 Found 35 hits with Last Name = 'ferreira' and Initial = 'lg'
Found 35 hits with Last Name = 'ferreira' and Initial = 'lg' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8960

((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...)Show SMILES COc1cc2CC(CC3CCN(Cc4ccccc4)CC3)C(=O)c2cc1OC Show InChI InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of AChE (unknown origin) using acetylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition and measured ... |
Citation and Details
Article DOI: 10.1039/d0md00009d
BindingDB Entry DOI: 10.7270/Q2C82DXM |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590189
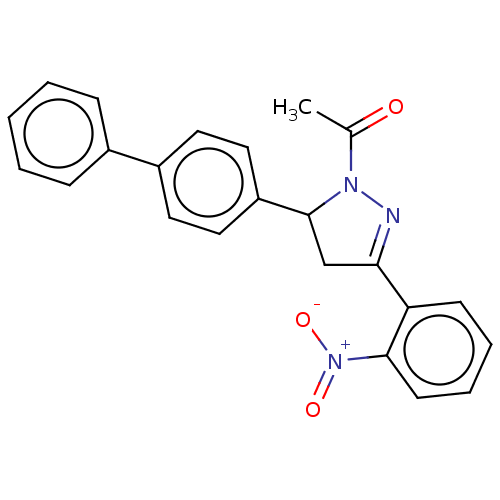
(CHEMBL5191187)Show SMILES CC(=O)N1N=C(CC1c1ccc(cc1)-c1ccccc1)c1ccccc1[N+]([O-])=O |c:4| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 52 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590196
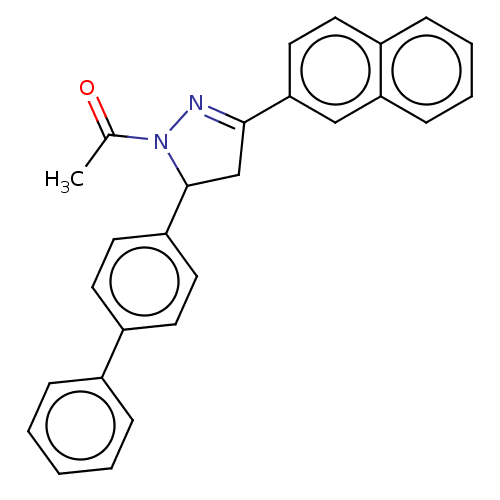
(CHEMBL5187436)Show SMILES CC(=O)N1N=C(CC1c1ccc(cc1)-c1ccccc1)c1ccc2ccccc2c1 |c:4| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 68 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8960

((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...)Show SMILES COc1cc2CC(CC3CCN(Cc4ccccc4)CC3)C(=O)c2cc1OC Show InChI InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 87 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8961

(1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...)Show InChI InChI=1S/C13H14N2/c14-13-9-5-1-3-7-11(9)15-12-8-4-2-6-10(12)13/h1,3,5,7H,2,4,6,8H2,(H2,14,15) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 178 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50004000

((3aS,8aR)-1,3a,8-trimethyl-1,2,3,3a,8,8a-hexahydro...)Show SMILES CNC(=O)Oc1ccc2N(C)[C@H]3N(C)CC[C@@]3(C)c2c1 |r| Show InChI InChI=1S/C15H21N3O2/c1-15-7-8-17(3)13(15)18(4)12-6-5-10(9-11(12)15)20-14(19)16-2/h5-6,9,13H,7-8H2,1-4H3,(H,16,19)/t13-,15+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Sodium/potassium-transporting ATPase subunit alpha-1
(Homo sapiens (Human)) | BDBM46355
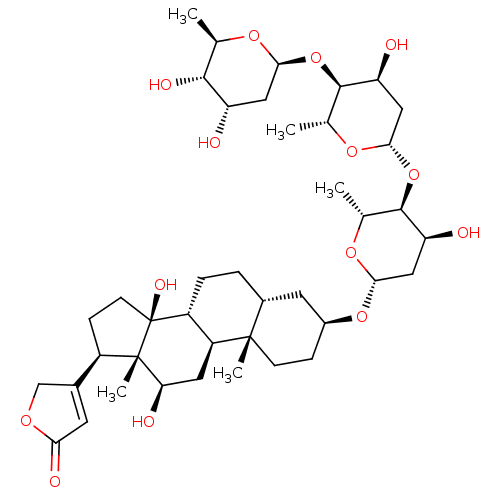
(DIGOXIN | MLS000069819 | SMR000059217 | US10668094...)Show SMILES C[C@H]1O[C@H](C[C@H](O)[C@@H]1O)O[C@H]1[C@@H](O)C[C@H](O[C@H]2[C@@H](O)C[C@H](O[C@H]3CC[C@@]4(C)[C@H](CC[C@@H]5[C@@H]4C[C@@H](O)[C@]4(C)[C@H](CC[C@]54O)C4=CC(=O)OC4)C3)O[C@@H]2C)O[C@@H]1C |t:46| Show InChI InChI=1S/C41H64O14/c1-19-36(47)28(42)15-34(50-19)54-38-21(3)52-35(17-30(38)44)55-37-20(2)51-33(16-29(37)43)53-24-8-10-39(4)23(13-24)6-7-26-27(39)14-31(45)40(5)25(9-11-41(26,40)48)22-12-32(46)49-18-22/h12,19-21,23-31,33-38,42-45,47-48H,6-11,13-18H2,1-5H3/t19-,20-,21-,23-,24+,25-,26-,27+,28+,29+,30+,31-,33+,34+,35+,36-,37-,38-,39+,40+,41+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade Federal de S£o Jo£o Del Rei
Curated by ChEMBL
| Assay Description
Inhibition of human kidney Na(+)/K(+) ATPase alpha-1 assessed as amount of Pi release after 1 hr by colorimetric method |
Bioorg Med Chem 23: 4397-404 (2015)
Article DOI: 10.1016/j.bmc.2015.06.028
BindingDB Entry DOI: 10.7270/Q2MG7R8C |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Sodium/potassium-transporting ATPase subunit alpha-1
(Homo sapiens (Human)) | BDBM50096255
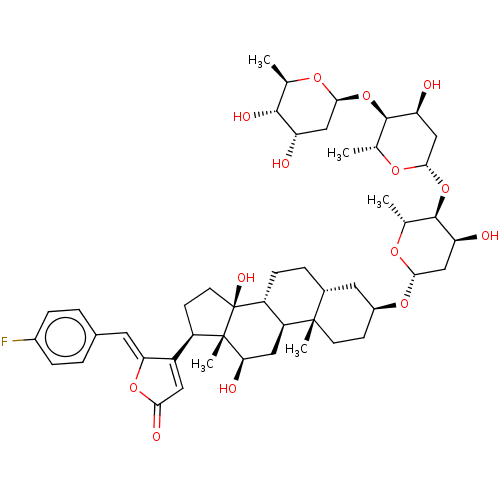
(CHEMBL3594278)Show SMILES [H][C@@]1(C[C@H](O)[C@H](O[C@@]2([H])C[C@H](O)[C@H](O[C@@]3([H])C[C@H](O)[C@H](O)[C@@H](C)O3)[C@@H](C)O2)[C@@H](C)O1)O[C@H]1CC[C@@]2(C)[C@]([H])(CC[C@]3([H])[C@]2([H])C[C@@H](O)[C@]2(C)[C@H](CC[C@]32O)C2=CC(=O)O\C2=C/c2ccc(F)cc2)C1 |r,t:60| Show InChI InChI=1S/C35H52N6O5/c1-4-23(3)31-35(46)41-19-12-11-17-30(41)34(45)37-27(15-8-6-7-13-25(42)5-2)32(43)38-28(33(44)39-31)21-24-22-40(20-18-36)29-16-10-9-14-26(24)29/h9-10,14,16,22-23,27-28,30-31H,4-8,11-13,15,17-21,36H2,1-3H3,(H,37,45)(H,38,43)(H,39,44)/t23?,27-,28-,30+,31-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade Federal de S£o Jo£o Del Rei
Curated by ChEMBL
| Assay Description
Inhibition of human kidney Na(+)/K(+) ATPase alpha-1 assessed as amount of Pi release after 1 hr by colorimetric method |
Bioorg Med Chem 23: 4397-404 (2015)
Article DOI: 10.1016/j.bmc.2015.06.028
BindingDB Entry DOI: 10.7270/Q2MG7R8C |
More data for this
Ligand-Target Pair | |
Sodium/potassium-transporting ATPase subunit alpha-1
(Homo sapiens (Human)) | BDBM50096254

(CHEMBL3594276)Show SMILES [H][C@@]1(C[C@H](O)[C@H](O[C@@]2([H])C[C@H](O)[C@H](O[C@@]3([H])C[C@H](O)[C@H](O)[C@@H](C)O3)[C@@H](C)O2)[C@@H](C)O1)O[C@H]1CC[C@@]2(C)[C@]([H])(CC[C@]3([H])[C@]2([H])C[C@@H](O)[C@]2(C)[C@H](CC[C@]32O)C2=CC(=O)O\C2=C/c2ccc(OC)cc2)C1 |r,t:60| Show InChI InChI=1S/C33H45N5O6/c1-4-20(3)27-33(44)38-18-12-11-17-26(38)31(42)35-25(16-8-6-7-13-21(39)5-2)30(41)37-28(32(43)36-27)23-19-34-24-15-10-9-14-22(24)29(23)40/h9-10,14-15,19-20,25-28H,4-8,11-13,16-18H2,1-3H3,(H,34,40)(H,35,42)(H,36,43)(H,37,41)/t20?,25-,26+,27-,28+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 570 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade Federal de S£o Jo£o Del Rei
Curated by ChEMBL
| Assay Description
Inhibition of human kidney Na(+)/K(+) ATPase alpha-1 assessed as amount of Pi release after 1 hr by colorimetric method |
Bioorg Med Chem 23: 4397-404 (2015)
Article DOI: 10.1016/j.bmc.2015.06.028
BindingDB Entry DOI: 10.7270/Q2MG7R8C |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590180
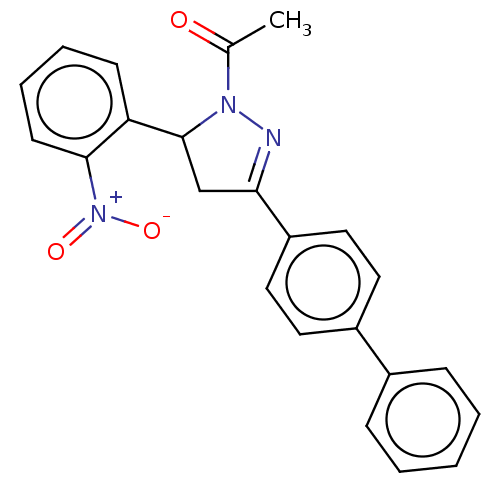
(CHEMBL5179832)Show SMILES CC(=O)N1N=C(CC1c1ccccc1[N+]([O-])=O)c1ccc(cc1)-c1ccccc1 |c:4| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 640 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590187
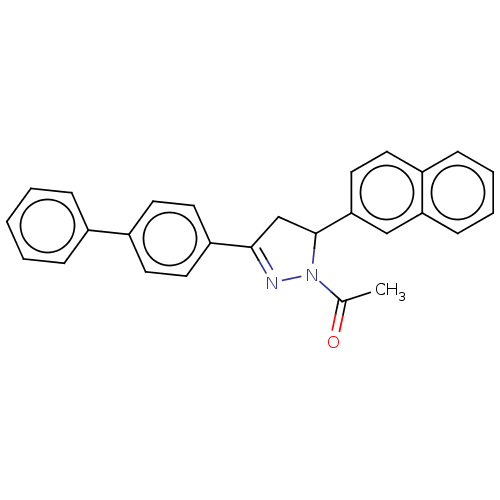
(CHEMBL5197832)Show SMILES CC(=O)N1N=C(CC1c1ccc2ccccc2c1)c1ccc(cc1)-c1ccccc1 |c:4| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 799 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590188
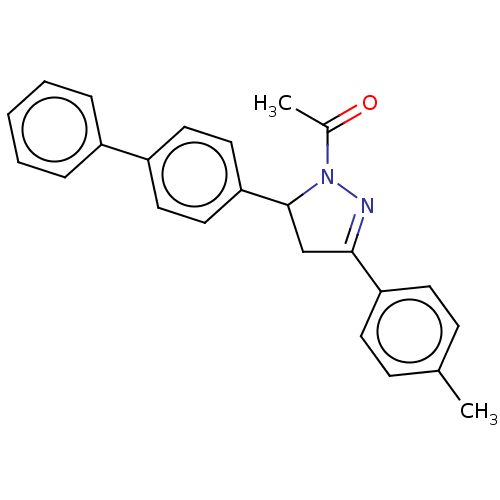
(CHEMBL5189505)Show SMILES CC(=O)N1N=C(CC1c1ccc(cc1)-c1ccccc1)c1ccc(C)cc1 |c:4| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 845 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590190

(CHEMBL5177419)Show SMILES CC(=O)N1N=C(CC1c1ccc(cc1)-c1ccccc1)c1ccc(Br)cc1 |c:4| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 903 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590194
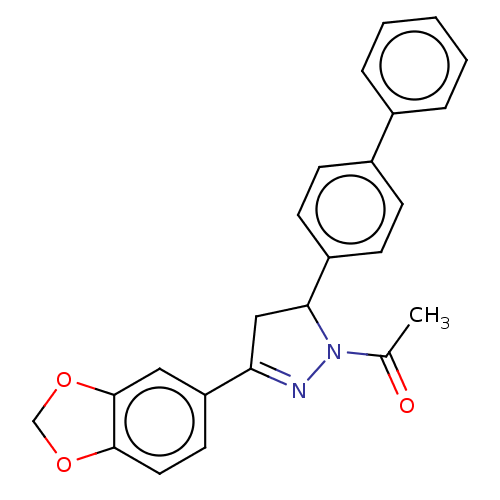
(CHEMBL5184984)Show SMILES CC(=O)N1N=C(CC1c1ccc(cc1)-c1ccccc1)c1ccc2OCOc2c1 |c:4| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 912 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590186

(CHEMBL5188500)Show SMILES CC(=O)N1N=C(CC1c1ccc2OCOc2c1)c1ccc(cc1)-c1ccccc1 |c:4| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 988 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50548032

(CHEMBL4785620) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.64E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of AChE (unknown origin) using acetylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition and measured ... |
Citation and Details
Article DOI: 10.1039/d0md00009d
BindingDB Entry DOI: 10.7270/Q2C82DXM |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50548029
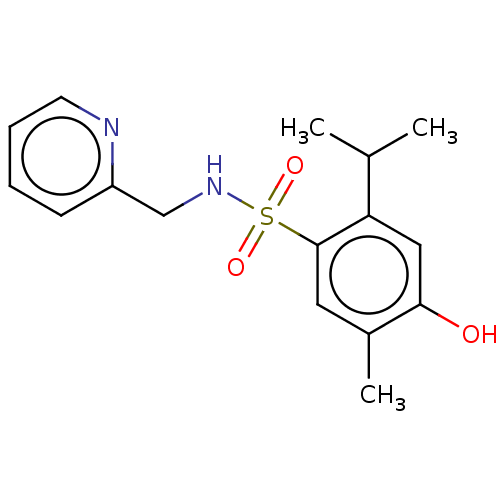
(CHEMBL4797608) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.99E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of AChE (unknown origin) using acetylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition and measured ... |
Citation and Details
Article DOI: 10.1039/d0md00009d
BindingDB Entry DOI: 10.7270/Q2C82DXM |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50548031

(CHEMBL4741157) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.34E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of AChE (unknown origin) using acetylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition and measured ... |
Citation and Details
Article DOI: 10.1039/d0md00009d
BindingDB Entry DOI: 10.7270/Q2C82DXM |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50548028
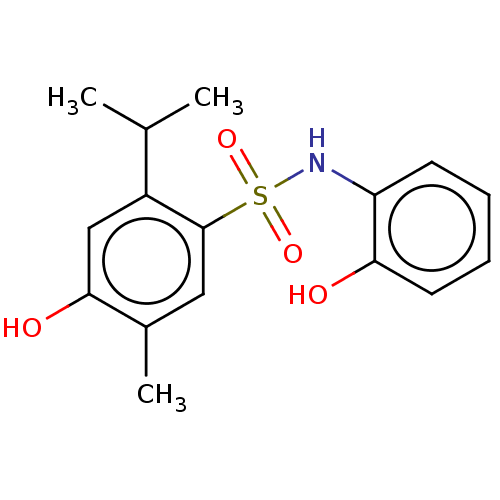
(CHEMBL4800270) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8.74E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of AChE (unknown origin) using acetylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition and measured ... |
Citation and Details
Article DOI: 10.1039/d0md00009d
BindingDB Entry DOI: 10.7270/Q2C82DXM |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50548030
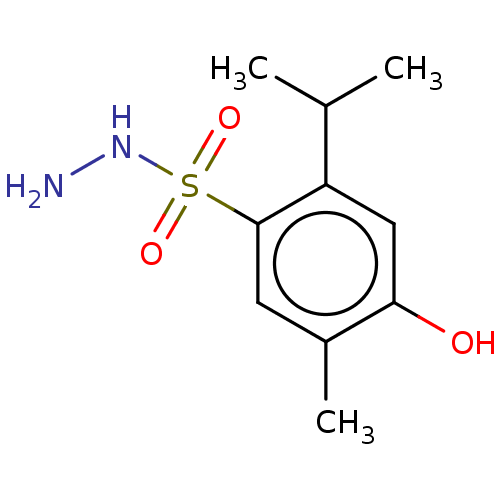
(CHEMBL4762463) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9.24E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of AChE (unknown origin) using acetylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition and measured ... |
Citation and Details
Article DOI: 10.1039/d0md00009d
BindingDB Entry DOI: 10.7270/Q2C82DXM |
More data for this
Ligand-Target Pair | |
Sodium/potassium-transporting ATPase subunit alpha-1
(Homo sapiens (Human)) | BDBM50096256
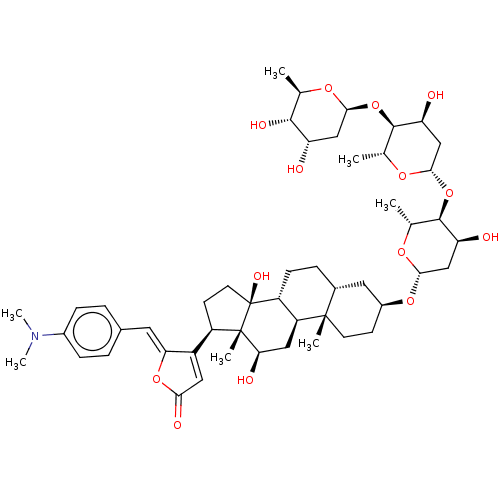
(CHEMBL3594279)Show SMILES [H][C@@]1(C[C@H](O)[C@H](O[C@@]2([H])C[C@H](O)[C@H](O[C@@]3([H])C[C@H](O)[C@H](O)[C@@H](C)O3)[C@@H](C)O2)[C@@H](C)O1)O[C@H]1CC[C@@]2(C)[C@]([H])(CC[C@]3([H])[C@]2([H])C[C@@H](O)[C@]2(C)[C@H](CC[C@]32O)C2=CC(=O)O\C2=C/c2ccc(cc2)N(C)C)C1 |r,t:60| Show InChI InChI=1S/C50H73NO14/c1-25-45(57)36(52)22-43(59-25)64-47-27(3)61-44(24-38(47)54)65-46-26(2)60-42(23-37(46)53)62-31-14-16-48(4)29(19-31)10-13-34-35(48)21-40(55)49(5)33(15-17-50(34,49)58)32-20-41(56)63-39(32)18-28-8-11-30(12-9-28)51(6)7/h8-9,11-12,18,20,25-27,29,31,33-38,40,42-47,52-55,57-58H,10,13-17,19,21-24H2,1-7H3/b39-18-/t25-,26-,27-,29-,31+,33-,34-,35+,36+,37+,38+,40-,42+,43+,44+,45-,46-,47-,48+,49+,50+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade Federal de S£o Jo£o Del Rei
Curated by ChEMBL
| Assay Description
Inhibition of human kidney Na(+)/K(+) ATPase alpha-1 assessed as amount of Pi release after 1 hr by colorimetric method |
Bioorg Med Chem 23: 4397-404 (2015)
Article DOI: 10.1016/j.bmc.2015.06.028
BindingDB Entry DOI: 10.7270/Q2MG7R8C |
More data for this
Ligand-Target Pair | |
Sodium/potassium-transporting ATPase subunit alpha-1
(Homo sapiens (Human)) | BDBM50096257
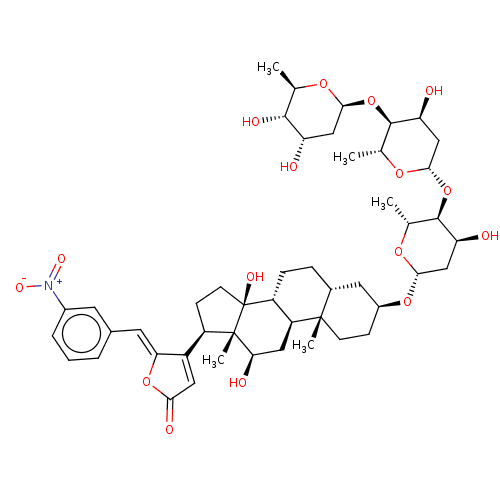
(CHEMBL3594280)Show SMILES [H][C@@]1(C[C@H](O)[C@H](O[C@@]2([H])C[C@H](O)[C@H](O[C@@]3([H])C[C@H](O)[C@H](O)[C@@H](C)O3)[C@@H](C)O2)[C@@H](C)O1)O[C@H]1CC[C@@]2(C)[C@]([H])(CC[C@]3([H])[C@]2([H])C[C@@H](O)[C@]2(C)[C@H](CC[C@]32O)C2=CC(=O)O\C2=C/c2cccc(c2)[N+]([O-])=O)C1 |r,t:60| Show InChI InChI=1S/C48H67NO16/c1-23-43(55)34(50)20-41(59-23)64-45-25(3)61-42(22-36(45)52)65-44-24(2)60-40(21-35(44)51)62-29-11-13-46(4)27(17-29)9-10-32-33(46)19-38(53)47(5)31(12-14-48(32,47)56)30-18-39(54)63-37(30)16-26-7-6-8-28(15-26)49(57)58/h6-8,15-16,18,23-25,27,29,31-36,38,40-45,50-53,55-56H,9-14,17,19-22H2,1-5H3/b37-16-/t23-,24-,25-,27-,29+,31-,32-,33+,34+,35+,36+,38-,40+,41+,42+,43-,44-,45-,46+,47+,48+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade Federal de S£o Jo£o Del Rei
Curated by ChEMBL
| Assay Description
Inhibition of human kidney Na(+)/K(+) ATPase alpha-1 assessed as amount of Pi release after 1 hr by colorimetric method |
Bioorg Med Chem 23: 4397-404 (2015)
Article DOI: 10.1016/j.bmc.2015.06.028
BindingDB Entry DOI: 10.7270/Q2MG7R8C |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM10404

((1S,12S,14R)-9-methoxy-4-methyl-11-oxa-4-azatetrac...)Show SMILES COc1ccc2CN(C)CC[C@@]34C=C[C@H](O)C[C@@H]3Oc1c24 |r,c:12| Show InChI InChI=1S/C17H21NO3/c1-18-8-7-17-6-5-12(19)9-14(17)21-16-13(20-2)4-3-11(10-18)15(16)17/h3-6,12,14,19H,7-10H2,1-2H3/t12-,14-,17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 1.71E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of AChE (unknown origin) using acetylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition and measured ... |
Citation and Details
Article DOI: 10.1039/d0md00009d
BindingDB Entry DOI: 10.7270/Q2C82DXM |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50240433
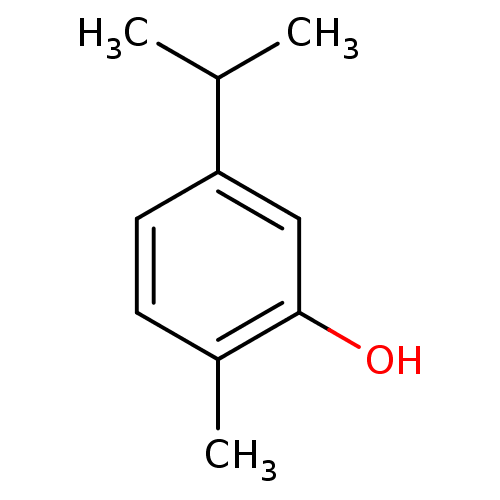
(5-Isopropyl-2-methyl-phenol | CARVACROL | CHEMBL28...)Show InChI InChI=1S/C10H14O/c1-7(2)9-5-4-8(3)10(11)6-9/h4-7,11H,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.84E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of AChE (unknown origin) using acetylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition and measured ... |
Citation and Details
Article DOI: 10.1039/d0md00009d
BindingDB Entry DOI: 10.7270/Q2C82DXM |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590195
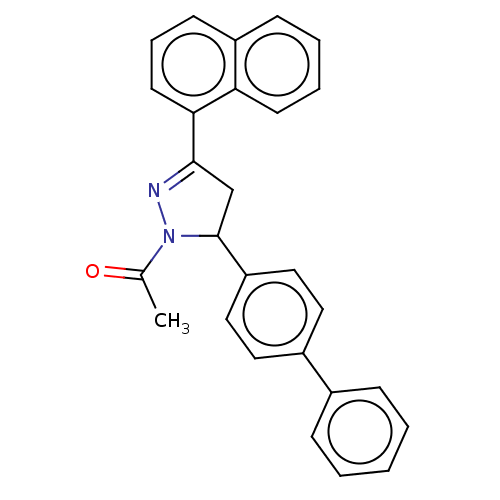
(CHEMBL5185580)Show SMILES CC(=O)N1N=C(CC1c1ccc(cc1)-c1ccccc1)c1cccc2ccccc12 |c:4| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.01E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590181
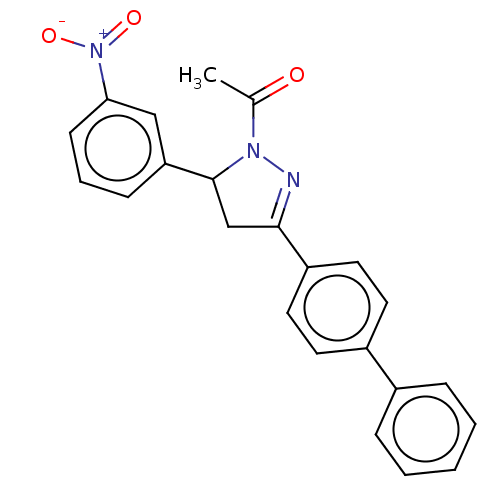
(CHEMBL5175686)Show SMILES CC(=O)N1N=C(CC1c1cccc(c1)[N+]([O-])=O)c1ccc(cc1)-c1ccccc1 |c:4| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.02E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590192
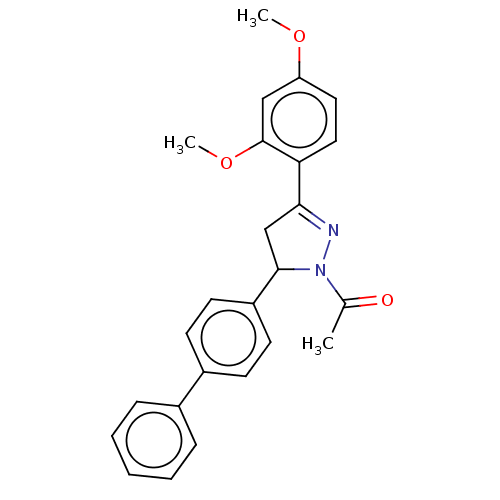
(CHEMBL5183314)Show SMILES COc1ccc(C2=NN(C(C2)c2ccc(cc2)-c2ccccc2)C(C)=O)c(OC)c1 |t:6| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.12E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590179

(CHEMBL5169783)Show SMILES CC(=O)N1N=C(CC1c1ccccc1)c1ccc(cc1)-c1ccccc1 |c:4| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.32E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590184

(CHEMBL5208439)Show SMILES COc1ccc(OC)c(c1)C1CC(=NN1C(C)=O)c1ccc(cc1)-c1ccccc1 |c:13| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.42E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590185
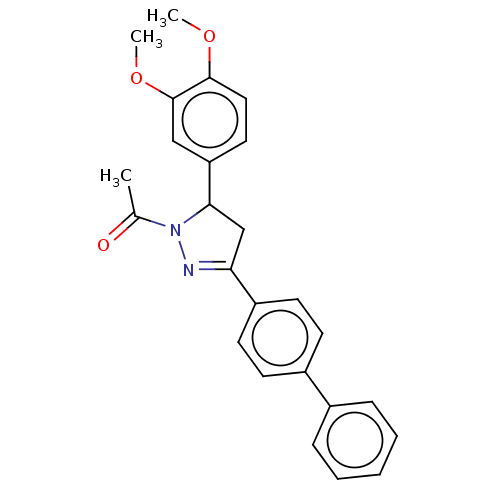
(CHEMBL5169502)Show SMILES COc1ccc(cc1OC)C1CC(=NN1C(C)=O)c1ccc(cc1)-c1ccccc1 |c:13| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.42E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590183

(CHEMBL5171946)Show SMILES CC(=O)N1N=C(CC1c1ccc(cc1)C(O)=O)c1ccc(cc1)-c1ccccc1 |c:4| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.54E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590182
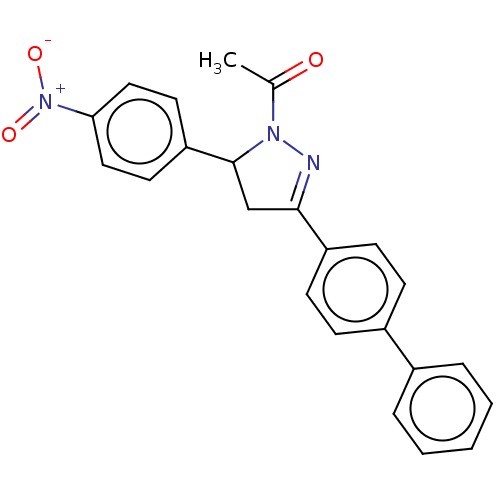
(CHEMBL5182185)Show SMILES CC(=O)N1N=C(CC1c1ccc(cc1)[N+]([O-])=O)c1ccc(cc1)-c1ccccc1 |c:4| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.55E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590193

(CHEMBL5191224)Show SMILES COc1ccc(OC)c(c1)C1=NN(C(C1)c1ccc(cc1)-c1ccccc1)C(C)=O |t:11| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.70E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM10404

((1S,12S,14R)-9-methoxy-4-methyl-11-oxa-4-azatetrac...)Show SMILES COc1ccc2CN(C)CC[C@@]34C=C[C@H](O)C[C@@H]3Oc1c24 |r,c:12| Show InChI InChI=1S/C17H21NO3/c1-18-8-7-17-6-5-12(19)9-14(17)21-16-13(20-2)4-3-11(10-18)15(16)17/h3-6,12,14,19H,7-10H2,1-2H3/t12-,14-,17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 1.72E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50590191
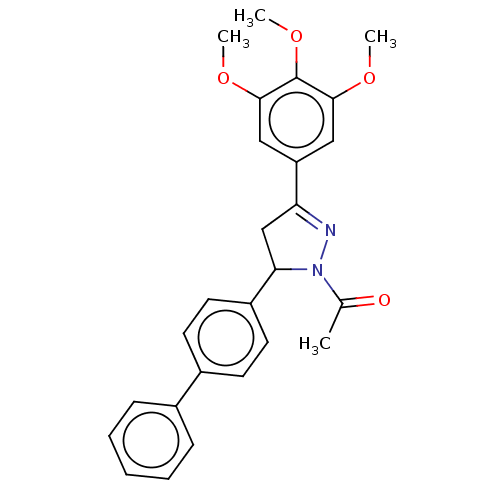
(CHEMBL5184154)Show SMILES COc1cc(cc(OC)c1OC)C1=NN(C(C1)c1ccc(cc1)-c1ccccc1)C(C)=O |t:13| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.94E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1039/d2md00262k
BindingDB Entry DOI: 10.7270/Q2J67MWD |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data