 Found 139 hits with Last Name = 'de la motte' and Initial = 'h'
Found 139 hits with Last Name = 'de la motte' and Initial = 'h' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Phospholipase A2, membrane associated
(Homo sapiens (Human)) | BDBM50055366
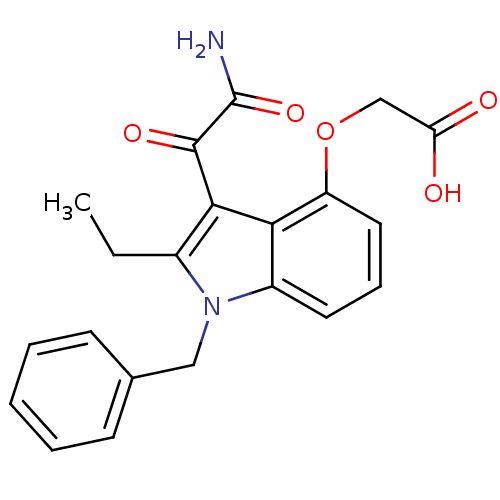
((3-Aminooxalyl-1-benzyl-2-ethyl-1H-indol-4-yloxy)-...)Show SMILES CCc1c(C(=O)C(N)=O)c2c(OCC(O)=O)cccc2n1Cc1ccccc1 Show InChI InChI=1S/C21H20N2O5/c1-2-14-19(20(26)21(22)27)18-15(9-6-10-16(18)28-12-17(24)25)23(14)11-13-7-4-3-5-8-13/h3-10H,2,11-12H2,1H3,(H2,22,27)(H,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-2A expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458617

(CHEMBL4215835)Show SMILES CC(CC(O)=O)c1cccc(n1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(7-17(26)27)13-3-2-4-16(24-13)25-14-9-12(29-19(20,21)22)6-5-11(14)8-15(25)18(23)28/h2-6,8-10H,7H2,1H3,(H2,23,28)(H,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458617

(CHEMBL4215835)Show SMILES CC(CC(O)=O)c1cccc(n1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(7-17(26)27)13-3-2-4-16(24-13)25-14-9-12(29-19(20,21)22)6-5-11(14)8-15(25)18(23)28/h2-6,8-10H,7H2,1H3,(H2,23,28)(H,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458614

(CHEMBL4210991)Show SMILES C[C@@H](CC(O)=O)c1cccc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O |r| Show InChI InChI=1S/C20H17F3N2O4/c1-11(7-18(26)27)12-3-2-4-14(8-12)25-16-10-15(29-20(21,22)23)6-5-13(16)9-17(25)19(24)28/h2-6,8-11H,7H2,1H3,(H2,24,28)(H,26,27)/t11-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366784
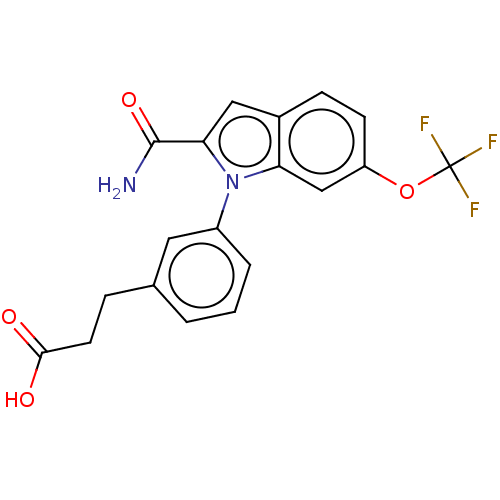
(CHEMBL4171084)Show SMILES NC(=O)c1cc2ccc(OC(F)(F)F)cc2n1-c1cccc(CCC(O)=O)c1 Show InChI InChI=1S/C19H15F3N2O4/c20-19(21,22)28-14-6-5-12-9-16(18(23)27)24(15(12)10-14)13-3-1-2-11(8-13)4-7-17(25)26/h1-3,5-6,8-10H,4,7H2,(H2,23,27)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366784

(CHEMBL4171084)Show SMILES NC(=O)c1cc2ccc(OC(F)(F)F)cc2n1-c1cccc(CCC(O)=O)c1 Show InChI InChI=1S/C19H15F3N2O4/c20-19(21,22)28-14-6-5-12-9-16(18(23)27)24(15(12)10-14)13-3-1-2-11(8-13)4-7-17(25)26/h1-3,5-6,8-10H,4,7H2,(H2,23,27)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458605
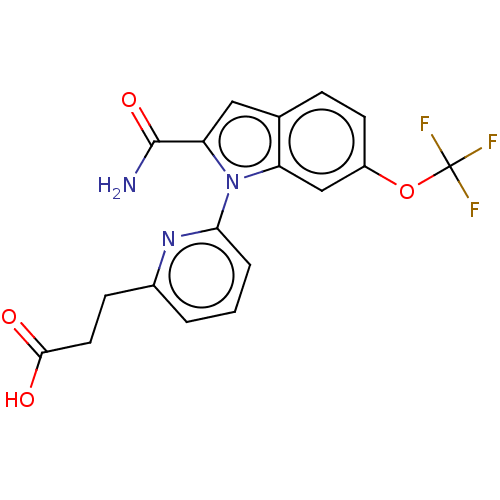
(CHEMBL4217510)Show SMILES NC(=O)c1cc2ccc(OC(F)(F)F)cc2n1-c1cccc(CCC(O)=O)n1 Show InChI InChI=1S/C18H14F3N3O4/c19-18(20,21)28-12-6-4-10-8-14(17(22)27)24(13(10)9-12)15-3-1-2-11(23-15)5-7-16(25)26/h1-4,6,8-9H,5,7H2,(H2,22,27)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458612
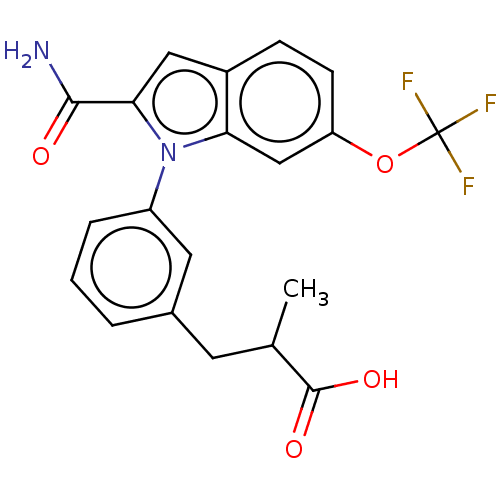
(CHEMBL4214052)Show SMILES CC(Cc1cccc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C20H17F3N2O4/c1-11(19(27)28)7-12-3-2-4-14(8-12)25-16-10-15(29-20(21,22)23)6-5-13(16)9-17(25)18(24)26/h2-6,8-11H,7H2,1H3,(H2,24,26)(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458612

(CHEMBL4214052)Show SMILES CC(Cc1cccc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C20H17F3N2O4/c1-11(19(27)28)7-12-3-2-4-14(8-12)25-16-10-15(29-20(21,22)23)6-5-13(16)9-17(25)18(24)26/h2-6,8-11H,7H2,1H3,(H2,24,26)(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458613
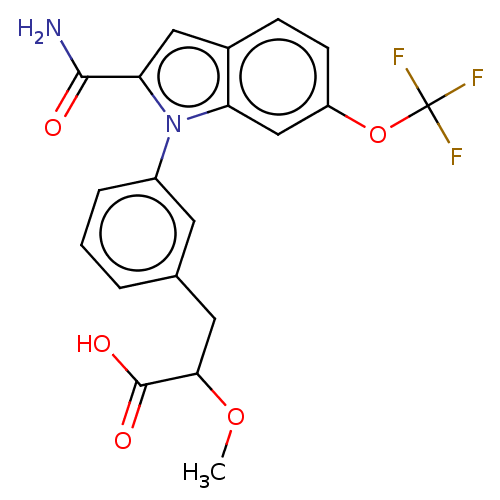
(CHEMBL4204172)Show SMILES COC(Cc1cccc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C20H17F3N2O5/c1-29-17(19(27)28)8-11-3-2-4-13(7-11)25-15-10-14(30-20(21,22)23)6-5-12(15)9-16(25)18(24)26/h2-7,9-10,17H,8H2,1H3,(H2,24,26)(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458613

(CHEMBL4204172)Show SMILES COC(Cc1cccc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C20H17F3N2O5/c1-29-17(19(27)28)8-11-3-2-4-13(7-11)25-15-10-14(30-20(21,22)23)6-5-12(15)9-16(25)18(24)26/h2-7,9-10,17H,8H2,1H3,(H2,24,26)(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50055366

((3-Aminooxalyl-1-benzyl-2-ethyl-1H-indol-4-yloxy)-...)Show SMILES CCc1c(C(=O)C(N)=O)c2c(OCC(O)=O)cccc2n1Cc1ccccc1 Show InChI InChI=1S/C21H20N2O5/c1-2-14-19(20(26)21(22)27)18-15(9-6-10-16(18)28-12-17(24)25)23(14)11-13-7-4-3-5-8-13/h3-10H,2,11-12H2,1H3,(H2,22,27)(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458617

(CHEMBL4215835)Show SMILES CC(CC(O)=O)c1cccc(n1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(7-17(26)27)13-3-2-4-16(24-13)25-14-9-12(29-19(20,21)22)6-5-11(14)8-15(25)18(23)28/h2-6,8-10H,7H2,1H3,(H2,23,28)(H,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 42 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 using human HDL as substrate pretreated for 20 mins followed by substrate addition and measured after 60 min... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366811
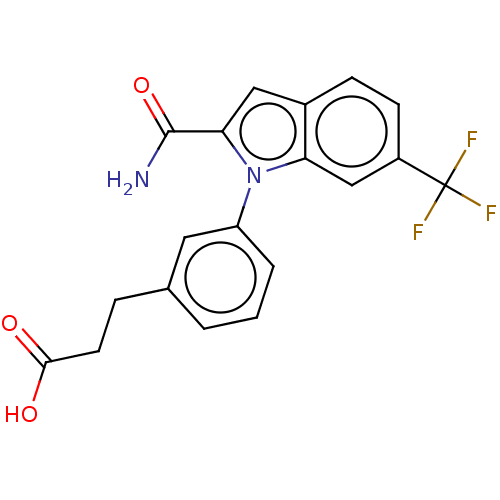
(CHEMBL4176544)Show SMILES NC(=O)c1cc2ccc(cc2n1-c1cccc(CCC(O)=O)c1)C(F)(F)F Show InChI InChI=1S/C19H15F3N2O3/c20-19(21,22)13-6-5-12-9-16(18(23)27)24(15(12)10-13)14-3-1-2-11(8-14)4-7-17(25)26/h1-3,5-6,8-10H,4,7H2,(H2,23,27)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 43 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Phospholipase A2, membrane associated
(Homo sapiens (Human)) | BDBM50458608
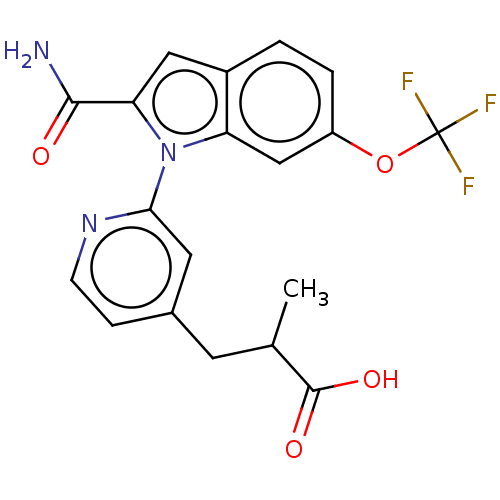
(CHEMBL4205008)Show SMILES CC(Cc1ccnc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(18(27)28)6-11-4-5-24-16(7-11)25-14-9-13(29-19(20,21)22)3-2-12(14)8-15(25)17(23)26/h2-5,7-10H,6H2,1H3,(H2,23,26)(H,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 44 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-2A (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Phospholipase A2, membrane associated
(Homo sapiens (Human)) | BDBM50458608

(CHEMBL4205008)Show SMILES CC(Cc1ccnc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(18(27)28)6-11-4-5-24-16(7-11)25-14-9-13(29-19(20,21)22)3-2-12(14)8-15(25)17(23)26/h2-5,7-10H,6H2,1H3,(H2,23,26)(H,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 44 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-2A (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Phospholipase A2, membrane associated
(Homo sapiens (Human)) | BDBM50458612

(CHEMBL4214052)Show SMILES CC(Cc1cccc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C20H17F3N2O4/c1-11(19(27)28)7-12-3-2-4-14(8-12)25-16-10-15(29-20(21,22)23)6-5-13(16)9-17(25)18(24)26/h2-6,8-11H,7H2,1H3,(H2,24,26)(H,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 57 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-2A (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Phospholipase A2, membrane associated
(Homo sapiens (Human)) | BDBM50458612

(CHEMBL4214052)Show SMILES CC(Cc1cccc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C20H17F3N2O4/c1-11(19(27)28)7-12-3-2-4-14(8-12)25-16-10-15(29-20(21,22)23)6-5-13(16)9-17(25)18(24)26/h2-6,8-11H,7H2,1H3,(H2,24,26)(H,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 57 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-2A (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458606
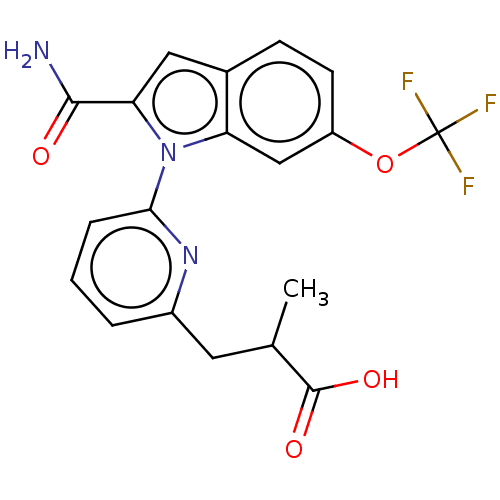
(CHEMBL4205511)Show SMILES CC(Cc1cccc(n1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(18(27)28)7-12-3-2-4-16(24-12)25-14-9-13(29-19(20,21)22)6-5-11(14)8-15(25)17(23)26/h2-6,8-10H,7H2,1H3,(H2,23,26)(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 65 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458606

(CHEMBL4205511)Show SMILES CC(Cc1cccc(n1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(18(27)28)7-12-3-2-4-16(24-12)25-14-9-13(29-19(20,21)22)6-5-11(14)8-15(25)17(23)26/h2-6,8-10H,7H2,1H3,(H2,23,26)(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 65 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366983
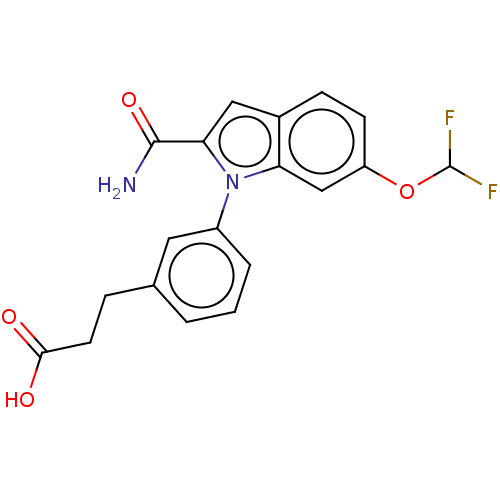
(CHEMBL4160483)Show SMILES NC(=O)c1cc2ccc(OC(F)F)cc2n1-c1cccc(CCC(O)=O)c1 Show InChI InChI=1S/C19H16F2N2O4/c20-19(21)27-14-6-5-12-9-16(18(22)26)23(15(12)10-14)13-3-1-2-11(8-13)4-7-17(24)25/h1-3,5-6,8-10,19H,4,7H2,(H2,22,26)(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 68 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Mus musculus) | BDBM50458617

(CHEMBL4215835)Show SMILES CC(CC(O)=O)c1cccc(n1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(7-17(26)27)13-3-2-4-16(24-13)25-14-9-12(29-19(20,21)22)6-5-11(14)8-15(25)18(23)28/h2-6,8-10H,7H2,1H3,(H2,23,28)(H,26,27) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 75 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of mouse sPLA2-10 |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366807

(CHEMBL4172222)Show InChI InChI=1S/C30H47N5O15/c1-11(2)5-4-6-16(39)33-20-23(43)21(41)14(47-29(20)50-28-19(32-12(3)37)22(42)18(31)15(10-36)48-28)9-13(38)26-24(44)25(45)27(49-26)35-8-7-17(40)34-30(35)46/h4,6-8,11,13-15,18-29,36,38,41-45H,5,9-10,31H2,1-3H3,(H,32,37)(H,33,39)(H,34,40,46)/b6-4+/t13?,14-,15-,18-,19-,20-,21+,22+,23-,24+,25-,26-,27-,28-,29+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Phospholipase A2, membrane associated
(Homo sapiens (Human)) | BDBM50458606

(CHEMBL4205511)Show SMILES CC(Cc1cccc(n1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(18(27)28)7-12-3-2-4-16(24-12)25-14-9-13(29-19(20,21)22)6-5-11(14)8-15(25)17(23)26/h2-6,8-10H,7H2,1H3,(H2,23,26)(H,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 92 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-2A (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Phospholipase A2, membrane associated
(Homo sapiens (Human)) | BDBM50458606

(CHEMBL4205511)Show SMILES CC(Cc1cccc(n1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(18(27)28)7-12-3-2-4-16(24-12)25-14-9-13(29-19(20,21)22)6-5-11(14)8-15(25)17(23)26/h2-6,8-10H,7H2,1H3,(H2,23,26)(H,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 92 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-2A (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366839
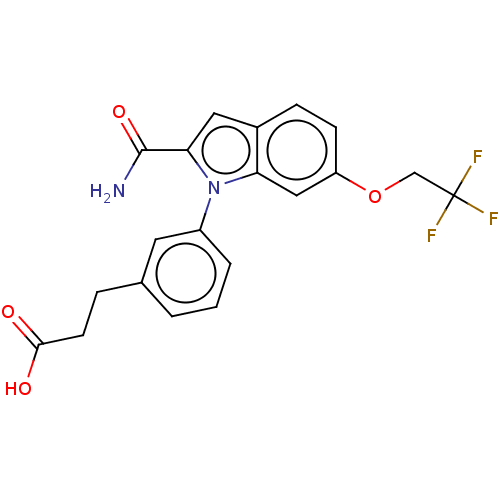
(CHEMBL4175583)Show SMILES NC(=O)c1cc2ccc(OCC(F)(F)F)cc2n1-c1cccc(CCC(O)=O)c1 Show InChI InChI=1S/C20H17F3N2O4/c21-20(22,23)11-29-15-6-5-13-9-17(19(24)28)25(16(13)10-15)14-3-1-2-12(8-14)4-7-18(26)27/h1-3,5-6,8-10H,4,7,11H2,(H2,24,28)(H,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 93 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Mus musculus) | BDBM50366784

(CHEMBL4171084)Show SMILES NC(=O)c1cc2ccc(OC(F)(F)F)cc2n1-c1cccc(CCC(O)=O)c1 Show InChI InChI=1S/C19H15F3N2O4/c20-19(21,22)28-14-6-5-12-9-16(18(23)27)24(15(12)10-14)13-3-1-2-11(8-13)4-7-17(25)26/h1-3,5-6,8-10H,4,7H2,(H2,23,27)(H,25,26) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 96 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of mouse sPLA2-10 using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substrate additio... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366840
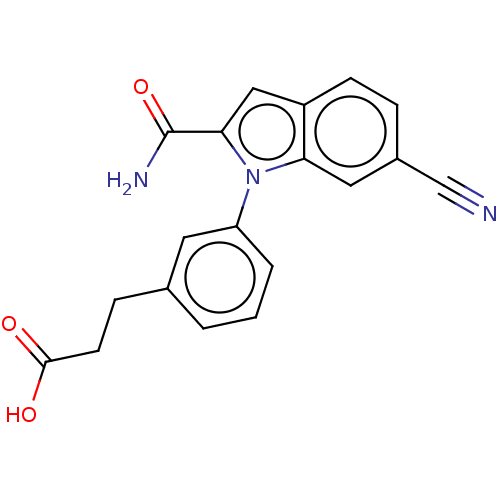
(CHEMBL4174522)Show SMILES NC(=O)c1cc2ccc(cc2n1-c1cccc(CCC(O)=O)c1)C#N Show InChI InChI=1S/C19H15N3O3/c20-11-13-4-6-14-10-17(19(21)25)22(16(14)9-13)15-3-1-2-12(8-15)5-7-18(23)24/h1-4,6,8-10H,5,7H2,(H2,21,25)(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458614

(CHEMBL4210991)Show SMILES C[C@@H](CC(O)=O)c1cccc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O |r| Show InChI InChI=1S/C20H17F3N2O4/c1-11(7-18(26)27)12-3-2-4-14(8-12)25-16-10-15(29-20(21,22)23)6-5-13(16)9-17(25)19(24)28/h2-6,8-11H,7H2,1H3,(H2,24,28)(H,26,27)/t11-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 using human HDL as substrate pretreated for 20 mins followed by substrate addition and measured after 60 min... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458609
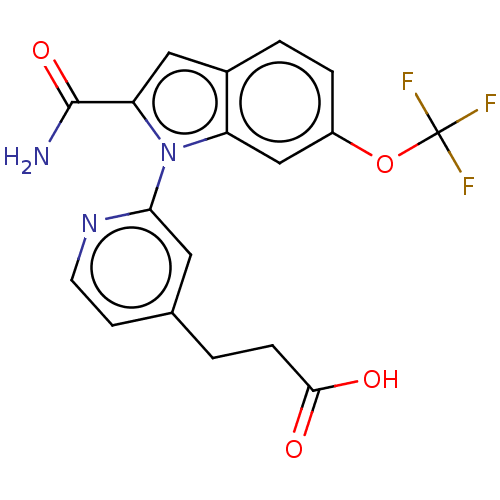
(CHEMBL4213094)Show SMILES NC(=O)c1cc2ccc(OC(F)(F)F)cc2n1-c1cc(CCC(O)=O)ccn1 Show InChI InChI=1S/C18H14F3N3O4/c19-18(20,21)28-12-3-2-11-8-14(17(22)27)24(13(11)9-12)15-7-10(5-6-23-15)1-4-16(25)26/h2-3,5-9H,1,4H2,(H2,22,27)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Phospholipase A2 group V
(Homo sapiens (Human)) | BDBM50055366

((3-Aminooxalyl-1-benzyl-2-ethyl-1H-indol-4-yloxy)-...)Show SMILES CCc1c(C(=O)C(N)=O)c2c(OCC(O)=O)cccc2n1Cc1ccccc1 Show InChI InChI=1S/C21H20N2O5/c1-2-14-19(20(26)21(22)27)18-15(9-6-10-16(18)28-12-17(24)25)23(14)11-13-7-4-3-5-8-13/h3-10H,2,11-12H2,1H3,(H2,22,27)(H,24,25) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 124 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-5 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate p... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Phospholipase A2, membrane associated
(Homo sapiens (Human)) | BDBM50458613

(CHEMBL4204172)Show SMILES COC(Cc1cccc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C20H17F3N2O5/c1-29-17(19(27)28)8-11-3-2-4-13(7-11)25-15-10-14(30-20(21,22)23)6-5-12(15)9-16(25)18(24)26/h2-7,9-10,17H,8H2,1H3,(H2,24,26)(H,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-2A (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458608

(CHEMBL4205008)Show SMILES CC(Cc1ccnc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(18(27)28)6-11-4-5-24-16(7-11)25-14-9-13(29-19(20,21)22)3-2-12(14)8-15(25)17(23)26/h2-5,7-10H,6H2,1H3,(H2,23,26)(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458608

(CHEMBL4205008)Show SMILES CC(Cc1ccnc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(18(27)28)6-11-4-5-24-16(7-11)25-14-9-13(29-19(20,21)22)3-2-12(14)8-15(25)17(23)26/h2-5,7-10H,6H2,1H3,(H2,23,26)(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Phospholipase A2, membrane associated
(Homo sapiens (Human)) | BDBM50458613

(CHEMBL4204172)Show SMILES COC(Cc1cccc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C20H17F3N2O5/c1-29-17(19(27)28)8-11-3-2-4-13(7-11)25-15-10-14(30-20(21,22)23)6-5-12(15)9-16(25)18(24)26/h2-7,9-10,17H,8H2,1H3,(H2,24,26)(H,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-2A (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458613

(CHEMBL4204172)Show SMILES COC(Cc1cccc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C20H17F3N2O5/c1-29-17(19(27)28)8-11-3-2-4-13(7-11)25-15-10-14(30-20(21,22)23)6-5-12(15)9-16(25)18(24)26/h2-7,9-10,17H,8H2,1H3,(H2,24,26)(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 using human HDL as substrate pretreated for 20 mins followed by substrate addition and measured after 60 min... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50055366

((3-Aminooxalyl-1-benzyl-2-ethyl-1H-indol-4-yloxy)-...)Show SMILES CCc1c(C(=O)C(N)=O)c2c(OCC(O)=O)cccc2n1Cc1ccccc1 Show InChI InChI=1S/C21H20N2O5/c1-2-14-19(20(26)21(22)27)18-15(9-6-10-16(18)28-12-17(24)25)23(14)11-13-7-4-3-5-8-13/h3-10H,2,11-12H2,1H3,(H2,22,27)(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using HDL as substrate pretreated for 20 mins followed by substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366905
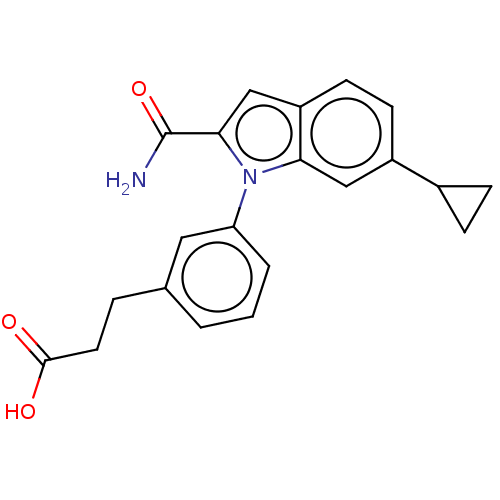
(CHEMBL4173359)Show SMILES NC(=O)c1cc2ccc(cc2n1-c1cccc(CCC(O)=O)c1)C1CC1 Show InChI InChI=1S/C21H20N2O3/c22-21(26)19-12-16-8-7-15(14-5-6-14)11-18(16)23(19)17-3-1-2-13(10-17)4-9-20(24)25/h1-3,7-8,10-12,14H,4-6,9H2,(H2,22,26)(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458615
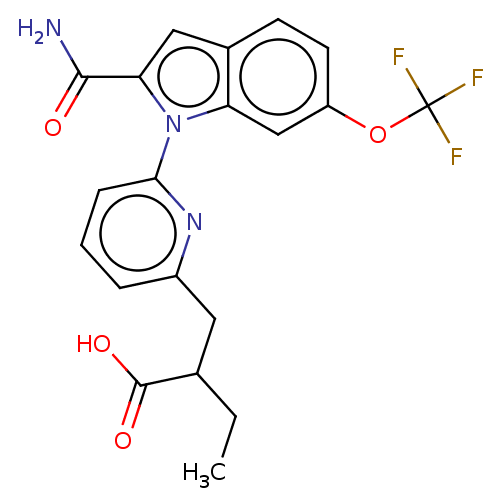
(CHEMBL4203027)Show SMILES CCC(Cc1cccc(n1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C20H18F3N3O4/c1-2-11(19(28)29)8-13-4-3-5-17(25-13)26-15-10-14(30-20(21,22)23)7-6-12(15)9-16(26)18(24)27/h3-7,9-11H,2,8H2,1H3,(H2,24,27)(H,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Phospholipase A2, membrane associated
(Homo sapiens (Human)) | BDBM50458615

(CHEMBL4203027)Show SMILES CCC(Cc1cccc(n1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C20H18F3N3O4/c1-2-11(19(28)29)8-13-4-3-5-17(25-13)26-15-10-14(30-20(21,22)23)7-6-12(15)9-16(26)18(24)27/h3-7,9-11H,2,8H2,1H3,(H2,24,27)(H,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-2A (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458615

(CHEMBL4203027)Show SMILES CCC(Cc1cccc(n1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C20H18F3N3O4/c1-2-11(19(28)29)8-13-4-3-5-17(25-13)26-15-10-14(30-20(21,22)23)7-6-12(15)9-16(26)18(24)27/h3-7,9-11H,2,8H2,1H3,(H2,24,27)(H,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366889
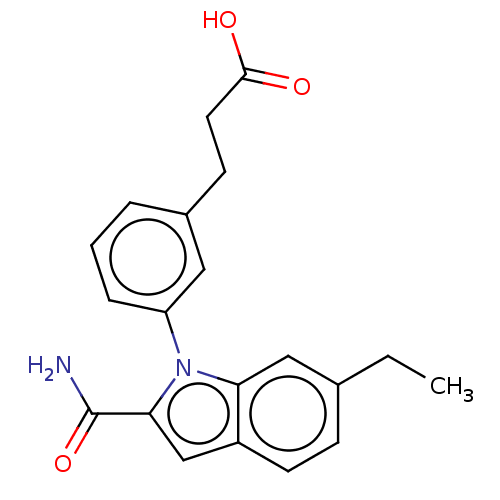
(CHEMBL4175643)Show SMILES CCc1ccc2cc(C(N)=O)n(-c3cccc(CCC(O)=O)c3)c2c1 Show InChI InChI=1S/C20H22F2N4O/c1-19-9-8-14-13-5-3-12(20(21,22)18-23-25-26-24-18)10-11(13)2-4-15(14)16(19)6-7-17(19)27/h3,5,10,14-16H,2,4,6-9H2,1H3,(H,23,24,25,26)/t14-,15-,16+,19+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Phospholipase A2, membrane associated
(Homo sapiens (Human)) | BDBM50458615

(CHEMBL4203027)Show SMILES CCC(Cc1cccc(n1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C20H18F3N3O4/c1-2-11(19(28)29)8-13-4-3-5-17(25-13)26-15-10-14(30-20(21,22)23)7-6-12(15)9-16(26)18(24)27/h3-7,9-11H,2,8H2,1H3,(H2,24,27)(H,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-2A (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Phospholipase A2 group V
(Homo sapiens (Human)) | BDBM50458608

(CHEMBL4205008)Show SMILES CC(Cc1ccnc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(18(27)28)6-11-4-5-24-16(7-11)25-14-9-13(29-19(20,21)22)3-2-12(14)8-15(25)17(23)26/h2-5,7-10H,6H2,1H3,(H2,23,26)(H,27,28) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-5 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substra... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Phospholipase A2 group V
(Homo sapiens (Human)) | BDBM50458608

(CHEMBL4205008)Show SMILES CC(Cc1ccnc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(18(27)28)6-11-4-5-24-16(7-11)25-14-9-13(29-19(20,21)22)3-2-12(14)8-15(25)17(23)26/h2-5,7-10H,6H2,1H3,(H2,23,26)(H,27,28) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-5 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substra... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458619
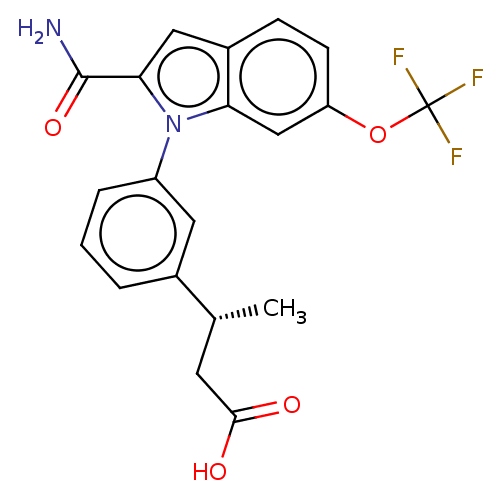
(CHEMBL4207266)Show SMILES C[C@H](CC(O)=O)c1cccc(c1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O |r| Show InChI InChI=1S/C20H17F3N2O4/c1-11(7-18(26)27)12-3-2-4-14(8-12)25-16-10-15(29-20(21,22)23)6-5-13(16)9-17(25)19(24)28/h2-6,8-11H,7H2,1H3,(H2,24,28)(H,26,27)/t11-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366876
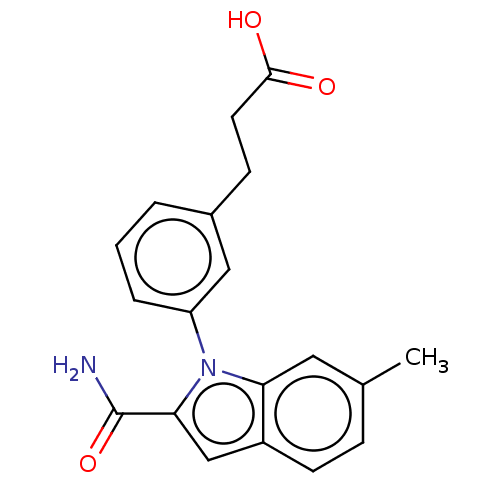
(CHEMBL4171797)Show SMILES Cc1ccc2cc(C(N)=O)n(-c3cccc(CCC(O)=O)c3)c2c1 Show InChI InChI=1S/C19H18N2O3/c1-12-5-7-14-11-17(19(20)24)21(16(14)9-12)15-4-2-3-13(10-15)6-8-18(22)23/h2-5,7,9-11H,6,8H2,1H3,(H2,20,24)(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 230 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458618
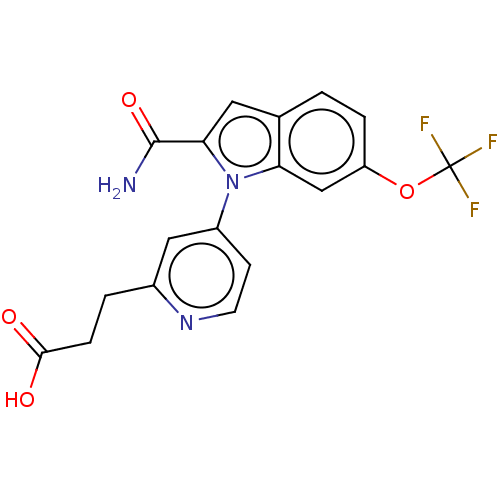
(CHEMBL4206163)Show SMILES NC(=O)c1cc2ccc(OC(F)(F)F)cc2n1-c1ccnc(CCC(O)=O)c1 Show InChI InChI=1S/C18H14F3N3O4/c19-18(20,21)28-13-3-1-10-7-15(17(22)27)24(14(10)9-13)12-5-6-23-11(8-12)2-4-16(25)26/h1,3,5-9H,2,4H2,(H2,22,27)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 240 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of sPLA2-10 (unknown origin) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substr... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458605

(CHEMBL4217510)Show SMILES NC(=O)c1cc2ccc(OC(F)(F)F)cc2n1-c1cccc(CCC(O)=O)n1 Show InChI InChI=1S/C18H14F3N3O4/c19-18(20,21)28-12-6-4-10-8-14(17(22)27)24(13(10)9-12)15-3-1-2-11(23-15)5-7-16(25)26/h1-4,6,8-9H,5,7H2,(H2,22,27)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 using human HDL as substrate pretreated for 20 mins followed by substrate addition and measured after 60 min... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50458606

(CHEMBL4205511)Show SMILES CC(Cc1cccc(n1)-n1c(cc2ccc(OC(F)(F)F)cc12)C(N)=O)C(O)=O Show InChI InChI=1S/C19H16F3N3O4/c1-10(18(27)28)7-12-3-2-4-16(24-12)25-14-9-13(29-19(20,21)22)6-5-11(14)8-15(25)17(23)26/h2-6,8-10H,7H2,1H3,(H2,23,26)(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 using human HDL as substrate pretreated for 20 mins followed by substrate addition and measured after 60 min... |
ACS Med Chem Lett 9: 600-605 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00507
BindingDB Entry DOI: 10.7270/Q2Z32272 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data