 Found 7 hits with Last Name = 'yoshizaki' and Initial = 'm'
Found 7 hits with Last Name = 'yoshizaki' and Initial = 'm' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM21642

((2S)-1-[(2S)-2-methyl-3-sulfanylpropanoyl]pyrrolid...)Show InChI InChI=1S/C9H15NO3S/c1-6(5-14)8(11)10-4-2-3-7(10)9(12)13/h6-7,14H,2-5H2,1H3,(H,12,13)/t6-,7+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of ACE by fluorometric assay |
J Nat Prod 51: 357-359 (1988)
Article DOI: 10.1021/np50056a033
BindingDB Entry DOI: 10.7270/Q2XW4M1C |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Xanthine dehydrogenase/oxidase
(Bos taurus (Bovine)) | BDBM35440
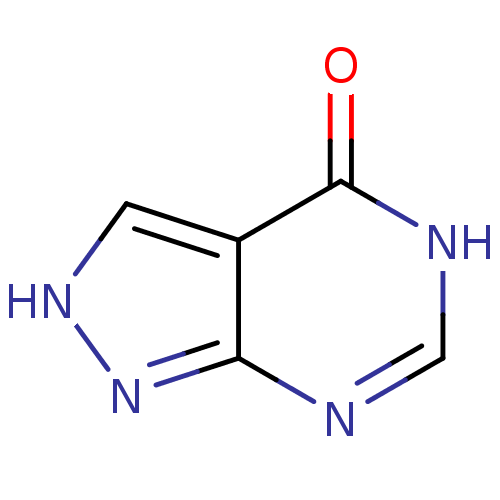
(ALLOPURINOL | MLS000069453 | SMR000059083 | cid_20...)Show InChI InChI=1S/C5H4N4O/c10-5-3-1-8-9-4(3)6-2-7-5/h1-2H,(H2,6,7,8,9,10) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of cow milk xanthine oxidase |
J Nat Prod 52: 210-211 (1989)
Article DOI: 10.1021/np50061a035
BindingDB Entry DOI: 10.7270/Q24749W5 |
More data for this
Ligand-Target Pair | |
Xanthine dehydrogenase/oxidase
(Bos taurus (Bovine)) | BDBM50241052

(1,2,3,4,6-Pgg | 1,2,3,4,6-pentakis-O-(3,4,5-trihyd...)Show SMILES Oc1cc(cc(O)c1O)C(=O)OC[C@H]1O[C@@H](OC(=O)c2cc(O)c(O)c(O)c2)[C@H](OC(=O)c2cc(O)c(O)c(O)c2)[C@@H](OC(=O)c2cc(O)c(O)c(O)c2)[C@@H]1OC(=O)c1cc(O)c(O)c(O)c1 |r| Show InChI InChI=1S/C41H32O26/c42-17-1-12(2-18(43)28(17)52)36(57)62-11-27-33(64-37(58)13-3-19(44)29(53)20(45)4-13)34(65-38(59)14-5-21(46)30(54)22(47)6-14)35(66-39(60)15-7-23(48)31(55)24(49)8-15)41(63-27)67-40(61)16-9-25(50)32(56)26(51)10-16/h1-10,27,33-35,41-56H,11H2/t27-,33-,34+,35-,41+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of cow milk xanthine oxidase |
J Nat Prod 52: 210-211 (1989)
Article DOI: 10.1021/np50061a035
BindingDB Entry DOI: 10.7270/Q24749W5 |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50242278
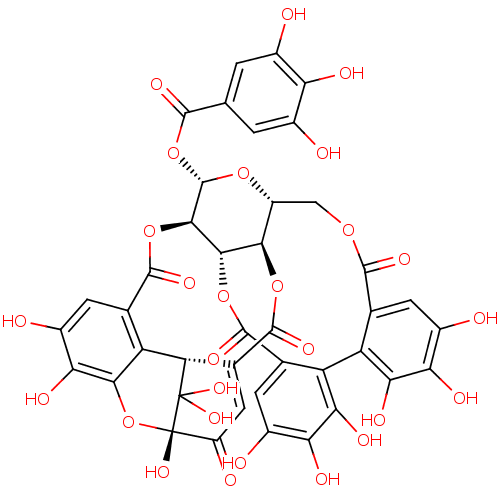
(CHEMBL506069 | Geraniin | geraniiin)Show SMILES Oc1cc(cc(O)c1O)C(=O)O[C@@H]1O[C@@H]2COC(=O)c3cc(O)c(O)c(O)c3-c3c(O)c(O)c(O)cc3C(=O)O[C@H]3[C@@H]2OC(=O)C2=CC(=O)[C@]4(O)Oc5c(O)c(O)cc(c5[C@H]2C4(O)O)C(=O)O[C@@H]13 |r,t:49,THB:43:45:61:52.59.51,48:47:61:52.59.51| Show InChI InChI=1S/C41H28O27/c42-13-1-8(2-14(43)24(13)48)34(54)67-39-33-32-30(64-38(58)12-6-19(47)41(61)40(59,60)23(12)22-11(37(57)66-33)5-17(46)27(51)31(22)68-41)18(63-39)7-62-35(55)9-3-15(44)25(49)28(52)20(9)21-10(36(56)65-32)4-16(45)26(50)29(21)53/h1-6,18,23,30,32-33,39,42-46,48-53,59-61H,7H2/t18-,23+,30-,32+,33-,39+,41+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of ACE by fluorometric assay |
J Nat Prod 51: 357-359 (1988)
Article DOI: 10.1021/np50056a033
BindingDB Entry DOI: 10.7270/Q2XW4M1C |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50242279

(6,7,8,11,12,13,22,23-octahydroxy-3,16-dioxo-21-(3,...)Show SMILES O[C@@H]1[C@H]2COC(=O)c3cc(O)c(O)c(O)c3-c3c(O)c(O)c(O)cc3C(=O)O[C@@H]1[C@@H](O)[C@H](OC(=O)c1cc(O)c(O)c(O)c1)O2 |r| Show InChI InChI=1S/C27H22O18/c28-9-1-6(2-10(29)16(9)32)24(39)45-27-22(38)23-19(35)13(43-27)5-42-25(40)7-3-11(30)17(33)20(36)14(7)15-8(26(41)44-23)4-12(31)18(34)21(15)37/h1-4,13,19,22-23,27-38H,5H2/t13-,19-,22-,23+,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.70E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of ACE by fluorometric assay |
J Nat Prod 51: 357-359 (1988)
Article DOI: 10.1021/np50056a033
BindingDB Entry DOI: 10.7270/Q2XW4M1C |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM4078

(6,7,13,14-tetrahydroxy-2,9-dioxatetracyclo[6.6.2.0...)Show InChI InChI=1S/C14H6O8/c15-5-1-3-7-8-4(14(20)22-11(7)9(5)17)2-6(16)10(18)12(8)21-13(3)19/h1-2,15-18H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of ACE by fluorometric assay |
J Nat Prod 51: 357-359 (1988)
Article DOI: 10.1021/np50056a033
BindingDB Entry DOI: 10.7270/Q2XW4M1C |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50085536
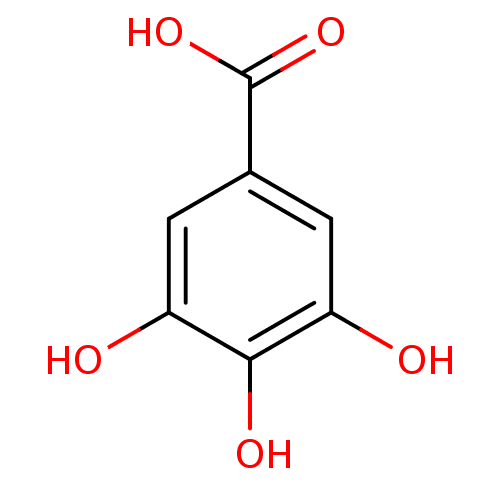
(3,4,5-Trihydroxybenzoate, X | 3,4,5-trihydroxybenz...)Show InChI InChI=1S/C7H6O5/c8-4-1-3(7(11)12)2-5(9)6(4)10/h1-2,8-10H,(H,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7.70E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of ACE by fluorometric assay |
J Nat Prod 51: 357-359 (1988)
Article DOI: 10.1021/np50056a033
BindingDB Entry DOI: 10.7270/Q2XW4M1C |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data