

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50320022 ((S)-1-((S)-2-amino-3-(4-hydroxyphenyl)propanoyl)-N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 8.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Roma Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat mu opioid receptor expressed in mouse HN9.10 cells | J Med Chem 53: 4550-4 (2010) Article DOI: 10.1021/jm1001343 BindingDB Entry DOI: 10.7270/Q23F4PTK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50139013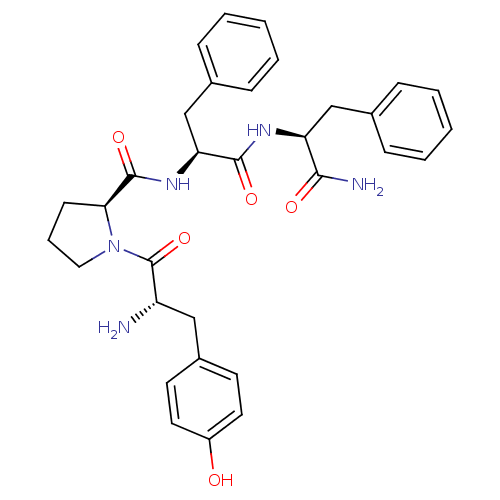 ((S)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 9.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Roma Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat mu opioid receptor expressed in mouse HN9.10 cells | J Med Chem 53: 4550-4 (2010) Article DOI: 10.1021/jm1001343 BindingDB Entry DOI: 10.7270/Q23F4PTK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50320023 ((S)-1-((S)-2-amino-3-(4-hydroxyphenyl)propanoyl)-N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Roma Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat mu opioid receptor expressed in mouse HN9.10 cells | J Med Chem 53: 4550-4 (2010) Article DOI: 10.1021/jm1001343 BindingDB Entry DOI: 10.7270/Q23F4PTK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50320024 ((S)-N-(3-((S)-1-amino-1-oxo-3-phenylpropan-2-ylami...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Roma Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat mu opioid receptor expressed in mouse HN9.10 cells | J Med Chem 53: 4550-4 (2010) Article DOI: 10.1021/jm1001343 BindingDB Entry DOI: 10.7270/Q23F4PTK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50320023 ((S)-1-((S)-2-amino-3-(4-hydroxyphenyl)propanoyl)-N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Roma Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from human delta opioid receptor expressed in HEK293 cells | J Med Chem 53: 4550-4 (2010) Article DOI: 10.1021/jm1001343 BindingDB Entry DOI: 10.7270/Q23F4PTK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50320022 ((S)-1-((S)-2-amino-3-(4-hydroxyphenyl)propanoyl)-N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Roma Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from human delta opioid receptor expressed in HEK293 cells | J Med Chem 53: 4550-4 (2010) Article DOI: 10.1021/jm1001343 BindingDB Entry DOI: 10.7270/Q23F4PTK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 6 (Homo sapiens (Human)) | BDBM50005711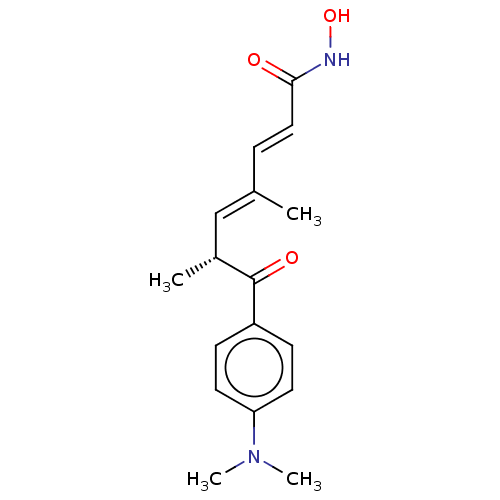 (CHEBI:46024 | GNF-Pf-1011 | TRICHOSTATIN | Trichos...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE PC cid PC sid PDB UniChem | PDB Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC6 using RHK-K(Ac) as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Histone deacetylase 6 (Homo sapiens (Human)) | BDBM50506775 (CHEMBL4593437) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC6 using RHK-K(Ac) as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM50005711 (CHEBI:46024 | GNF-Pf-1011 | TRICHOSTATIN | Trichos...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE PC cid PC sid PDB UniChem | Article PubMed | n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC1 using RHK-K(Ac) as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 6 (Homo sapiens (Human)) | BDBM50506777 (CHEMBL4476057) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC6 using RHK-K(Ac) as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50139013 ((S)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Roma Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | J Med Chem 53: 4550-4 (2010) Article DOI: 10.1021/jm1001343 BindingDB Entry DOI: 10.7270/Q23F4PTK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 6 (Homo sapiens (Human)) | BDBM50506780 (CHEMBL4550522) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC6 using RHK-K(Ac) as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 6 (Homo sapiens (Human)) | BDBM50506778 (CHEMBL4469596) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC6 using RHK-K(Ac) as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50320022 ((S)-1-((S)-2-amino-3-(4-hydroxyphenyl)propanoyl)-N...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Roma Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | J Med Chem 53: 4550-4 (2010) Article DOI: 10.1021/jm1001343 BindingDB Entry DOI: 10.7270/Q23F4PTK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 6 (Homo sapiens (Human)) | BDBM50506779 (CHEMBL4521590) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC6 using RHK-K(Ac) as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 6 (Homo sapiens (Human)) | BDBM50506781 (CHEMBL4435005) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC6 using RHK-K(Ac) as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM50506775 (CHEMBL4593437) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC1 using RHK-K(Ac) as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM50506780 (CHEMBL4550522) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC1 using RHK-K(Ac) as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50320024 ((S)-N-(3-((S)-1-amino-1-oxo-3-phenylpropan-2-ylami...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Roma Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | J Med Chem 53: 4550-4 (2010) Article DOI: 10.1021/jm1001343 BindingDB Entry DOI: 10.7270/Q23F4PTK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM50506781 (CHEMBL4435005) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC1 using RHK-K(Ac) as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM50506779 (CHEMBL4521590) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC1 using RHK-K(Ac) as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 4 (Homo sapiens (Human)) | BDBM50446481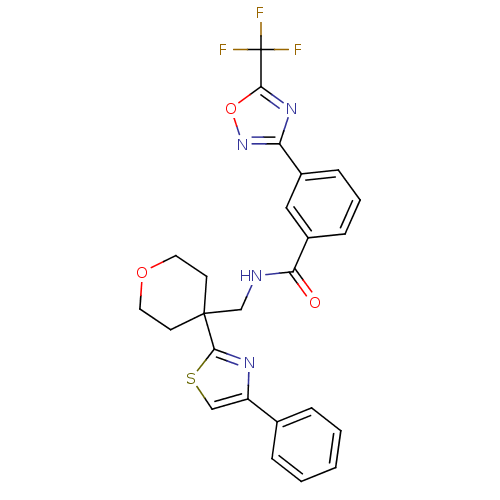 (CHEMBL3110004 | US10011611, TMP269 | US10722597, C...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem | Article PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC4 using Boc-Lys(trifluoroacetyl)-AMC as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50320023 ((S)-1-((S)-2-amino-3-(4-hydroxyphenyl)propanoyl)-N...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 330 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Roma Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | J Med Chem 53: 4550-4 (2010) Article DOI: 10.1021/jm1001343 BindingDB Entry DOI: 10.7270/Q23F4PTK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50320022 ((S)-1-((S)-2-amino-3-(4-hydroxyphenyl)propanoyl)-N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 390 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Roma Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically evoked contraction | J Med Chem 53: 4550-4 (2010) Article DOI: 10.1021/jm1001343 BindingDB Entry DOI: 10.7270/Q23F4PTK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50139013 ((S)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 510 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Roma Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically evoked contraction | J Med Chem 53: 4550-4 (2010) Article DOI: 10.1021/jm1001343 BindingDB Entry DOI: 10.7270/Q23F4PTK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM50506778 (CHEMBL4469596) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC1 using RHK-K(Ac) as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50320023 ((S)-1-((S)-2-amino-3-(4-hydroxyphenyl)propanoyl)-N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Roma Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically evoked contraction | J Med Chem 53: 4550-4 (2010) Article DOI: 10.1021/jm1001343 BindingDB Entry DOI: 10.7270/Q23F4PTK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM50506777 (CHEMBL4476057) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC1 using RHK-K(Ac) as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50320024 ((S)-N-(3-((S)-1-amino-1-oxo-3-phenylpropan-2-ylami...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Roma Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically evoked contraction | J Med Chem 53: 4550-4 (2010) Article DOI: 10.1021/jm1001343 BindingDB Entry DOI: 10.7270/Q23F4PTK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 4 (Homo sapiens (Human)) | BDBM50506780 (CHEMBL4550522) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC4 using Boc-Lys(trifluoroacetyl)-AMC as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 4 (Homo sapiens (Human)) | BDBM50005711 (CHEBI:46024 | GNF-Pf-1011 | TRICHOSTATIN | Trichos...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE PC cid PC sid PDB UniChem | Article PubMed | n/a | n/a | 3.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC4 using Boc-Lys(trifluoroacetyl)-AMC as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 4 (Homo sapiens (Human)) | BDBM50506781 (CHEMBL4435005) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC4 using Boc-Lys(trifluoroacetyl)-AMC as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Smoothened homolog (Mus musculus) | BDBM50233217 (CHEMBL4090942) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.38E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Sapienza University Curated by ChEMBL | Assay Description Inhibition of Bodipy-cyclopamine binding to mouse FLAG-tagged Smo expressed in HEK293 cells after 3 hrs by Hoechst staining based fluorescence assay | J Med Chem 60: 1469-1477 (2017) Article DOI: 10.1021/acs.jmedchem.6b01135 BindingDB Entry DOI: 10.7270/Q25X2C51 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Smoothened homolog (Mus musculus) | BDBM50233218 (CHEMBL4063037) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Sapienza University Curated by ChEMBL | Assay Description Inhibition of Bodipy-cyclopamine binding to mouse FLAG-tagged Smo expressed in HEK293 cells after 3 hrs by Hoechst staining based fluorescence assay | J Med Chem 60: 1469-1477 (2017) Article DOI: 10.1021/acs.jmedchem.6b01135 BindingDB Entry DOI: 10.7270/Q25X2C51 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 4 (Homo sapiens (Human)) | BDBM50506778 (CHEMBL4469596) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC4 using Boc-Lys(trifluoroacetyl)-AMC as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 4 (Homo sapiens (Human)) | BDBM50506779 (CHEMBL4521590) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC4 using Boc-Lys(trifluoroacetyl)-AMC as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 4 (Homo sapiens (Human)) | BDBM50506777 (CHEMBL4476057) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC4 using Boc-Lys(trifluoroacetyl)-AMC as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 4 (Homo sapiens (Human)) | BDBM50506775 (CHEMBL4593437) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC4 using Boc-Lys(trifluoroacetyl)-AMC as substrate by homogeneous fluorescence release assay | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA (cytosine-5)-methyltransferase 3A (Homo sapiens (Human)) | BDBM50506774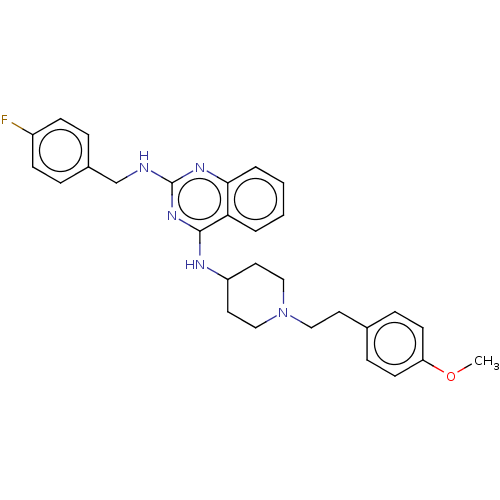 (CHEMBL4470149) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 4.80E+3 | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human DNMT3a C-terminal catalytic domain (623 to 908 residues) using 5'-biotinylated/3'-FAM-oligonucleotide as substrate me... | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone-lysine N-methyltransferase EHMT2 (Homo sapiens (Human)) | BDBM50506774 (CHEMBL4470149) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 5.09E+4 | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal GST-fused G9a (786 to 1210 residues) expressed in Escherichia coli using biotinylated H3 (1 to 21 residues... | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA (cytosine-5)-methyltransferase 1 (Homo sapiens (Human)) | BDBM50506776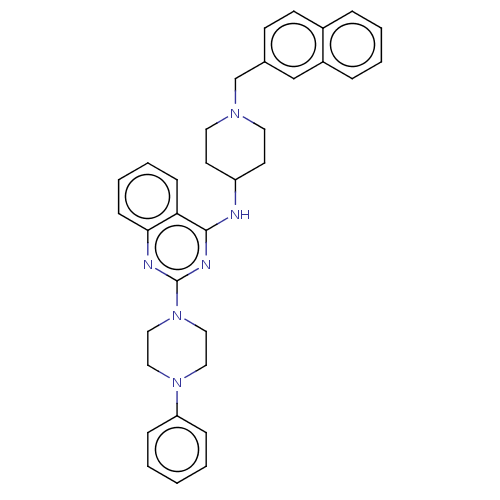 (CHEMBL4442279) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human DNMT1 using biotinylated DNA duplex as substrate measured after 2 hrs in presence of Adomet/[methyl-3H]Adomet by topc... | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA (cytosine-5)-methyltransferase 1 (Homo sapiens (Human)) | BDBM50506774 (CHEMBL4470149) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human DNMT1 using biotinylated DNA duplex as substrate measured after 2 hrs in presence of Adomet/[methyl-3H]Adomet by topc... | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA (cytosine-5)-methyltransferase 1 (Homo sapiens (Human)) | BDBM50506782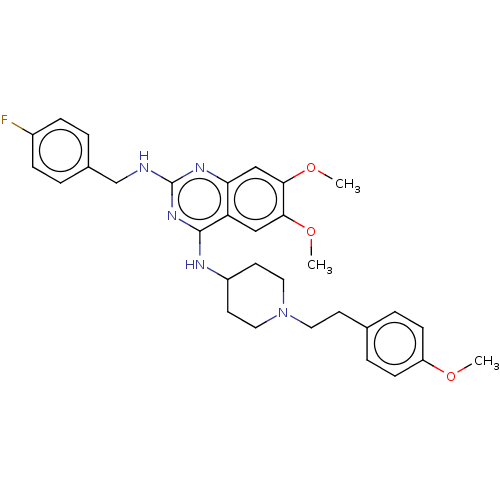 (CHEMBL4461581) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human DNMT1 using biotinylated DNA duplex as substrate measured after 2 hrs in presence of Adomet/[methyl-3H]Adomet by topc... | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA (cytosine-5)-methyltransferase 1 (Homo sapiens (Human)) | BDBM50506783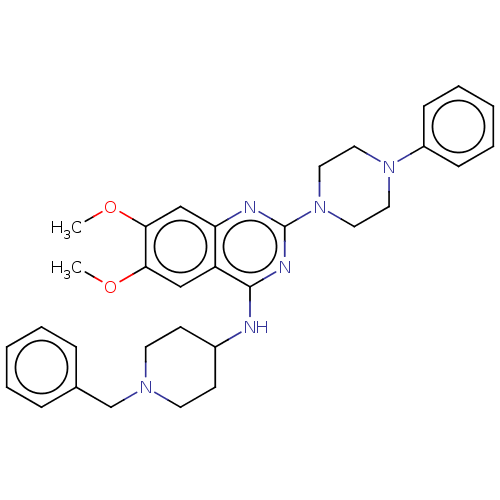 (CHEMBL4582042) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human DNMT1 using biotinylated DNA duplex as substrate measured after 2 hrs in presence of Adomet/[methyl-3H]Adomet by topc... | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA (cytosine-5)-methyltransferase 1 (Homo sapiens (Human)) | BDBM50506784 (CHEMBL4527662) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human DNMT1 using biotinylated DNA duplex as substrate measured after 2 hrs in presence of Adomet/[methyl-3H]Adomet by topc... | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA (cytosine-5)-methyltransferase 1 (Homo sapiens (Human)) | BDBM50506785 (CHEMBL4537137) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human DNMT1 using biotinylated DNA duplex as substrate measured after 2 hrs in presence of Adomet/[methyl-3H]Adomet by topc... | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA (cytosine-5)-methyltransferase 1 (Homo sapiens (Human)) | BDBM50506786 (CHEMBL4469152) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human DNMT1 using biotinylated DNA duplex as substrate measured after 2 hrs in presence of Adomet/[methyl-3H]Adomet by topc... | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA (cytosine-5)-methyltransferase 3A (Homo sapiens (Human)) | BDBM50506787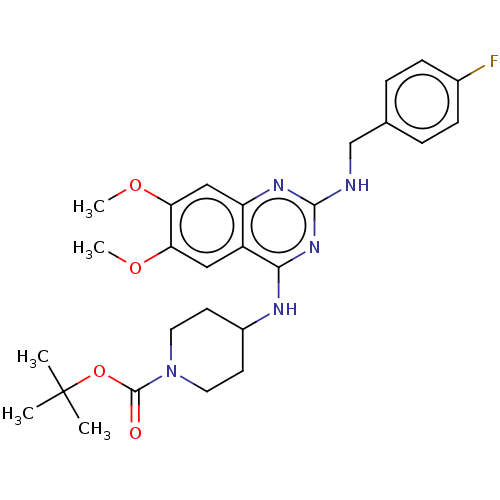 (CHEMBL4558658) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 8.70E+3 | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human DNMT3a C-terminal catalytic domain (623 to 908 residues) using 5'-biotinylated/3'-FAM-oligonucleotide as substrate me... | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA (cytosine-5)-methyltransferase 3A (Homo sapiens (Human)) | BDBM50506788 (CHEMBL4526979) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 5.40E+3 | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human DNMT3a C-terminal catalytic domain (623 to 908 residues) using 5'-biotinylated/3'-FAM-oligonucleotide as substrate me... | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA (cytosine-5)-methyltransferase 3A (Homo sapiens (Human)) | BDBM50506789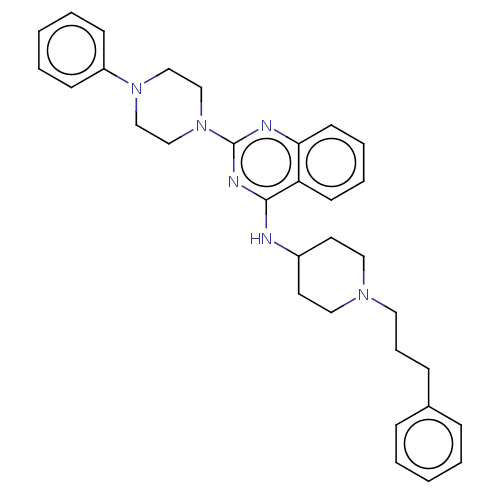 (CHEMBL4470318) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 5.10E+3 | n/a | n/a | n/a | n/a |
Universit£ de Lille Curated by ChEMBL | Assay Description Inhibition of recombinant human DNMT3a C-terminal catalytic domain (623 to 908 residues) using 5'-biotinylated/3'-FAM-oligonucleotide as substrate me... | Eur J Med Chem 161: 277-291 (2019) Article DOI: 10.1016/j.ejmech.2018.10.041 BindingDB Entry DOI: 10.7270/Q21V5J76 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 65 total ) | Next | Last >> |