 Found 80 hits with Last Name = 'qvortrup' and Initial = 'k'
Found 80 hits with Last Name = 'qvortrup' and Initial = 'k' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50215115
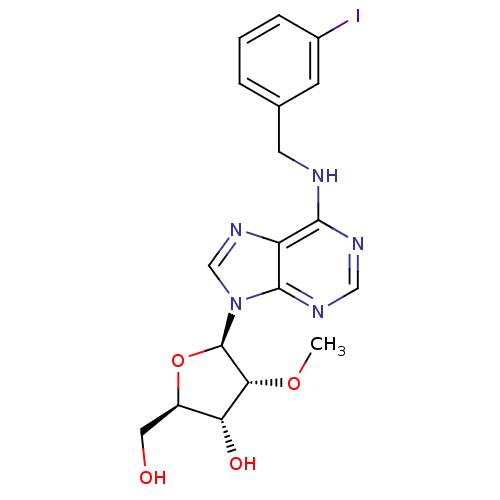
(CHEMBL428377 | N6-(3-Iodobenzyl)-2'-O-methyladenos...)Show SMILES CO[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1n1cnc2c(NCc3cccc(I)c3)ncnc12 Show InChI InChI=1S/C18H20IN5O4/c1-27-15-14(26)12(7-25)28-18(15)24-9-23-13-16(21-8-22-17(13)24)20-6-10-3-2-4-11(19)5-10/h2-5,8-9,12,14-15,18,25-26H,6-7H2,1H3,(H,20,21,22)/t12-,14-,15-,18-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50215111
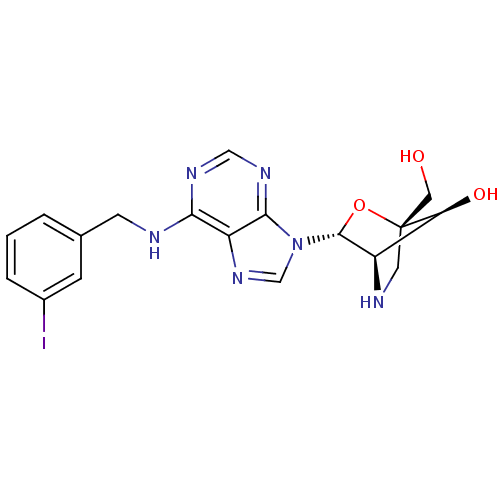
((1S,3R,4R,7S)-7-hydroxy-1-hydroxymethyl-3-(N6-(3-i...)Show SMILES OC[C@]12CN[C@H]([C@@H]1O)[C@@H](O2)n1cnc2c(NCc3cccc(I)c3)ncnc12 Show InChI InChI=1S/C18H19IN6O3/c19-11-3-1-2-10(4-11)5-20-15-13-16(23-8-22-15)25(9-24-13)17-12-14(27)18(7-26,28-17)6-21-12/h1-4,8-9,12,14,17,21,26-27H,5-7H2,(H,20,22,23)/t12-,14+,17-,18-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50215110
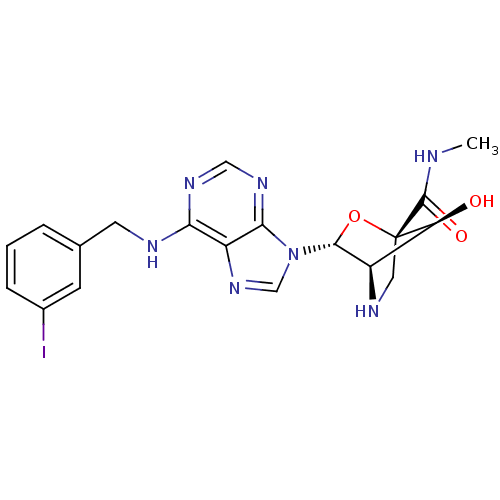
((1R,3R,4R,7S)-(7-hydroxy-3-(N6-(3-iodobenzyl)-aden...)Show SMILES CNC(=O)[C@]12CN[C@H]([C@@H]1O)[C@@H](O2)n1cnc2c(NCc3cccc(I)c3)ncnc12 Show InChI InChI=1S/C19H20IN7O3/c1-21-18(29)19-7-23-12(14(19)28)17(30-19)27-9-26-13-15(24-8-25-16(13)27)22-6-10-3-2-4-11(20)5-10/h2-5,8-9,12,14,17,23,28H,6-7H2,1H3,(H,21,29)(H,22,24,25)/t12-,14+,17-,19+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50144945
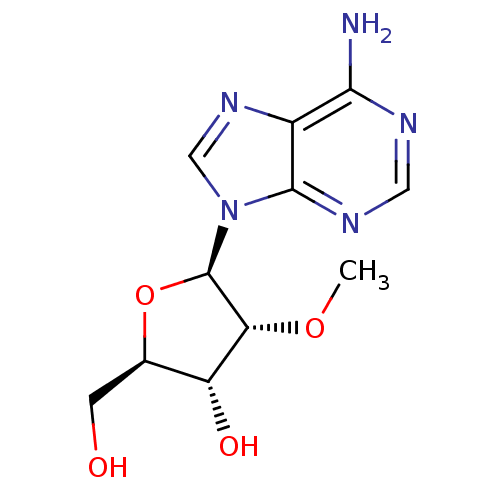
((2R,3R,4R,5R)-2-Hydroxymethyl-4-methoxy-5-(6-methy...)Show SMILES CO[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1n1cnc2c(N)ncnc12 Show InChI InChI=1S/C11H15N5O4/c1-19-8-7(18)5(2-17)20-11(8)16-4-15-6-9(12)13-3-14-10(6)16/h3-5,7-8,11,17-18H,2H2,1H3,(H2,12,13,14)/t5-,7-,8-,11-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50215118
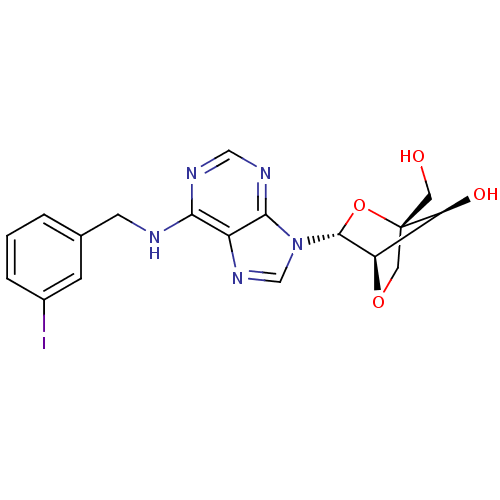
((1S,3R,4R,7S)-7-hydroxy-1-hydroxymethyl-3-(N6-(3-i...)Show SMILES OC[C@]12CO[C@H]([C@@H]1O)[C@@H](O2)n1cnc2c(NCc3cccc(I)c3)ncnc12 Show InChI InChI=1S/C18H18IN5O4/c19-11-3-1-2-10(4-11)5-20-15-12-16(22-8-21-15)24(9-23-12)17-13-14(26)18(6-25,28-17)7-27-13/h1-4,8-9,13-14,17,25-26H,5-7H2,(H,20,21,22)/t13-,14+,17-,18+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50215109

((1R,3R,4R,7S)-(7-hydroxy-3-(adenin-9-yl)-2-oxa-5-a...)Show SMILES CNC(=O)[C@]12CN[C@H]([C@@H]1O)[C@@H](O2)n1cnc2c(N)ncnc12 Show InChI InChI=1S/C12H15N7O3/c1-14-11(21)12-2-15-5(7(12)20)10(22-12)19-4-18-6-8(13)16-3-17-9(6)19/h3-5,7,10,15,20H,2H2,1H3,(H,14,21)(H2,13,16,17)/t5-,7+,10-,12+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM50596272
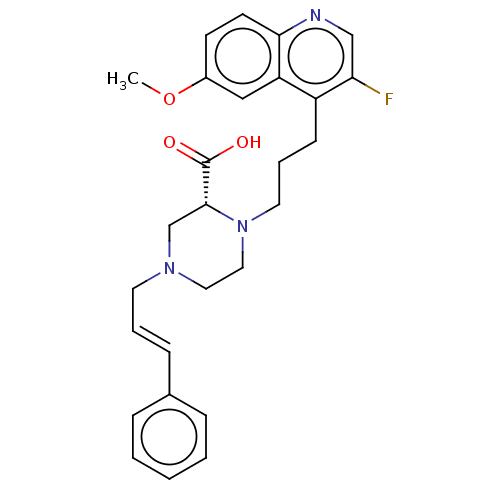
(CHEMBL5199813)Show SMILES COc1ccc2ncc(F)c(CCCN3CCN(C\C=C\c4ccccc4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM21690
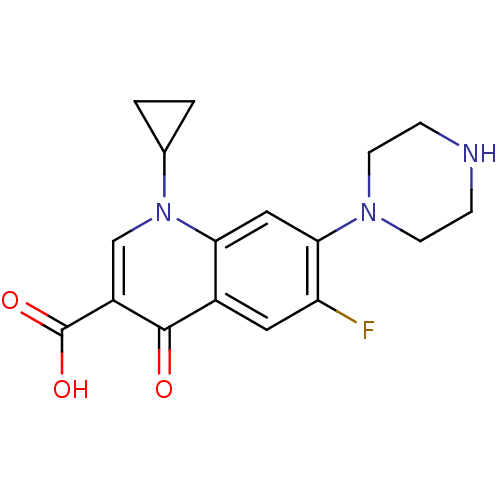
(1-cyclopropyl-6-fluoro-4-oxo-7-(piperazin-1-yl)-1,...)Show InChI InChI=1S/C17H18FN3O3/c18-13-7-11-14(8-15(13)20-5-3-19-4-6-20)21(10-1-2-10)9-12(16(11)22)17(23)24/h7-10,19H,1-6H2,(H,23,24) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 230 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM50596263

(CHEMBL5207657)Show SMILES COc1ccc2ncc(F)c(\C=C/CN3CCN(CCSc4cccs4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 280 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM50596278
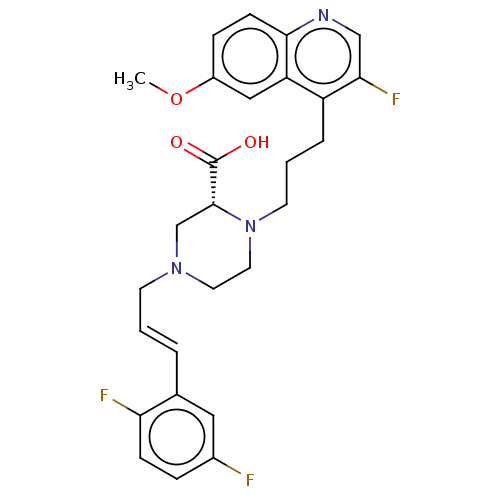
(CHEMBL5178214)Show SMILES COc1ccc2ncc(F)c(CCCN3CCN(C\C=C\c4cc(F)ccc4F)C[C@@H]3C(O)=O)c2c1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 280 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM50393503
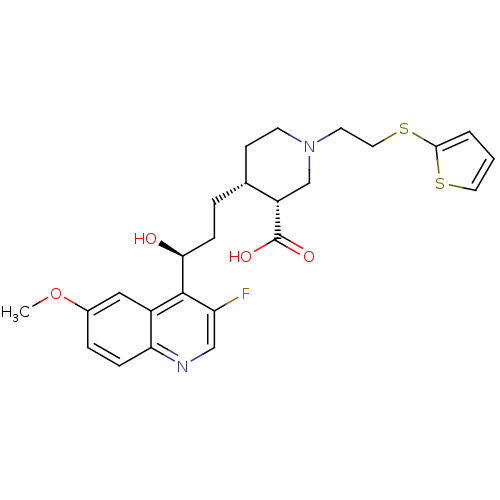
(CHEMBL2158050)Show SMILES COc1ccc2ncc(F)c([C@@H](O)CC[C@@H]3CCN(CCSc4cccs4)C[C@@H]3C(O)=O)c2c1 |r| Show InChI InChI=1S/C25H29FN2O4S2/c1-32-17-5-6-21-18(13-17)24(20(26)14-27-21)22(29)7-4-16-8-9-28(15-19(16)25(30)31)10-12-34-23-3-2-11-33-23/h2-3,5-6,11,13-14,16,19,22,29H,4,7-10,12,15H2,1H3,(H,30,31)/t16-,19+,22+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM50596275
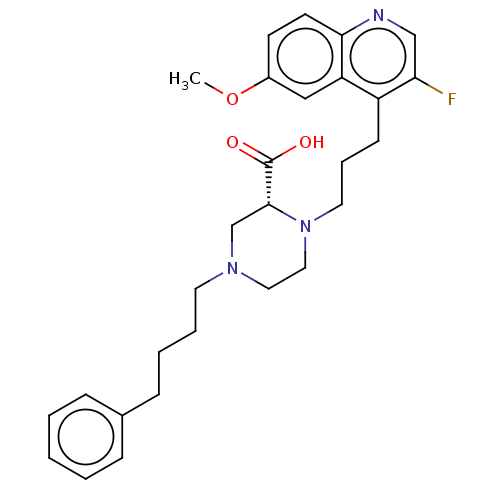
(CHEMBL5176348)Show SMILES COc1ccc2ncc(F)c(CCCN3CCN(CCCCc4ccccc4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 370 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM50596261
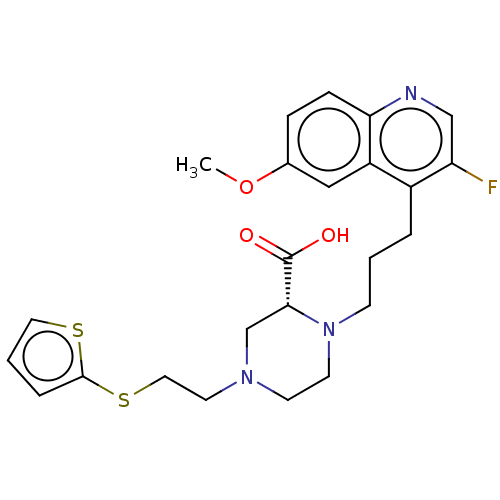
(CHEMBL5176134)Show SMILES COc1ccc2ncc(F)c(CCCN3CCN(CCSc4cccs4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 370 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM50596271
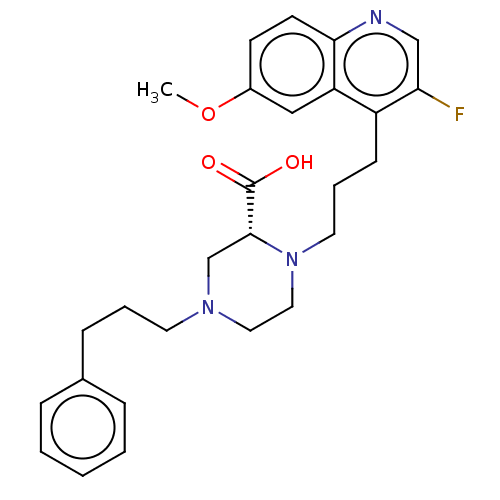
(CHEMBL5176046)Show SMILES COc1ccc2ncc(F)c(CCCN3CCN(CCCc4ccccc4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 460 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM50596264

(CHEMBL5206355)Show SMILES COc1ccc2ncc(F)c(C#CCN3CCN(CCSc4cccs4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 480 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM50596274

(CHEMBL5204986)Show SMILES COc1ccc2ncc(F)c(CCCN3CCN(CCc4ccccc4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 540 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM50596262
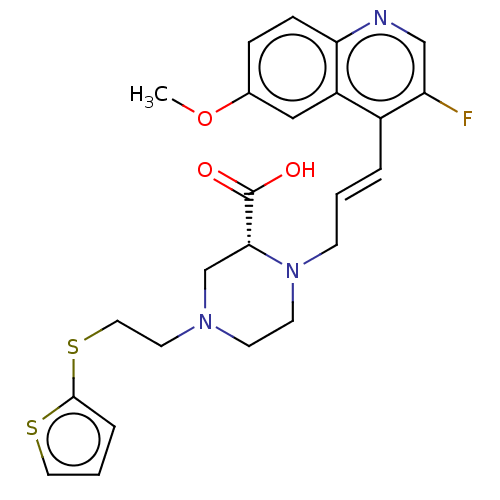
(CHEMBL5176331)Show SMILES COc1ccc2ncc(F)c(\C=C\CN3CCN(CCSc4cccs4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 680 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA topoisomerase 4 subunit A/B
(Staphylococcus aureus) | BDBM50596262

(CHEMBL5176331)Show SMILES COc1ccc2ncc(F)c(\C=C\CN3CCN(CCSc4cccs4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.81E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA topoisomerase 4 subunit A/B
(Staphylococcus aureus) | BDBM50596278

(CHEMBL5178214)Show SMILES COc1ccc2ncc(F)c(CCCN3CCN(C\C=C\c4cc(F)ccc4F)C[C@@H]3C(O)=O)c2c1 |r| | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA topoisomerase 4 subunit A/B
(Staphylococcus aureus) | BDBM50596264

(CHEMBL5206355)Show SMILES COc1ccc2ncc(F)c(C#CCN3CCN(CCSc4cccs4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.47E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50215110

((1R,3R,4R,7S)-(7-hydroxy-3-(N6-(3-iodobenzyl)-aden...)Show SMILES CNC(=O)[C@]12CN[C@H]([C@@H]1O)[C@@H](O2)n1cnc2c(NCc3cccc(I)c3)ncnc12 Show InChI InChI=1S/C19H20IN7O3/c1-21-18(29)19-7-23-12(14(19)28)17(30-19)27-9-26-13-15(24-8-25-16(13)27)22-6-10-3-2-4-11(20)5-10/h2-5,8-9,12,14,17,23,28H,6-7H2,1H3,(H,21,29)(H,22,24,25)/t12-,14+,17-,19+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A1 receptor |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50144945

((2R,3R,4R,5R)-2-Hydroxymethyl-4-methoxy-5-(6-methy...)Show SMILES CO[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1n1cnc2c(N)ncnc12 Show InChI InChI=1S/C11H15N5O4/c1-19-8-7(18)5(2-17)20-11(8)16-4-15-6-9(12)13-3-14-10(6)16/h3-5,7-8,11,17-18H,2H2,1H3,(H2,12,13,14)/t5-,7-,8-,11-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A1 receptor |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50215116
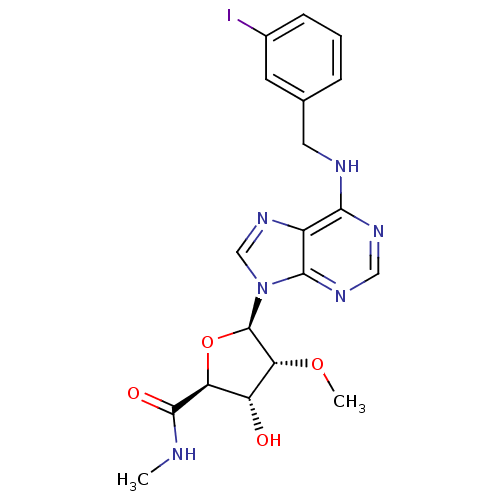
(CHEMBL393362 | N6-(3-iodobenzyl)-2'-O-methyladenos...)Show SMILES CNC(=O)[C@H]1O[C@H]([C@H](OC)[C@@H]1O)n1cnc2c(NCc3cccc(I)c3)ncnc12 Show InChI InChI=1S/C19H21IN6O4/c1-21-18(28)14-13(27)15(29-2)19(30-14)26-9-25-12-16(23-8-24-17(12)26)22-7-10-4-3-5-11(20)6-10/h3-6,8-9,13-15,19,27H,7H2,1-2H3,(H,21,28)(H,22,23,24)/t13-,14+,15-,19-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A1 receptor |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50215115

(CHEMBL428377 | N6-(3-Iodobenzyl)-2'-O-methyladenos...)Show SMILES CO[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1n1cnc2c(NCc3cccc(I)c3)ncnc12 Show InChI InChI=1S/C18H20IN5O4/c1-27-15-14(26)12(7-25)28-18(15)24-9-23-13-16(21-8-22-17(13)24)20-6-10-3-2-4-11(19)5-10/h2-5,8-9,12,14-15,18,25-26H,6-7H2,1H3,(H,20,21,22)/t12-,14-,15-,18-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A1 receptor |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50215111

((1S,3R,4R,7S)-7-hydroxy-1-hydroxymethyl-3-(N6-(3-i...)Show SMILES OC[C@]12CN[C@H]([C@@H]1O)[C@@H](O2)n1cnc2c(NCc3cccc(I)c3)ncnc12 Show InChI InChI=1S/C18H19IN6O3/c19-11-3-1-2-10(4-11)5-20-15-13-16(23-8-22-15)25(9-24-13)17-12-14(27)18(7-26,28-17)6-21-12/h1-4,8-9,12,14,17,21,26-27H,5-7H2,(H,20,22,23)/t12-,14+,17-,18-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A1 receptor |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
DNA topoisomerase 4 subunit A/B
(Staphylococcus aureus) | BDBM50596272

(CHEMBL5199813)Show SMILES COc1ccc2ncc(F)c(CCCN3CCN(C\C=C\c4ccccc4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.36E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA topoisomerase 4 subunit A/B
(Staphylococcus aureus) | BDBM50596275

(CHEMBL5176348)Show SMILES COc1ccc2ncc(F)c(CCCN3CCN(CCCCc4ccccc4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.82E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM21690

(1-cyclopropyl-6-fluoro-4-oxo-7-(piperazin-1-yl)-1,...)Show InChI InChI=1S/C17H18FN3O3/c18-13-7-11-14(8-15(13)20-5-3-19-4-6-20)21(10-1-2-10)9-12(16(11)22)17(23)24/h7-10,19H,1-6H2,(H,23,24) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 3.84E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM50393503

(CHEMBL2158050)Show SMILES COc1ccc2ncc(F)c([C@@H](O)CC[C@@H]3CCN(CCSc4cccs4)C[C@@H]3C(O)=O)c2c1 |r| Show InChI InChI=1S/C25H29FN2O4S2/c1-32-17-5-6-21-18(13-17)24(20(26)14-27-21)22(29)7-4-16-8-9-28(15-19(16)25(30)31)10-12-34-23-3-2-11-33-23/h2-3,5-6,11,13-14,16,19,22,29H,4,7-10,12,15H2,1H3,(H,30,31)/t16-,19+,22+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM50596261

(CHEMBL5176134)Show SMILES COc1ccc2ncc(F)c(CCCN3CCN(CCSc4cccs4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM50596278

(CHEMBL5178214)Show SMILES COc1ccc2ncc(F)c(CCCN3CCN(C\C=C\c4cc(F)ccc4F)C[C@@H]3C(O)=O)c2c1 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.46E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA topoisomerase 4 subunit A/B
(Staphylococcus aureus) | BDBM50596261

(CHEMBL5176134)Show SMILES COc1ccc2ncc(F)c(CCCN3CCN(CCSc4cccs4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.64E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA topoisomerase 4 subunit A/B
(Staphylococcus aureus) | BDBM50393503

(CHEMBL2158050)Show SMILES COc1ccc2ncc(F)c([C@@H](O)CC[C@@H]3CCN(CCSc4cccs4)C[C@@H]3C(O)=O)c2c1 |r| Show InChI InChI=1S/C25H29FN2O4S2/c1-32-17-5-6-21-18(13-17)24(20(26)14-27-21)22(29)7-4-16-8-9-28(15-19(16)25(30)31)10-12-34-23-3-2-11-33-23/h2-3,5-6,11,13-14,16,19,22,29H,4,7-10,12,15H2,1H3,(H,30,31)/t16-,19+,22+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA topoisomerase 4 subunit A/B
(Staphylococcus aureus) | BDBM21690

(1-cyclopropyl-6-fluoro-4-oxo-7-(piperazin-1-yl)-1,...)Show InChI InChI=1S/C17H18FN3O3/c18-13-7-11-14(8-15(13)20-5-3-19-4-6-20)21(10-1-2-10)9-12(16(11)22)17(23)24/h7-10,19H,1-6H2,(H,23,24) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9.16E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA topoisomerase 4 subunit A/B
(Staphylococcus aureus) | BDBM50596263

(CHEMBL5207657)Show SMILES COc1ccc2ncc(F)c(\C=C/CN3CCN(CCSc4cccs4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9.92E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA topoisomerase 4 subunit A/B
(Staphylococcus aureus) | BDBM50596271

(CHEMBL5176046)Show SMILES COc1ccc2ncc(F)c(CCCN3CCN(CCCc4ccccc4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.02E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA topoisomerase 4 subunit A/B
(Staphylococcus aureus) | BDBM50596274

(CHEMBL5204986)Show SMILES COc1ccc2ncc(F)c(CCCN3CCN(CCc4ccccc4)C[C@@H]3C(O)=O)c2c1 |r| | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.26E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM50596269

(CHEMBL5183842)Show SMILES COC(=O)[C@H]1CN(CCSc2cccs2)CCN1CCCc1c(F)cnc2ccc(OC)cc12 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.84E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50215112

((1R,3R,4R,7S)-3-(6-amino-purin-9-yl)-1-hydroxymeth...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@@]2(CO)CN[C@@H]1[C@@H]2O Show InChI InChI=1S/C11H14N6O3/c12-8-6-9(15-3-14-8)17(4-16-6)10-5-7(19)11(2-18,20-10)1-13-5/h3-5,7,10,13,18-19H,1-2H2,(H2,12,14,15)/t5-,7+,10-,11-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50215113
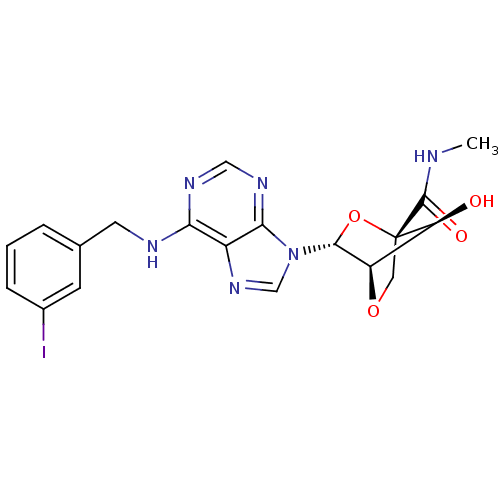
((1R,3R,4R,7S)-(7-hydroxy-3-(N6-(3-iodobenzyl)-aden...)Show SMILES CNC(=O)[C@]12CO[C@H]([C@@H]1O)[C@@H](O2)n1cnc2c(NCc3cccc(I)c3)ncnc12 Show InChI InChI=1S/C19H19IN6O4/c1-21-18(28)19-7-29-13(14(19)27)17(30-19)26-9-25-12-15(23-8-24-16(12)26)22-6-10-3-2-4-11(20)5-10/h2-5,8-9,13-14,17,27H,6-7H2,1H3,(H,21,28)(H,22,23,24)/t13-,14+,17-,19+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50215117

((1S,3R,4R,7S)-3-(6-amino-purin-9-yl)-1-hydroxymeth...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@@]2(CO)CO[C@@H]1[C@@H]2O Show InChI InChI=1S/C11H13N5O4/c12-8-5-9(14-3-13-8)16(4-15-5)10-6-7(18)11(1-17,20-10)2-19-6/h3-4,6-7,10,17-18H,1-2H2,(H2,12,13,14)/t6-,7+,10-,11+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50215114

((1R,3R,4R,7S)-(7-hydroxy-3-(adenin-9-yl)-2,5-dioxa...)Show SMILES CNC(=O)[C@]12CO[C@H]([C@@H]1O)[C@@H](O2)n1cnc2c(N)ncnc12 Show InChI InChI=1S/C12H14N6O4/c1-14-11(20)12-2-21-6(7(12)19)10(22-12)18-4-17-5-8(13)15-3-16-9(5)18/h3-4,6-7,10,19H,2H2,1H3,(H,14,20)(H2,13,15,16)/t6-,7+,10-,12+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM50596269

(CHEMBL5183842)Show SMILES COC(=O)[C@H]1CN(CCSc2cccs2)CCN1CCCc1c(F)cnc2ccc(OC)cc12 |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128499
BindingDB Entry DOI: 10.7270/Q2X06C35 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50215110

((1R,3R,4R,7S)-(7-hydroxy-3-(N6-(3-iodobenzyl)-aden...)Show SMILES CNC(=O)[C@]12CN[C@H]([C@@H]1O)[C@@H](O2)n1cnc2c(NCc3cccc(I)c3)ncnc12 Show InChI InChI=1S/C19H20IN7O3/c1-21-18(29)19-7-23-12(14(19)28)17(30-19)27-9-26-13-15(24-8-25-16(13)27)22-6-10-3-2-4-11(20)5-10/h2-5,8-9,12,14,17,23,28H,6-7H2,1H3,(H,21,29)(H,22,24,25)/t12-,14+,17-,19+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Agonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50215112

((1R,3R,4R,7S)-3-(6-amino-purin-9-yl)-1-hydroxymeth...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@@]2(CO)CN[C@@H]1[C@@H]2O Show InChI InChI=1S/C11H14N6O3/c12-8-6-9(15-3-14-8)17(4-16-6)10-5-7(19)11(2-18,20-10)1-13-5/h3-5,7,10,13,18-19H,1-2H2,(H2,12,14,15)/t5-,7+,10-,11-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Agonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50144945

((2R,3R,4R,5R)-2-Hydroxymethyl-4-methoxy-5-(6-methy...)Show SMILES CO[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1n1cnc2c(N)ncnc12 Show InChI InChI=1S/C11H15N5O4/c1-19-8-7(18)5(2-17)20-11(8)16-4-15-6-9(12)13-3-14-10(6)16/h3-5,7-8,11,17-18H,2H2,1H3,(H2,12,13,14)/t5-,7-,8-,11-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Agonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50215113

((1R,3R,4R,7S)-(7-hydroxy-3-(N6-(3-iodobenzyl)-aden...)Show SMILES CNC(=O)[C@]12CO[C@H]([C@@H]1O)[C@@H](O2)n1cnc2c(NCc3cccc(I)c3)ncnc12 Show InChI InChI=1S/C19H19IN6O4/c1-21-18(28)19-7-29-13(14(19)27)17(30-19)26-9-25-12-15(23-8-24-16(12)26)22-6-10-3-2-4-11(20)5-10/h2-5,8-9,13-14,17,27H,6-7H2,1H3,(H,21,28)(H,22,23,24)/t13-,14+,17-,19+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Agonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50215110

((1R,3R,4R,7S)-(7-hydroxy-3-(N6-(3-iodobenzyl)-aden...)Show SMILES CNC(=O)[C@]12CN[C@H]([C@@H]1O)[C@@H](O2)n1cnc2c(NCc3cccc(I)c3)ncnc12 Show InChI InChI=1S/C19H20IN7O3/c1-21-18(29)19-7-23-12(14(19)28)17(30-19)27-9-26-13-15(24-8-25-16(13)27)22-6-10-3-2-4-11(20)5-10/h2-5,8-9,12,14,17,23,28H,6-7H2,1H3,(H,21,29)(H,22,24,25)/t12-,14+,17-,19+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >3.00E+3 | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Agonist activity at human adenosine A1 receptor |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50215109

((1R,3R,4R,7S)-(7-hydroxy-3-(adenin-9-yl)-2-oxa-5-a...)Show SMILES CNC(=O)[C@]12CN[C@H]([C@@H]1O)[C@@H](O2)n1cnc2c(N)ncnc12 Show InChI InChI=1S/C12H15N7O3/c1-14-11(21)12-2-15-5(7(12)20)10(22-12)19-4-18-6-8(13)16-3-17-9(6)19/h3-5,7,10,15,20H,2H2,1H3,(H,14,21)(H2,13,16,17)/t5-,7+,10-,12+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Agonist activity at human adenosine A3 receptor after 15 to 30 mins |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50215111

((1S,3R,4R,7S)-7-hydroxy-1-hydroxymethyl-3-(N6-(3-i...)Show SMILES OC[C@]12CN[C@H]([C@@H]1O)[C@@H](O2)n1cnc2c(NCc3cccc(I)c3)ncnc12 Show InChI InChI=1S/C18H19IN6O3/c19-11-3-1-2-10(4-11)5-20-15-13-16(23-8-22-15)25(9-24-13)17-12-14(27)18(7-26,28-17)6-21-12/h1-4,8-9,12,14,17,21,26-27H,5-7H2,(H,20,22,23)/t12-,14+,17-,18-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >3.00E+3 | n/a | n/a | n/a | n/a |
Santaris Pharma A/S
Curated by ChEMBL
| Assay Description
Agonist activity at human adenosine A1 receptor |
Bioorg Med Chem 15: 5440-7 (2007)
Article DOI: 10.1016/j.bmc.2007.05.056
BindingDB Entry DOI: 10.7270/Q2086515 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data