 Found 49 hits with Last Name = 'zyk' and Initial = 'nv'
Found 49 hits with Last Name = 'zyk' and Initial = 'nv' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587810
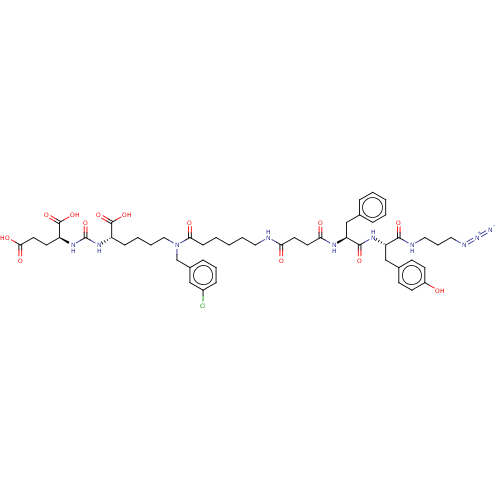
(CHEMBL5193353)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1cccc(Cl)c1)C(=O)CCCCCNC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587817

(CHEMBL5186540)Show SMILES CN1\C(=C\C=C\C=C\C2=[N+](CCCCCC(=O)NCc3cn(CCCNC(=O)[C@H](Cc4ccc(O)cc4)NC(=O)[C@H](Cc4ccccc4)NC(=O)CCC(=O)NCCCCCC(=O)N(CCCC[C@H](NC(=O)N[C@@H](CCC(O)=O)C(O)=O)C(O)=O)Cc4cccc(Cl)c4)nn3)c3ccc(cc3C2(C)C)S([O-])(=O)=O)C(C)(C)c2cc(ccc12)S(O)(=O)=O |r,c:8| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587818

(CHEMBL5206903)Show SMILES CN1\C(=C\C=C2/CCCC(\C=C\C3=[N+](CCCCCC(=O)NCc4cn(CCCNC(=O)[C@H](Cc5ccc(O)cc5)NC(=O)[C@H](Cc5ccccc5)NC(=O)CCC(=O)NCCCCCC(=O)N(CCCC[C@H](NC(=O)N[C@@H](CCC(O)=O)C(O)=O)C(O)=O)Cc5cccc(Cl)c5)nn4)c4ccc(cc4C3(C)C)S([O-])(=O)=O)=C2)C(C)(C)c2cc(ccc12)S(O)(=O)=O |r,c:12,117| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587810

(CHEMBL5193353)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1cccc(Cl)c1)C(=O)CCCCCNC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587810

(CHEMBL5193353)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1cccc(Cl)c1)C(=O)CCCCCNC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113936
BindingDB Entry DOI: 10.7270/Q2V69PKR |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587806
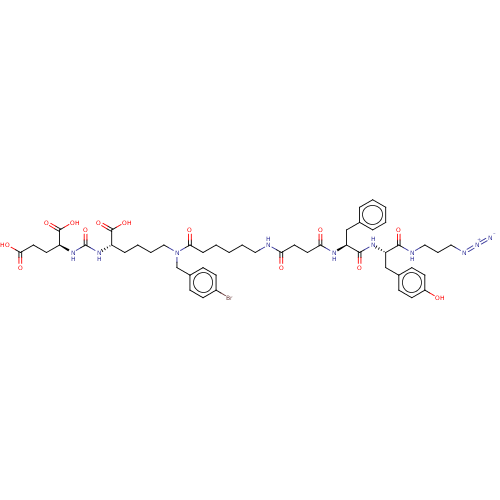
(CHEMBL5175556)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Br)cc1)C(=O)CCCCCNC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587801
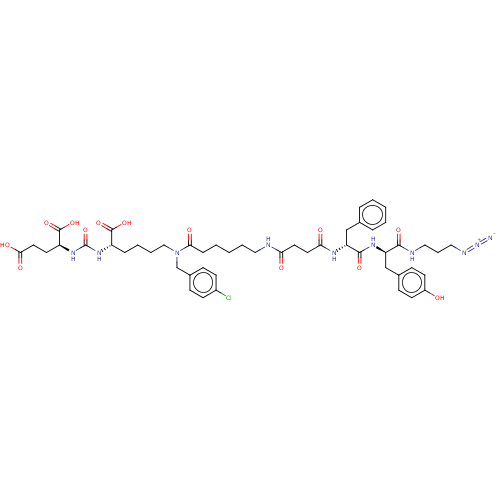
(CHEMBL5199943)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Cl)cc1)C(=O)CCCCCNC(=O)CCC(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccc(O)cc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587797
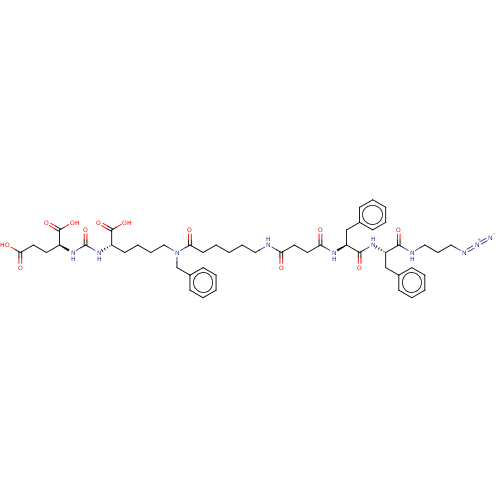
(CHEMBL5174173)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccccc1)C(=O)CCCCCNC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587797

(CHEMBL5174173)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccccc1)C(=O)CCCCCNC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113936
BindingDB Entry DOI: 10.7270/Q2V69PKR |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587808
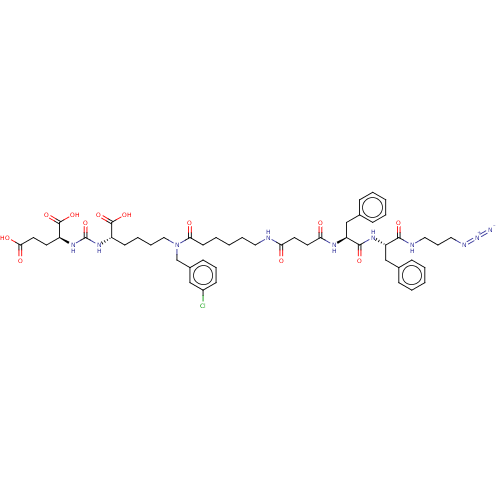
(CHEMBL5191246)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1cccc(Cl)c1)C(=O)CCCCCNC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 38 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587812
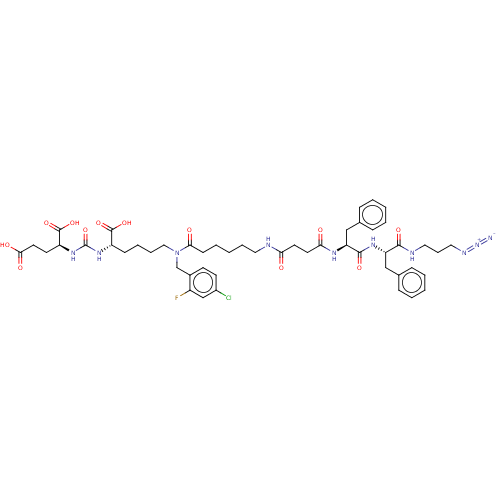
(CHEMBL5204105)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Cl)cc1F)C(=O)CCCCCNC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 43 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587800
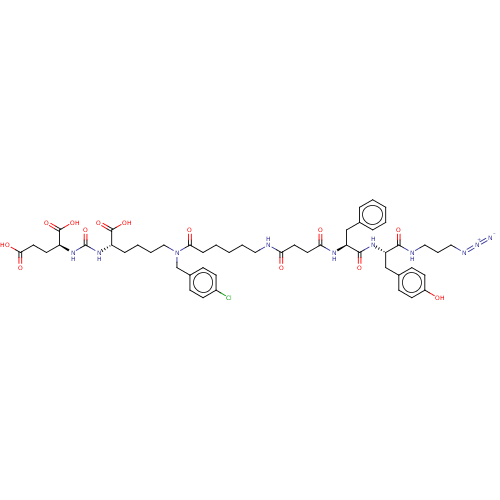
(CHEMBL5173940)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Cl)cc1)C(=O)CCCCCNC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 46 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587807
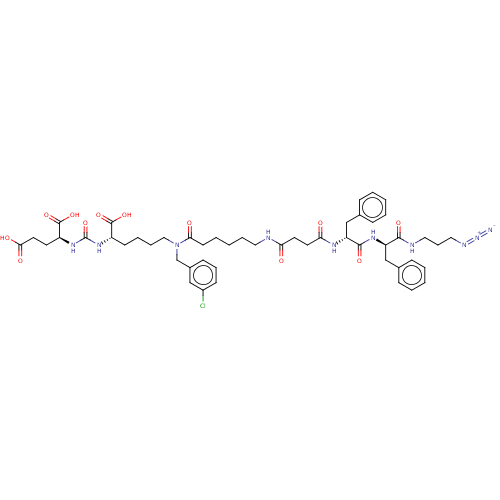
(CHEMBL5195613)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1cccc(Cl)c1)C(=O)CCCCCNC(=O)CCC(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccccc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 46 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587802
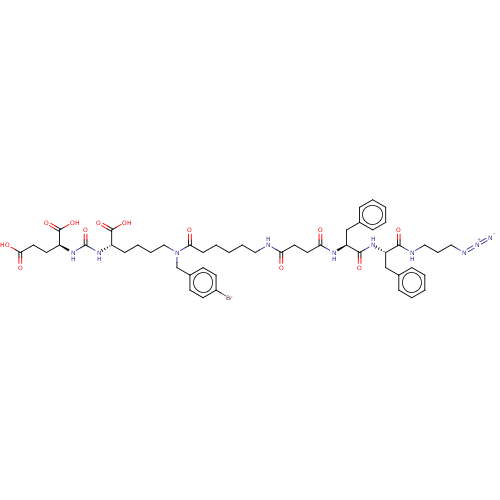
(CHEMBL5206132)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Br)cc1)C(=O)CCCCCNC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587798
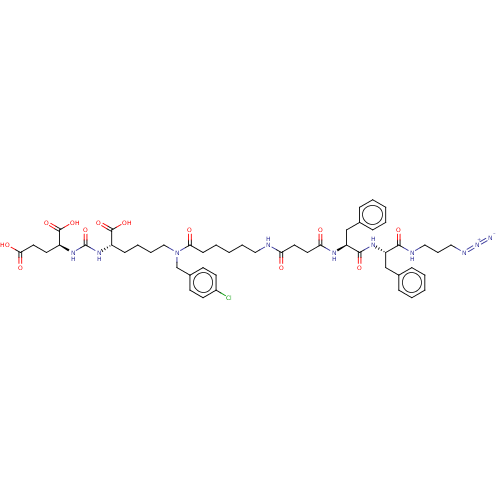
(CHEMBL5176891)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Cl)cc1)C(=O)CCCCCNC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 55 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50014323
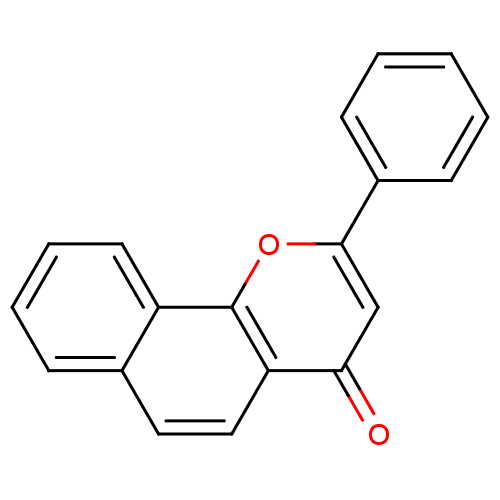
(2-PHENYL-4H-BENZO[H]CHROMEN-4-ONE | 2-Phenyl-benzo...)Show InChI InChI=1S/C19H12O2/c20-17-12-18(14-7-2-1-3-8-14)21-19-15-9-5-4-6-13(15)10-11-16(17)19/h1-12H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 57 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP1A2 in human liver microsome using phenacetin as substrate |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01157
BindingDB Entry DOI: 10.7270/Q2K64NZW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587816
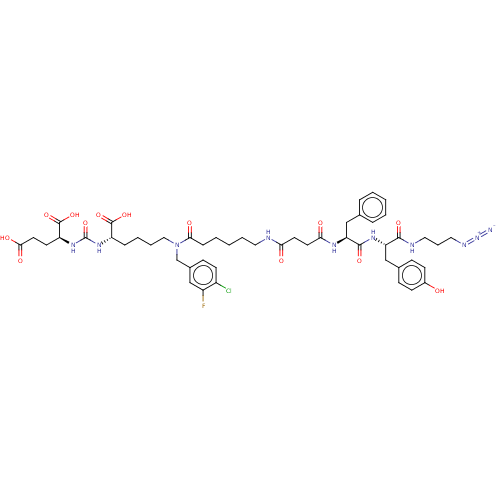
(CHEMBL5209061)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Cl)c(F)c1)C(=O)CCCCCNC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 62 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587813
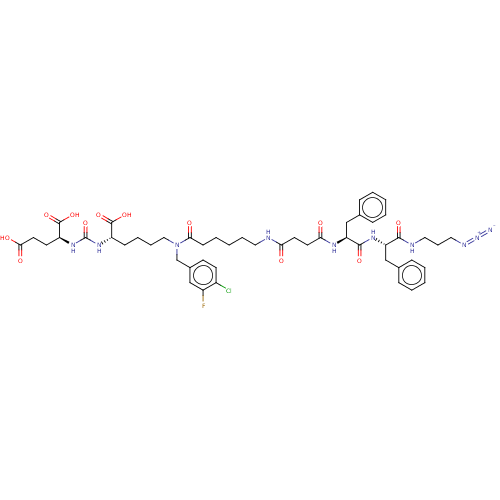
(CHEMBL5206082)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Cl)c(F)c1)C(=O)CCCCCNC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 63 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50121975
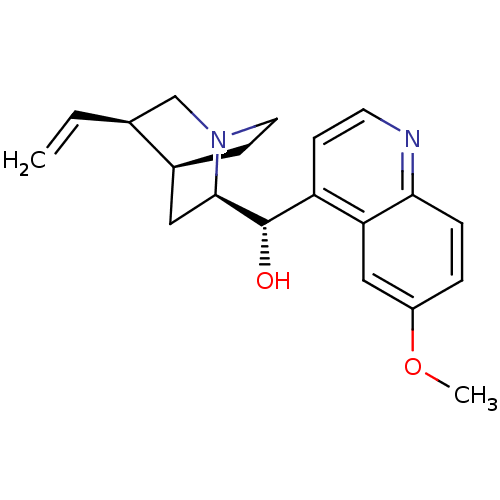
((6-Methoxy-quinolin-4-yl)-(5-vinyl-1-aza-bicyclo[2...)Show SMILES COc1ccc2nccc([C@H](O)[C@H]3C[C@@H]4CCN3C[C@@H]4C=C)c2c1 |r,THB:20:19:12.13:16.15,10:12:18.19:16.15| Show InChI InChI=1S/C20H24N2O2/c1-3-13-12-22-9-7-14(13)10-19(22)20(23)16-6-8-21-18-5-4-15(24-2)11-17(16)18/h3-6,8,11,13-14,19-20,23H,1,7,9-10,12H2,2H3/t13-,14-,19+,20-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 64 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2D6 in human liver microsome using dextromethorphan as substrate |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01157
BindingDB Entry DOI: 10.7270/Q2K64NZW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM17659
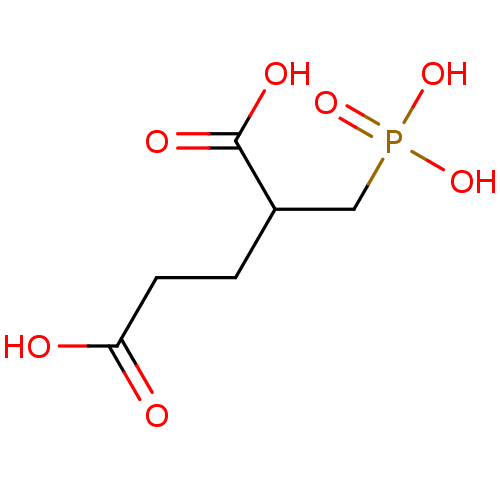
((R,S)-2-phosphonomethylpentanedioic acid | 2-(phos...)Show InChI InChI=1S/C6H11O7P/c7-5(8)2-1-4(6(9)10)3-14(11,12)13/h4H,1-3H2,(H,7,8)(H,9,10)(H2,11,12,13) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587811
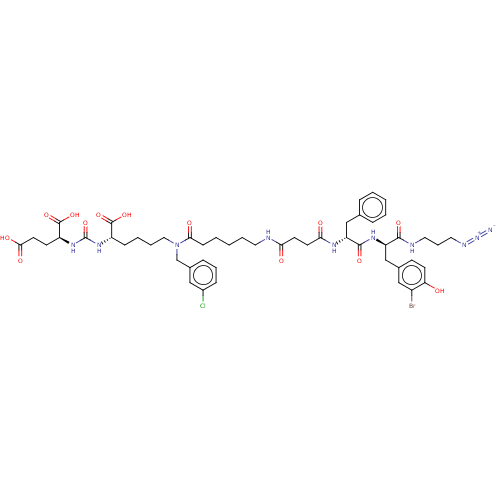
(CHEMBL5181465)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1cccc(Cl)c1)C(=O)CCCCCNC(=O)CCC(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccc(O)c(Br)c1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM17659

((R,S)-2-phosphonomethylpentanedioic acid | 2-(phos...)Show InChI InChI=1S/C6H11O7P/c7-5(8)2-1-4(6(9)10)3-14(11,12)13/h4H,1-3H2,(H,7,8)(H,9,10)(H2,11,12,13) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113936
BindingDB Entry DOI: 10.7270/Q2V69PKR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587805

(CHEMBL5182882)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Br)cc1)C(=O)CCCCCNC(=O)CCC(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccc(O)c(Br)c1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 89 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM8610

(1-[4-(4-{[(2R,4S)-2-(2,4-dichlorophenyl)-2-(1H-imi...)Show SMILES [H][C@]1(COc2ccc(cc2)N2CCN(CC2)C(C)=O)CO[C@@](Cn2ccnc2)(O1)c1ccc(Cl)cc1Cl |r| Show InChI InChI=1S/C26H28Cl2N4O4/c1-19(33)31-10-12-32(13-11-31)21-3-5-22(6-4-21)34-15-23-16-35-26(36-23,17-30-9-8-29-18-30)24-7-2-20(27)14-25(24)28/h2-9,14,18,23H,10-13,15-17H2,1H3/t23-,26-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP3A4 in human liver microsome using midazolam as substrate |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01157
BindingDB Entry DOI: 10.7270/Q2K64NZW |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587809
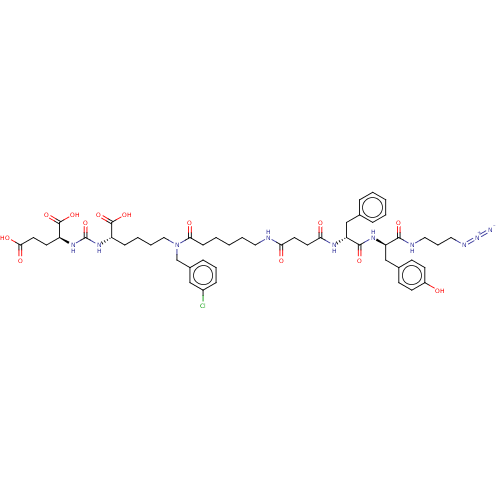
(CHEMBL5209008)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1cccc(Cl)c1)C(=O)CCCCCNC(=O)CCC(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccc(O)cc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 107 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587815
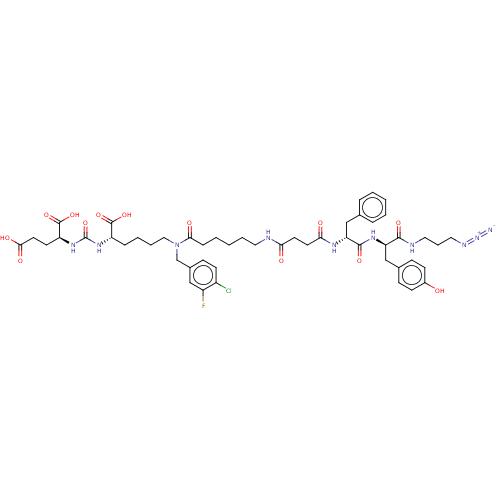
(CHEMBL5194398)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Cl)c(F)c1)C(=O)CCCCCNC(=O)CCC(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccc(O)cc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 108 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50522950

(CHEMBL4463310)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Cl)cc1)C(=O)CCCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| Show InChI InChI=1S/C25H35ClN6O8/c26-18-10-8-17(9-11-18)16-32(21(33)7-2-1-4-14-28-31-27)15-5-3-6-19(23(36)37)29-25(40)30-20(24(38)39)12-13-22(34)35/h8-11,19-20H,1-7,12-16H2,(H,34,35)(H,36,37)(H,38,39)(H2,29,30,40)/t19-,20-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
Lomonosov Moscow State University
Curated by ChEMBL
| Assay Description
Inhibition of PSMA in human LNCAP cells using NAAG as substrate measured after 2 hrs by amplex red reagent based fluorescence assay |
Bioorg Med Chem Lett 29: 2229-2235 (2019)
Article DOI: 10.1016/j.bmcl.2019.06.035
BindingDB Entry DOI: 10.7270/Q280561X |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50522948

(CHEMBL4444705)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(O)cc1)C(=O)CCCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| Show InChI InChI=1S/C25H36N6O9/c26-30-27-14-4-1-2-7-21(33)31(16-17-8-10-18(32)11-9-17)15-5-3-6-19(23(36)37)28-25(40)29-20(24(38)39)12-13-22(34)35/h8-11,19-20,32H,1-7,12-16H2,(H,34,35)(H,36,37)(H,38,39)(H2,28,29,40)/t19-,20-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
Lomonosov Moscow State University
Curated by ChEMBL
| Assay Description
Inhibition of PSMA in human LNCAP cells using NAAG as substrate measured after 2 hrs by amplex red reagent based fluorescence assay |
Bioorg Med Chem Lett 29: 2229-2235 (2019)
Article DOI: 10.1016/j.bmcl.2019.06.035
BindingDB Entry DOI: 10.7270/Q280561X |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50522949

(CHEMBL4449364)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Br)cc1)C(=O)CCCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| Show InChI InChI=1S/C25H35BrN6O8/c26-18-10-8-17(9-11-18)16-32(21(33)7-2-1-4-14-28-31-27)15-5-3-6-19(23(36)37)29-25(40)30-20(24(38)39)12-13-22(34)35/h8-11,19-20H,1-7,12-16H2,(H,34,35)(H,36,37)(H,38,39)(H2,29,30,40)/t19-,20-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
Lomonosov Moscow State University
Curated by ChEMBL
| Assay Description
Inhibition of PSMA in human LNCAP cells using NAAG as substrate measured after 2 hrs by amplex red reagent based fluorescence assay |
Bioorg Med Chem Lett 29: 2229-2235 (2019)
Article DOI: 10.1016/j.bmcl.2019.06.035
BindingDB Entry DOI: 10.7270/Q280561X |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587796

(CHEMBL5200623)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccccc1)C(=O)CCCNC(=O)CCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 175 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM8610

(1-[4-(4-{[(2R,4S)-2-(2,4-dichlorophenyl)-2-(1H-imi...)Show SMILES [H][C@]1(COc2ccc(cc2)N2CCN(CC2)C(C)=O)CO[C@@](Cn2ccnc2)(O1)c1ccc(Cl)cc1Cl |r| Show InChI InChI=1S/C26H28Cl2N4O4/c1-19(33)31-10-12-32(13-11-31)21-3-5-22(6-4-21)34-15-23-16-35-26(36-23,17-30-9-8-29-18-30)24-7-2-20(27)14-25(24)28/h2-9,14,18,23H,10-13,15-17H2,1H3/t23-,26-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 184 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP3A4 in human liver microsome using testosterone as substrate |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01157
BindingDB Entry DOI: 10.7270/Q2K64NZW |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587799
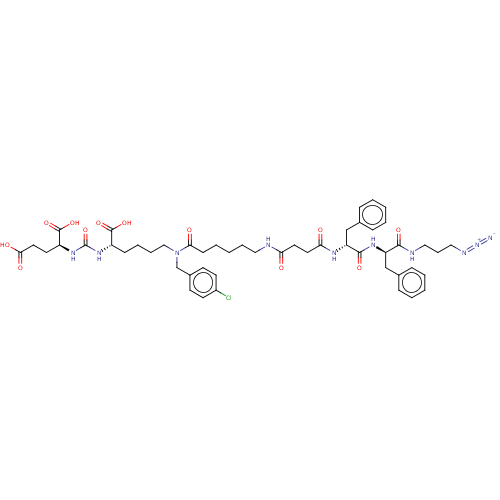
(CHEMBL5184603)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Cl)cc1)C(=O)CCCCCNC(=O)CCC(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccccc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 226 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587814
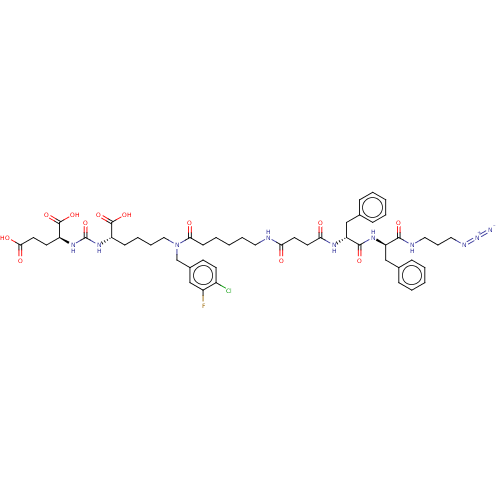
(CHEMBL5171260)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Cl)c(F)c1)C(=O)CCCCCNC(=O)CCC(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccccc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 294 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587814

(CHEMBL5171260)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Cl)c(F)c1)C(=O)CCCCCNC(=O)CCC(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccccc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 294 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113936
BindingDB Entry DOI: 10.7270/Q2V69PKR |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587803
(CHEMBL5202302)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Br)cc1)C(=O)CCCCCNC(=O)CCC(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccccc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 319 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50265784

((S)-2-(3-((S)-5-Amino-1-carboxypentyl)ureido)penta...)Show SMILES NCCCC[C@H](NC(=O)N[C@@H](CCC(O)=O)C(O)=O)C(O)=O |r| Show InChI InChI=1S/C12H21N3O7/c13-6-2-1-3-7(10(18)19)14-12(22)15-8(11(20)21)4-5-9(16)17/h7-8H,1-6,13H2,(H,16,17)(H,18,19)(H,20,21)(H2,14,15,22)/t7-,8-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 547 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50265784

((S)-2-(3-((S)-5-Amino-1-carboxypentyl)ureido)penta...)Show SMILES NCCCC[C@H](NC(=O)N[C@@H](CCC(O)=O)C(O)=O)C(O)=O |r| Show InChI InChI=1S/C12H21N3O7/c13-6-2-1-3-7(10(18)19)14-12(22)15-8(11(20)21)4-5-9(16)17/h7-8H,1-6,13H2,(H,16,17)(H,18,19)(H,20,21)(H2,14,15,22)/t7-,8-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 547 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113936
BindingDB Entry DOI: 10.7270/Q2V69PKR |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50090677
(4-Amino-N-(2-phenyl-2H-pyrazol-3-yl)-benzenesulfon...)Show InChI InChI=1S/C15H14N4O2S/c16-12-6-8-14(9-7-12)22(20,21)18-15-10-11-17-19(15)13-4-2-1-3-5-13/h1-11,18H,16H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 641 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2C9 in human liver microsome using tolbutamide as substrate |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01157
BindingDB Entry DOI: 10.7270/Q2K64NZW |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50052024

(CHEMBL787 | montelukast)Show SMILES CC(C)(O)c1ccccc1CC[C@@H](SCC1(CC(O)=O)CC1)c1cccc(\C=C\c2ccc3ccc(Cl)cc3n2)c1 |r| Show InChI InChI=1S/C35H36ClNO3S/c1-34(2,40)30-9-4-3-7-25(30)13-17-32(41-23-35(18-19-35)22-33(38)39)27-8-5-6-24(20-27)10-15-29-16-12-26-11-14-28(36)21-31(26)37-29/h3-12,14-16,20-21,32,40H,13,17-19,22-23H2,1-2H3,(H,38,39)/b15-10+/t32-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 1.27E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2C8 in human liver microsome using paclitaxel as substrate |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01157
BindingDB Entry DOI: 10.7270/Q2K64NZW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50265784

((S)-2-(3-((S)-5-Amino-1-carboxypentyl)ureido)penta...)Show SMILES NCCCC[C@H](NC(=O)N[C@@H](CCC(O)=O)C(O)=O)C(O)=O |r| Show InChI InChI=1S/C12H21N3O7/c13-6-2-1-3-7(10(18)19)14-12(22)15-8(11(20)21)4-5-9(16)17/h7-8H,1-6,13H2,(H,16,17)(H,18,19)(H,20,21)(H2,14,15,22)/t7-,8-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.47E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lomonosov Moscow State University
Curated by ChEMBL
| Assay Description
Inhibition of PSMA in human LNCAP cells using NAAG as substrate measured after 2 hrs by amplex red reagent based fluorescence assay |
Bioorg Med Chem Lett 29: 2229-2235 (2019)
Article DOI: 10.1016/j.bmcl.2019.06.035
BindingDB Entry DOI: 10.7270/Q280561X |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50587804
(CHEMBL5191610)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCN(Cc1ccc(Br)cc1)C(=O)CCCCCNC(=O)CCC(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccc(O)cc1)C(=O)NCCCN=[N+]=[N-])C(O)=O)C(O)=O |r| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.16E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01935
BindingDB Entry DOI: 10.7270/Q2NS0ZWP |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50581559

(CHEMBL5083665)Show SMILES [H][C@]1(CCCN1C(=O)C[C@@H](OC)[C@H]([C@@H](C)CC)N(C)C(=O)[C@@H](NC(=O)[C@H](C(C)C)N(C)C(=O)OCc1ccc(NC(=O)[C@H](CCCNC(N)=O)NC(=O)[C@@H](NC(=O)CCCc2cn(CCCNC(=O)[C@H](Cc3ccc(O)cc3)NC(=O)[C@H](Cc3ccccc3)NC(=O)CCC(=O)NCCCCCC(=O)N(CCCC[C@H](NC(=O)N[C@@H](CCC(O)=O)C(O)=O)C(O)=O)Cc3cccc(Cl)c3)nn2)C(C)C)cc1)C(C)C)[C@H](OC)[C@@H](C)C(=O)N[C@H](C)[C@@H](O)c1ccccc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.12E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP3A4 in human liver microsome using midazolam as substrate |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01157
BindingDB Entry DOI: 10.7270/Q2K64NZW |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50581559

(CHEMBL5083665)Show SMILES [H][C@]1(CCCN1C(=O)C[C@@H](OC)[C@H]([C@@H](C)CC)N(C)C(=O)[C@@H](NC(=O)[C@H](C(C)C)N(C)C(=O)OCc1ccc(NC(=O)[C@H](CCCNC(N)=O)NC(=O)[C@@H](NC(=O)CCCc2cn(CCCNC(=O)[C@H](Cc3ccc(O)cc3)NC(=O)[C@H](Cc3ccccc3)NC(=O)CCC(=O)NCCCCCC(=O)N(CCCC[C@H](NC(=O)N[C@@H](CCC(O)=O)C(O)=O)C(O)=O)Cc3cccc(Cl)c3)nn2)C(C)C)cc1)C(C)C)[C@H](OC)[C@@H](C)C(=O)N[C@H](C)[C@@H](O)c1ccccc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.12E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP3A4 in human liver microsome using testosterone as substrate |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01157
BindingDB Entry DOI: 10.7270/Q2K64NZW |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50240772
((1R,2S)-(-)-2-phenylcyclopropylamine | (1R,2S)-2-p...)Show InChI InChI=1S/C9H11N/c10-9-6-8(9)7-4-2-1-3-5-7/h1-5,8-9H,6,10H2/t8-,9+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.17E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2C19 in human liver microsome using mephenytoin as substrate |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01157
BindingDB Entry DOI: 10.7270/Q2K64NZW |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50581559

(CHEMBL5083665)Show SMILES [H][C@]1(CCCN1C(=O)C[C@@H](OC)[C@H]([C@@H](C)CC)N(C)C(=O)[C@@H](NC(=O)[C@H](C(C)C)N(C)C(=O)OCc1ccc(NC(=O)[C@H](CCCNC(N)=O)NC(=O)[C@@H](NC(=O)CCCc2cn(CCCNC(=O)[C@H](Cc3ccc(O)cc3)NC(=O)[C@H](Cc3ccccc3)NC(=O)CCC(=O)NCCCCCC(=O)N(CCCC[C@H](NC(=O)N[C@@H](CCC(O)=O)C(O)=O)C(O)=O)Cc3cccc(Cl)c3)nn2)C(C)C)cc1)C(C)C)[C@H](OC)[C@@H](C)C(=O)N[C@H](C)[C@@H](O)c1ccccc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP1A2 in human liver microsome using phenacetin as substrate |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01157
BindingDB Entry DOI: 10.7270/Q2K64NZW |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50581559

(CHEMBL5083665)Show SMILES [H][C@]1(CCCN1C(=O)C[C@@H](OC)[C@H]([C@@H](C)CC)N(C)C(=O)[C@@H](NC(=O)[C@H](C(C)C)N(C)C(=O)OCc1ccc(NC(=O)[C@H](CCCNC(N)=O)NC(=O)[C@@H](NC(=O)CCCc2cn(CCCNC(=O)[C@H](Cc3ccc(O)cc3)NC(=O)[C@H](Cc3ccccc3)NC(=O)CCC(=O)NCCCCCC(=O)N(CCCC[C@H](NC(=O)N[C@@H](CCC(O)=O)C(O)=O)C(O)=O)Cc3cccc(Cl)c3)nn2)C(C)C)cc1)C(C)C)[C@H](OC)[C@@H](C)C(=O)N[C@H](C)[C@@H](O)c1ccccc1 |r| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2D6 in human liver microsome using dextromethorphan as substrate |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01157
BindingDB Entry DOI: 10.7270/Q2K64NZW |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50581559

(CHEMBL5083665)Show SMILES [H][C@]1(CCCN1C(=O)C[C@@H](OC)[C@H]([C@@H](C)CC)N(C)C(=O)[C@@H](NC(=O)[C@H](C(C)C)N(C)C(=O)OCc1ccc(NC(=O)[C@H](CCCNC(N)=O)NC(=O)[C@@H](NC(=O)CCCc2cn(CCCNC(=O)[C@H](Cc3ccc(O)cc3)NC(=O)[C@H](Cc3ccccc3)NC(=O)CCC(=O)NCCCCCC(=O)N(CCCC[C@H](NC(=O)N[C@@H](CCC(O)=O)C(O)=O)C(O)=O)Cc3cccc(Cl)c3)nn2)C(C)C)cc1)C(C)C)[C@H](OC)[C@@H](C)C(=O)N[C@H](C)[C@@H](O)c1ccccc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2C19 in human liver microsome using mephenytoin as substrate |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01157
BindingDB Entry DOI: 10.7270/Q2K64NZW |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50581559

(CHEMBL5083665)Show SMILES [H][C@]1(CCCN1C(=O)C[C@@H](OC)[C@H]([C@@H](C)CC)N(C)C(=O)[C@@H](NC(=O)[C@H](C(C)C)N(C)C(=O)OCc1ccc(NC(=O)[C@H](CCCNC(N)=O)NC(=O)[C@@H](NC(=O)CCCc2cn(CCCNC(=O)[C@H](Cc3ccc(O)cc3)NC(=O)[C@H](Cc3ccccc3)NC(=O)CCC(=O)NCCCCCC(=O)N(CCCC[C@H](NC(=O)N[C@@H](CCC(O)=O)C(O)=O)C(O)=O)Cc3cccc(Cl)c3)nn2)C(C)C)cc1)C(C)C)[C@H](OC)[C@@H](C)C(=O)N[C@H](C)[C@@H](O)c1ccccc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2C9 in human liver microsome using tolbutamide as substrate |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01157
BindingDB Entry DOI: 10.7270/Q2K64NZW |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50581559

(CHEMBL5083665)Show SMILES [H][C@]1(CCCN1C(=O)C[C@@H](OC)[C@H]([C@@H](C)CC)N(C)C(=O)[C@@H](NC(=O)[C@H](C(C)C)N(C)C(=O)OCc1ccc(NC(=O)[C@H](CCCNC(N)=O)NC(=O)[C@@H](NC(=O)CCCc2cn(CCCNC(=O)[C@H](Cc3ccc(O)cc3)NC(=O)[C@H](Cc3ccccc3)NC(=O)CCC(=O)NCCCCCC(=O)N(CCCC[C@H](NC(=O)N[C@@H](CCC(O)=O)C(O)=O)C(O)=O)Cc3cccc(Cl)c3)nn2)C(C)C)cc1)C(C)C)[C@H](OC)[C@@H](C)C(=O)N[C@H](C)[C@@H](O)c1ccccc1 |r| | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CYP2C8 in human liver microsome using paclitaxel as substrate |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01157
BindingDB Entry DOI: 10.7270/Q2K64NZW |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data