlorophenyl)-2,2-dimethyl-7-phenyl-2,3-di...)Show SMILES CC1(C)Cc2c(c(c(CC(O)=O)n2C1)-c1ccc(Cl)cc1)-c1ccccc1 Show InChI InChI=1S/C23H22ClNO2/c1-23(2)13-19-22(15-6-4-3-5-7-15)21(16-8-10-17(24)11-9-16)18(12-20(26)27)25(19)14-23/h3-11H,12-14H2,1-2H3,(H,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| | n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50614345

(CHEMBL5277078)Show SMILES Cc1c(c(c2C(=O)C(C)(C)Cn12)-c1ccccc1)-c1ccc(Cl)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 230 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50614349

(CHEMBL5289423)Show SMILES CC1(C)Cc2c(c(-c3ccc(Cl)s3)c(CC(O)=O)n2C1)-c1ccccc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Mus musculus (mouse)) | BDBM50105335

(CHEMBL3597247)Show SMILES O=c1sc2ccccc2n1CCCCCCN1CCN(CCCCCCn2c3ccccc3sc2=O)CC1 Show InChI InChI=1S/C30H40N4O2S2/c35-29-33(25-13-5-7-15-27(25)37-29)19-11-3-1-9-17-31-21-23-32(24-22-31)18-10-2-4-12-20-34-26-14-6-8-16-28(26)38-30(34)36/h5-8,13-16H,1-4,9-12,17-24H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 280 | n/a | n/a | n/a | n/a | n/a | n/a |
Beni-Suef University
Curated by ChEMBL
| Assay Description
Antiinflammatory activity in mouse RAW264.7 cells assessed as inhibition of LPS-induced iNOS activity incubated for 30 mins prior to LPS challenge me... |
Bioorg Med Chem 23: 3248-59 (2015)
Article DOI: 10.1016/j.bmc.2015.04.057
BindingDB Entry DOI: 10.7270/Q26H4K5F |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Mus musculus (mouse)) | BDBM50105353
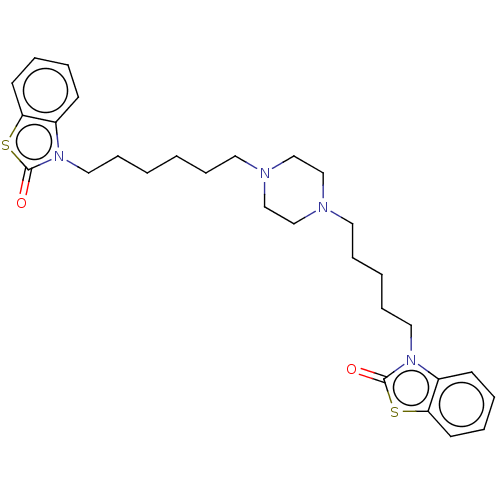
(CHEMBL3597266)Show SMILES O=c1sc2ccccc2n1CCCCCCN1CCN(CCCCCn2c3ccccc3sc2=O)CC1 Show InChI InChI=1S/C29H38N4O2S2/c34-28-32(24-12-4-6-14-26(24)36-28)18-10-2-1-8-16-30-20-22-31(23-21-30)17-9-3-11-19-33-25-13-5-7-15-27(25)37-29(33)35/h4-7,12-15H,1-3,8-11,16-23H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a |
Beni-Suef University
Curated by ChEMBL
| Assay Description
Antiinflammatory activity in mouse RAW264.7 cells assessed as inhibition of LPS-induced iNOS activity incubated for 30 mins prior to LPS challenge me... |
Bioorg Med Chem 23: 3248-59 (2015)
Article DOI: 10.1016/j.bmc.2015.04.057
BindingDB Entry DOI: 10.7270/Q26H4K5F |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Ovis aries (Sheep)) | BDBM50207432

(CHEMBL3909479)Show SMILES COc1ccc(cc1)-c1nc(NCC(O)=O)sc1-c1ccc(OC)cc1 Show InChI InChI=1S/C19H18N2O4S/c1-24-14-7-3-12(4-8-14)17-18(13-5-9-15(25-2)10-6-13)26-19(21-17)20-11-16(22)23/h3-10H,11H2,1-2H3,(H,20,21)(H,22,23) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 320 | n/a | n/a | n/a | n/a | n/a | n/a |
Beni-Suef University
Curated by ChEMBL
| Assay Description
Inhibition of ovine COX-1 assessed as reduction in oxidation of TMPD using arachidonic acid as substrate preincubated for 5 mins followed by substrat... |
Bioorg Med Chem 25: 665-676 (2017)
Article DOI: 10.1016/j.bmc.2016.11.037
BindingDB Entry DOI: 10.7270/Q2542QKM |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM11639

(4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyraz...)Show SMILES Cc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O2S/c1-11-2-4-12(5-3-11)15-10-16(17(18,19)20)22-23(15)13-6-8-14(9-7-13)26(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
Beni-Suef University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant COX-2 assessed as reduction in oxidation of TMPD using arachidonic acid as substrate preincubated for 5 mins followed... |
Bioorg Med Chem 25: 665-676 (2017)
Article DOI: 10.1016/j.bmc.2016.11.037
BindingDB Entry DOI: 10.7270/Q2542QKM |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50261644
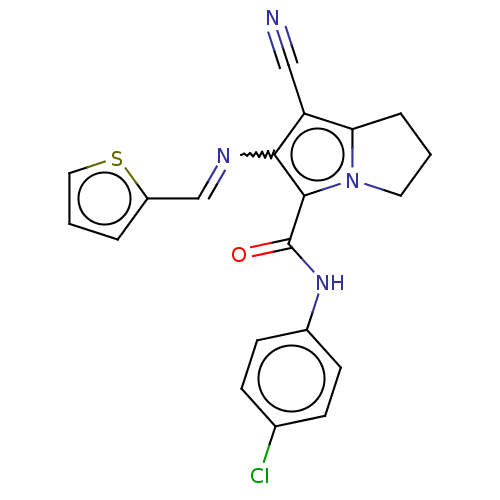
(CHEMBL4072244)Show SMILES Clc1ccc(NC(=O)c2c(N=Cc3cccs3)c(C#N)c3CCCn23)cc1 Show InChI InChI=1S/C20H15ClN4OS/c21-13-5-7-14(8-6-13)24-20(26)19-18(23-12-15-3-2-10-27-15)16(11-22)17-4-1-9-25(17)19/h2-3,5-8,10,12H,1,4,9H2,(H,24,26)/b23-12+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 360 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Umm Al-Qura University, Makkah 21955, Saudi Arabia; Department of Medicinal Chemistry, Faculty of Pharmacy, Beni-Suef University, Beni-Sue
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant COX2 assessed as reduction in PGH2 formation by measuring PGF2alpha by colorimetric assay |
Bioorg Med Chem 25: 5637-5651 (2017)
Article DOI: 10.1016/j.bmc.2017.08.039
BindingDB Entry DOI: 10.7270/Q2JQ13GQ |
More data for this
Ligand-Target Pair | |
Bifunctional epoxide hydrolase 2
(Homo sapiens (Human)) | BDBM50565488

(CHEMBL4783386)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(NC(=O)Nc2ccc(cc2)C(F)(F)F)cc1-c1ccccc1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human sEH using epoxy flour 7 as substrate measured after 30 mins by cell-based fluorescence assay |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112662
BindingDB Entry DOI: 10.7270/Q2B85CWK |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Mus musculus (mouse)) | BDBM50105355
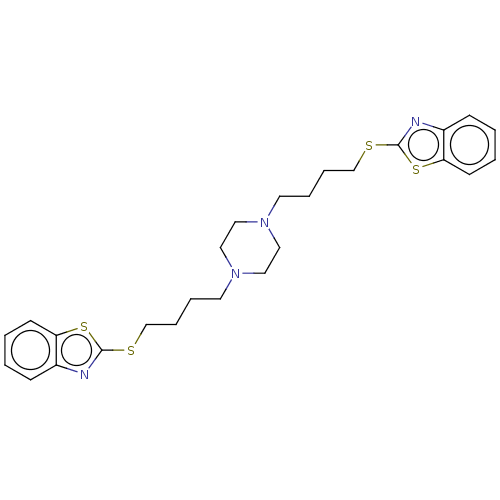
(CHEMBL3597268)Show SMILES C(CCN1CCN(CCCCSc2nc3ccccc3s2)CC1)CSc1nc2ccccc2s1 Show InChI InChI=1S/C26H32N4S4/c1-3-11-23-21(9-1)27-25(33-23)31-19-7-5-13-29-15-17-30(18-16-29)14-6-8-20-32-26-28-22-10-2-4-12-24(22)34-26/h1-4,9-12H,5-8,13-20H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 410 | n/a | n/a | n/a | n/a | n/a | n/a |
Beni-Suef University
Curated by ChEMBL
| Assay Description
Antiinflammatory activity in mouse RAW264.7 cells assessed as inhibition of LPS-induced iNOS activity incubated for 30 mins prior to LPS challenge me... |
Bioorg Med Chem 23: 3248-59 (2015)
Article DOI: 10.1016/j.bmc.2015.04.057
BindingDB Entry DOI: 10.7270/Q26H4K5F |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Mus musculus (mouse)) | BDBM50105333
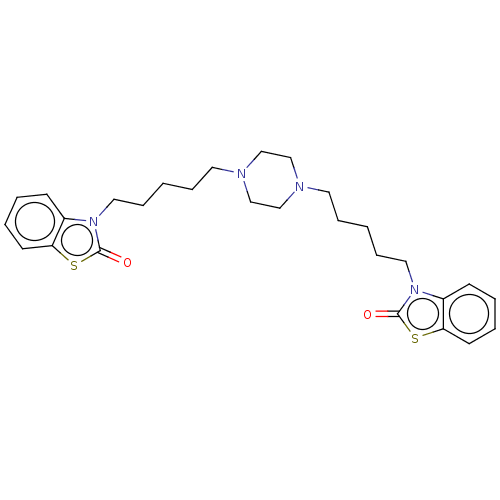
(CHEMBL3597245)Show SMILES O=c1sc2ccccc2n1CCCCCN1CCN(CCCCCn2c3ccccc3sc2=O)CC1 Show InChI InChI=1S/C28H36N4O2S2/c33-27-31(23-11-3-5-13-25(23)35-27)17-9-1-7-15-29-19-21-30(22-20-29)16-8-2-10-18-32-24-12-4-6-14-26(24)36-28(32)34/h3-6,11-14H,1-2,7-10,15-22H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 410 | n/a | n/a | n/a | n/a | n/a | n/a |
Beni-Suef University
Curated by ChEMBL
| Assay Description
Antiinflammatory activity in mouse RAW264.7 cells assessed as inhibition of LPS-induced iNOS activity incubated for 30 mins prior to LPS challenge me... |
Bioorg Med Chem 23: 3248-59 (2015)
Article DOI: 10.1016/j.bmc.2015.04.057
BindingDB Entry DOI: 10.7270/Q26H4K5F |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Ovis aries (Sheep)) | BDBM50207431
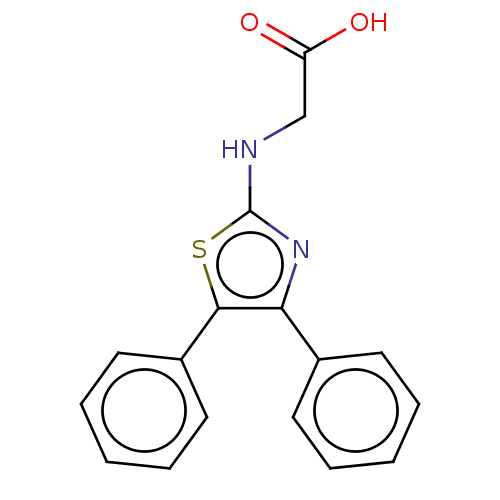
(CHEMBL3892572)Show InChI InChI=1S/C17H14N2O2S/c20-14(21)11-18-17-19-15(12-7-3-1-4-8-12)16(22-17)13-9-5-2-6-10-13/h1-10H,11H2,(H,18,19)(H,20,21) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 420 | n/a | n/a | n/a | n/a | n/a | n/a |
Beni-Suef University
Curated by ChEMBL
| Assay Description
Inhibition of ovine COX-1 assessed as reduction in oxidation of TMPD using arachidonic acid as substrate preincubated for 5 mins followed by substrat... |
Bioorg Med Chem 25: 665-676 (2017)
Article DOI: 10.1016/j.bmc.2016.11.037
BindingDB Entry DOI: 10.7270/Q2542QKM |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50261636

(CHEMBL4065185)Show SMILES Clc1ccc(NC(=O)c2c(N=Cc3ccco3)c(C#N)c3CCCn23)cc1 Show InChI InChI=1S/C20H15ClN4O2/c21-13-5-7-14(8-6-13)24-20(26)19-18(23-12-15-3-2-10-27-15)16(11-22)17-4-1-9-25(17)19/h2-3,5-8,10,12H,1,4,9H2,(H,24,26)/b23-12+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 450 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Umm Al-Qura University, Makkah 21955, Saudi Arabia; Department of Medicinal Chemistry, Faculty of Pharmacy, Beni-Suef University, Beni-Sue
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant COX2 assessed as reduction in PGH2 formation by measuring PGF2alpha by colorimetric assay |
Bioorg Med Chem 25: 5637-5651 (2017)
Article DOI: 10.1016/j.bmc.2017.08.039
BindingDB Entry DOI: 10.7270/Q2JQ13GQ |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM50391240

(CHEBI:76228 | CHEMBL496240)Show InChI InChI=1S/C11H12N4O/c12-10-9(6-14-11(13)15-10)16-7-8-4-2-1-3-5-8/h1-6H,7H2,(H4,12,13,14,15) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| | n/a | n/a | 460 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data