 Found 69 hits with Last Name = 'gan' and Initial = 'ls'
Found 69 hits with Last Name = 'gan' and Initial = 'ls' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50352206
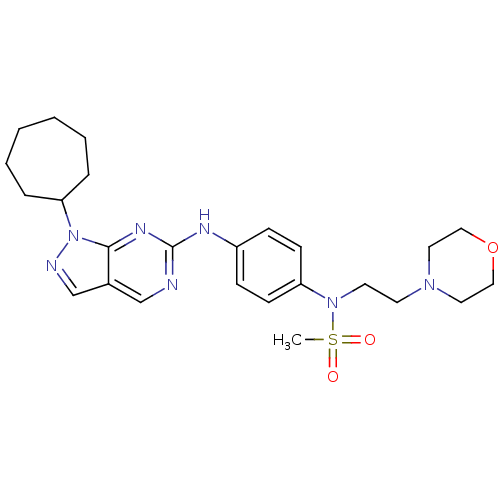
(CHEMBL1825089)Show SMILES CS(=O)(=O)N(CCN1CCOCC1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C25H35N7O3S/c1-36(33,34)31(13-12-30-14-16-35-17-15-30)22-10-8-21(9-11-22)28-25-26-18-20-19-27-32(24(20)29-25)23-6-4-2-3-5-7-23/h8-11,18-19,23H,2-7,12-17H2,1H3,(H,26,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 using a fluorescent probe 7-methoxy-4-trifluoromethylcoumarin |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352202

(CHEMBL1825101)Show SMILES CS(=O)(=O)N(CCN)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cn1 Show InChI InChI=1S/C20H28N8O2S/c1-31(29,30)27(11-10-21)18-9-8-16(14-22-18)25-20-23-12-15-13-24-28(19(15)26-20)17-6-4-2-3-5-7-17/h8-9,12-14,17H,2-7,10-11,21H2,1H3,(H,23,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352200

(CHEMBL1825100)Show SMILES CS(=O)(=O)N(CCO)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cn1 Show InChI InChI=1S/C20H27N7O3S/c1-31(29,30)26(10-11-28)18-9-8-16(14-21-18)24-20-22-12-15-13-23-27(19(15)25-20)17-6-4-2-3-5-7-17/h8-9,12-14,17,28H,2-7,10-11H2,1H3,(H,22,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352208
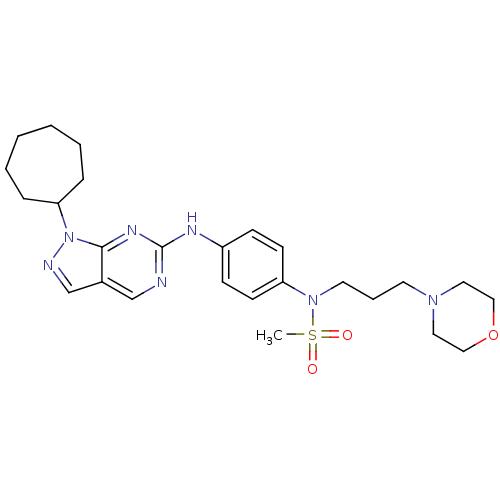
(CHEMBL1825090)Show SMILES CS(=O)(=O)N(CCCN1CCOCC1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C26H37N7O3S/c1-37(34,35)32(14-6-13-31-15-17-36-18-16-31)23-11-9-22(10-12-23)29-26-27-19-21-20-28-33(25(21)30-26)24-7-4-2-3-5-8-24/h9-12,19-20,24H,2-8,13-18H2,1H3,(H,27,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352209

(CHEMBL1825092)Show SMILES CC(C)N(c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1)S(C)(=O)=O Show InChI InChI=1S/C22H30N6O2S/c1-16(2)28(31(3,29)30)20-12-10-18(11-13-20)25-22-23-14-17-15-24-27(21(17)26-22)19-8-6-4-5-7-9-19/h10-16,19H,4-9H2,1-3H3,(H,23,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352208

(CHEMBL1825090)Show SMILES CS(=O)(=O)N(CCCN1CCOCC1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C26H37N7O3S/c1-37(34,35)32(14-6-13-31-15-17-36-18-16-31)23-11-9-22(10-12-23)29-26-27-19-21-20-28-33(25(21)30-26)24-7-4-2-3-5-8-24/h9-12,19-20,24H,2-8,13-18H2,1H3,(H,27,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352215

(CHEMBL1825093)Show SMILES CS(=O)(=O)N(CCN)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C21H29N7O2S/c1-31(29,30)27(13-12-22)18-10-8-17(9-11-18)25-21-23-14-16-15-24-28(20(16)26-21)19-6-4-2-3-5-7-19/h8-11,14-15,19H,2-7,12-13,22H2,1H3,(H,23,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352212

(CHEMBL1821761)Show SMILES CS(=O)(=O)N(CCO)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C21H28N6O3S/c1-31(29,30)26(12-13-28)18-10-8-17(9-11-18)24-21-22-14-16-15-23-27(20(16)25-21)19-6-4-2-3-5-7-19/h8-11,14-15,19,28H,2-7,12-13H2,1H3,(H,22,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352210

(CHEMBL1825091)Show SMILES CS(=O)(=O)N(CCCO)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C22H30N6O3S/c1-32(30,31)27(13-6-14-29)19-11-9-18(10-12-19)25-22-23-15-17-16-24-28(21(17)26-22)20-7-4-2-3-5-8-20/h9-12,15-16,20,29H,2-8,13-14H2,1H3,(H,23,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352211
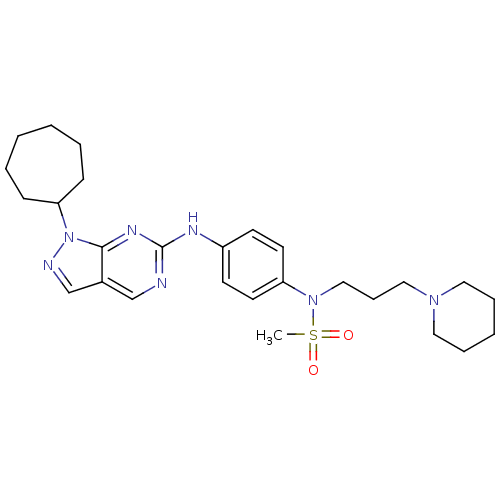
(CHEMBL1825095)Show SMILES CS(=O)(=O)N(CCCN1CCCCC1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C27H39N7O2S/c1-37(35,36)33(19-9-18-32-16-7-4-8-17-32)24-14-12-23(13-15-24)30-27-28-20-22-21-29-34(26(22)31-27)25-10-5-2-3-6-11-25/h12-15,20-21,25H,2-11,16-19H2,1H3,(H,28,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352210

(CHEMBL1825091)Show SMILES CS(=O)(=O)N(CCCO)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C22H30N6O3S/c1-32(30,31)27(13-6-14-29)19-11-9-18(10-12-19)25-22-23-15-17-16-24-28(21(17)26-22)20-7-4-2-3-5-8-20/h9-12,15-16,20,29H,2-8,13-14H2,1H3,(H,23,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352205
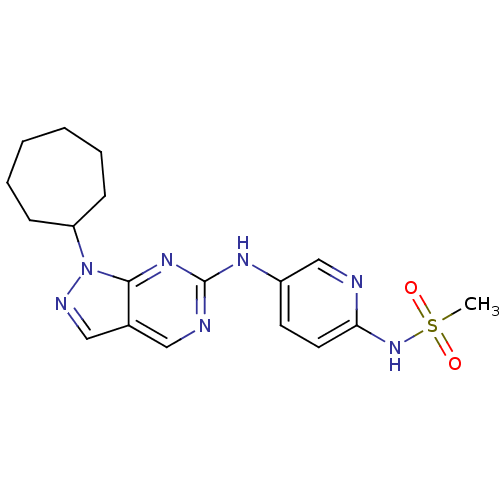
(CHEMBL1825096)Show SMILES CS(=O)(=O)Nc1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cn1 Show InChI InChI=1S/C18H23N7O2S/c1-28(26,27)24-16-9-8-14(12-19-16)22-18-20-10-13-11-21-25(17(13)23-18)15-6-4-2-3-5-7-15/h8-12,15H,2-7H2,1H3,(H,19,24)(H,20,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352211

(CHEMBL1825095)Show SMILES CS(=O)(=O)N(CCCN1CCCCC1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C27H39N7O2S/c1-37(35,36)33(19-9-18-32-16-7-4-8-17-32)24-14-12-23(13-15-24)30-27-28-20-22-21-29-34(26(22)31-27)25-10-5-2-3-6-11-25/h12-15,20-21,25H,2-11,16-19H2,1H3,(H,28,30,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352204
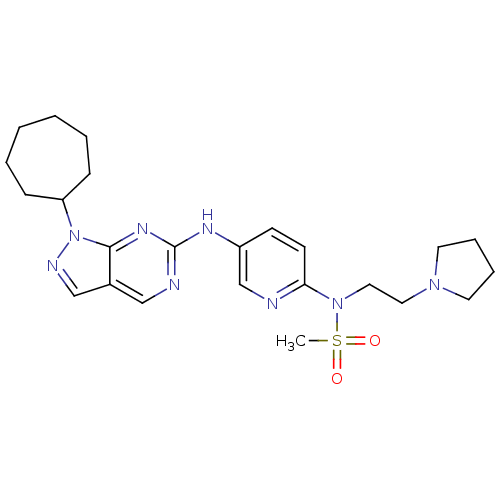
(CHEMBL1825098)Show SMILES CS(=O)(=O)N(CCN1CCCC1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cn1 Show InChI InChI=1S/C24H34N8O2S/c1-35(33,34)31(15-14-30-12-6-7-13-30)22-11-10-20(18-25-22)28-24-26-16-19-17-27-32(23(19)29-24)21-8-4-2-3-5-9-21/h10-11,16-18,21H,2-9,12-15H2,1H3,(H,26,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352203

(CHEMBL1825099)Show SMILES CN(C)CCN(c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cn1)S(C)(=O)=O Show InChI InChI=1S/C22H32N8O2S/c1-28(2)12-13-29(33(3,31)32)20-11-10-18(16-23-20)26-22-24-14-17-15-25-30(21(17)27-22)19-8-6-4-5-7-9-19/h10-11,14-16,19H,4-9,12-13H2,1-3H3,(H,24,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352212

(CHEMBL1821761)Show SMILES CS(=O)(=O)N(CCO)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C21H28N6O3S/c1-31(29,30)26(12-13-28)18-10-8-17(9-11-18)24-21-22-14-16-15-23-27(20(16)25-21)19-6-4-2-3-5-7-19/h8-11,14-15,19,28H,2-7,12-13H2,1H3,(H,22,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352214
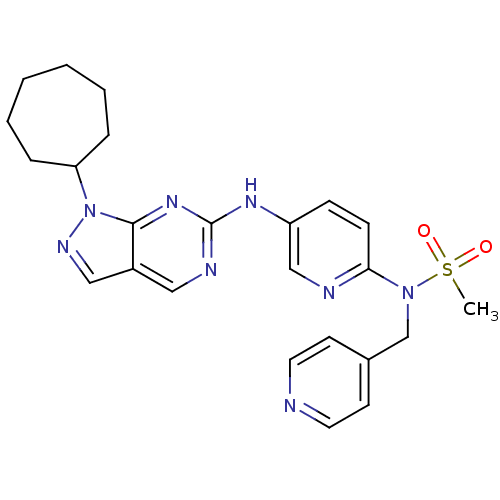
(CHEMBL1825102)Show SMILES CS(=O)(=O)N(Cc1ccncc1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cn1 Show InChI InChI=1S/C24H28N8O2S/c1-35(33,34)31(17-18-10-12-25-13-11-18)22-9-8-20(16-26-22)29-24-27-14-19-15-28-32(23(19)30-24)21-6-4-2-3-5-7-21/h8-16,21H,2-7,17H2,1H3,(H,27,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352206

(CHEMBL1825089)Show SMILES CS(=O)(=O)N(CCN1CCOCC1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C25H35N7O3S/c1-36(33,34)31(13-12-30-14-16-35-17-15-30)22-10-8-21(9-11-22)28-25-26-18-20-19-27-32(24(20)29-25)23-6-4-2-3-5-7-23/h8-11,18-19,23H,2-7,12-17H2,1H3,(H,26,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352205

(CHEMBL1825096)Show SMILES CS(=O)(=O)Nc1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cn1 Show InChI InChI=1S/C18H23N7O2S/c1-28(26,27)24-16-9-8-14(12-19-16)22-18-20-10-13-11-21-25(17(13)23-18)15-6-4-2-3-5-7-15/h8-12,15H,2-7H2,1H3,(H,19,24)(H,20,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352215

(CHEMBL1825093)Show SMILES CS(=O)(=O)N(CCN)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C21H29N7O2S/c1-31(29,30)27(13-12-22)18-10-8-17(9-11-18)25-21-23-14-16-15-24-28(20(16)26-21)19-6-4-2-3-5-7-19/h8-11,14-15,19H,2-7,12-13,22H2,1H3,(H,23,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352213
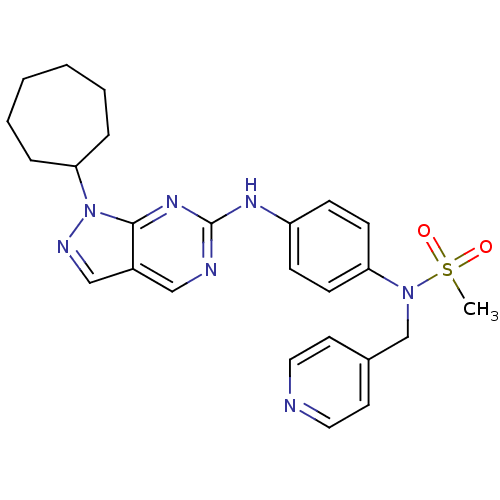
(CHEMBL1825094)Show SMILES CS(=O)(=O)N(Cc1ccncc1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C25H29N7O2S/c1-35(33,34)31(18-19-12-14-26-15-13-19)22-10-8-21(9-11-22)29-25-27-16-20-17-28-32(24(20)30-25)23-6-4-2-3-5-7-23/h8-17,23H,2-7,18H2,1H3,(H,27,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352206

(CHEMBL1825089)Show SMILES CS(=O)(=O)N(CCN1CCOCC1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C25H35N7O3S/c1-36(33,34)31(13-12-30-14-16-35-17-15-30)22-10-8-21(9-11-22)28-25-26-18-20-19-27-32(24(20)29-25)23-6-4-2-3-5-7-23/h8-11,18-19,23H,2-7,12-17H2,1H3,(H,26,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50352206

(CHEMBL1825089)Show SMILES CS(=O)(=O)N(CCN1CCOCC1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C25H35N7O3S/c1-36(33,34)31(13-12-30-14-16-35-17-15-30)22-10-8-21(9-11-22)28-25-26-18-20-19-27-32(24(20)29-25)23-6-4-2-3-5-7-23/h8-11,18-19,23H,2-7,12-17H2,1H3,(H,26,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352213

(CHEMBL1825094)Show SMILES CS(=O)(=O)N(Cc1ccncc1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C25H29N7O2S/c1-35(33,34)31(18-19-12-14-26-15-13-19)22-10-8-21(9-11-22)29-25-27-16-20-17-28-32(24(20)30-25)23-6-4-2-3-5-7-23/h8-17,23H,2-7,18H2,1H3,(H,27,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352201

(CHEMBL1825097)Show SMILES CS(=O)(=O)N(CCN1CCOCC1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cn1 Show InChI InChI=1S/C24H34N8O3S/c1-36(33,34)31(11-10-30-12-14-35-15-13-30)22-9-8-20(18-25-22)28-24-26-16-19-17-27-32(23(19)29-24)21-6-4-2-3-5-7-21/h8-9,16-18,21H,2-7,10-15H2,1H3,(H,26,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50352206

(CHEMBL1825089)Show SMILES CS(=O)(=O)N(CCN1CCOCC1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C25H35N7O3S/c1-36(33,34)31(13-12-30-14-16-35-17-15-30)22-10-8-21(9-11-22)28-25-26-18-20-19-27-32(24(20)29-25)23-6-4-2-3-5-7-23/h8-11,18-19,23H,2-7,12-17H2,1H3,(H,26,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 using a fluorescent probe 7-benzyloxy-4(trifluoromethyl)-coumarin |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50352206

(CHEMBL1825089)Show SMILES CS(=O)(=O)N(CCN1CCOCC1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1 Show InChI InChI=1S/C25H35N7O3S/c1-36(33,34)31(13-12-30-14-16-35-17-15-30)22-10-8-21(9-11-22)28-25-26-18-20-19-27-32(24(20)29-25)23-6-4-2-3-5-7-23/h8-11,18-19,23H,2-7,12-17H2,1H3,(H,26,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 using a fluorescent probe 7-benzyloxyquinoline |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
High affinity cGMP-specific 3',5'-cyclic phosphodiesterase 9A
(Homo sapiens (Human)) | BDBM50041010
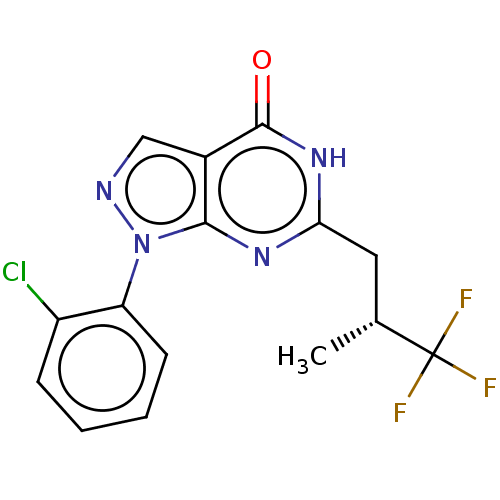
(BAY-736691 | CHEMBL1513993)Show SMILES C[C@H](Cc1nc2n(ncc2c(=O)[nH]1)-c1ccccc1Cl)C(F)(F)F Show InChI InChI=1S/C15H12ClF3N4O/c1-8(15(17,18)19)6-12-21-13-9(14(24)22-12)7-20-23(13)11-5-3-2-4-10(11)16/h2-5,7-8H,6H2,1H3,(H,21,22,24)/t8-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-sen University
Curated by ChEMBL
| Assay Description
Inhibition of recombinant PDE9A2 catalytic domain (unknown origin) expressed in Escherichia coli BL21 using [3H]cGMP as substrate after 15 mins by li... |
J Nat Prod 77: 2651-7 (2014)
Article DOI: 10.1021/np500528u
BindingDB Entry DOI: 10.7270/Q2N87CDV |
More data for this
Ligand-Target Pair | |
High affinity cGMP-specific 3',5'-cyclic phosphodiesterase 9A
(Homo sapiens (Human)) | BDBM50041010

(BAY-736691 | CHEMBL1513993)Show SMILES C[C@H](Cc1nc2n(ncc2c(=O)[nH]1)-c1ccccc1Cl)C(F)(F)F Show InChI InChI=1S/C15H12ClF3N4O/c1-8(15(17,18)19)6-12-21-13-9(14(24)22-12)7-20-23(13)11-5-3-2-4-10(11)16/h2-5,7-8H,6H2,1H3,(H,21,22,24)/t8-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 55 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-sen University
Curated by ChEMBL
| Assay Description
Inhibition of human PDE9A2 using [3H]cGMP as substrate by scintillation proximity assay |
J Nat Prod 77: 2651-7 (2014)
Article DOI: 10.1021/np500528u
BindingDB Entry DOI: 10.7270/Q2N87CDV |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50352209

(CHEMBL1825092)Show SMILES CC(C)N(c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cc1)S(C)(=O)=O Show InChI InChI=1S/C22H30N6O2S/c1-16(2)28(31(3,29)30)20-12-10-18(11-13-20)25-22-23-14-17-15-24-27(21(17)26-22)19-8-6-4-5-7-9-19/h10-16,19H,4-9H2,1-3H3,(H,23,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/Cyclin B assessed as phosphorylation of Z-lyte Peptide at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352214

(CHEMBL1825102)Show SMILES CS(=O)(=O)N(Cc1ccncc1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cn1 Show InChI InChI=1S/C24H28N8O2S/c1-35(33,34)31(17-18-10-12-25-13-11-18)22-9-8-20(16-26-22)29-24-27-14-19-15-28-32(23(19)30-24)21-6-4-2-3-5-7-21/h8-16,21H,2-7,17H2,1H3,(H,27,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 95 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352200

(CHEMBL1825100)Show SMILES CS(=O)(=O)N(CCO)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cn1 Show InChI InChI=1S/C20H27N7O3S/c1-31(29,30)26(10-11-28)18-9-8-16(14-21-18)24-20-22-12-15-13-23-27(19(15)25-20)17-6-4-2-3-5-7-17/h8-9,12-14,17,28H,2-7,10-11H2,1H3,(H,22,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352201

(CHEMBL1825097)Show SMILES CS(=O)(=O)N(CCN1CCOCC1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cn1 Show InChI InChI=1S/C24H34N8O3S/c1-36(33,34)31(11-10-30-12-14-35-15-13-30)22-9-8-20(18-25-22)28-24-26-16-19-17-27-32(23(19)29-24)21-6-4-2-3-5-7-21/h8-9,16-18,21H,2-7,10-15H2,1H3,(H,26,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352202

(CHEMBL1825101)Show SMILES CS(=O)(=O)N(CCN)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cn1 Show InChI InChI=1S/C20H28N8O2S/c1-31(29,30)27(11-10-21)18-9-8-16(14-22-18)25-20-23-12-15-13-24-28(19(15)26-20)17-6-4-2-3-5-7-17/h8-9,12-14,17H,2-7,10-11,21H2,1H3,(H,23,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352203

(CHEMBL1825099)Show SMILES CN(C)CCN(c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cn1)S(C)(=O)=O Show InChI InChI=1S/C22H32N8O2S/c1-28(2)12-13-29(33(3,31)32)20-11-10-18(16-23-20)26-22-24-14-17-15-25-30(21(17)27-22)19-8-6-4-5-7-9-19/h10-11,14-16,19H,4-9,12-13H2,1-3H3,(H,24,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50352204

(CHEMBL1825098)Show SMILES CS(=O)(=O)N(CCN1CCCC1)c1ccc(Nc2ncc3cnn(C4CCCCCC4)c3n2)cn1 Show InChI InChI=1S/C24H34N8O2S/c1-35(33,34)31(15-14-30-12-6-7-13-30)22-11-10-20(18-25-22)28-24-26-16-19-17-27-32(23(19)29-24)21-8-4-2-3-5-9-21/h10-11,16-18,21H,2-9,12-15H2,1H3,(H,26,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 350 | n/a | n/a | n/a | n/a | n/a | n/a |
Biogen Idec Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A assessed as phosphorylation of Lats2 substrate at 0.017 to 30 nM by FRET assay |
Bioorg Med Chem Lett 21: 5633-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.06.129
BindingDB Entry DOI: 10.7270/Q2HM58VW |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50199522

((+)-huperzine A | (+-)-HA | (-)-1-Amino-13-ethylid...)Show SMILES C\C=C1/[C@@H]2Cc3[nH]c(=O)ccc3[C@@]1(N)CC(C)=C2 |r,c:18,THB:1:2:14.15.17:5.11.4| Show InChI InChI=1S/C15H18N2O/c1-3-11-10-6-9(2)8-15(11,16)12-4-5-14(18)17-13(12)7-10/h3-6,10H,7-8,16H2,1-2H3,(H,17,18)/b11-3+/t10-,15+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
Xinjiang Technical Institute of Physics and Chemistry
Curated by ChEMBL
| Assay Description
Inhibition of AChE (unknown origin) using acetylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition measured afte... |
J Nat Prod 81: 2115-2119 (2018)
Article DOI: 10.1021/acs.jnatprod.8b00461
BindingDB Entry DOI: 10.7270/Q25D8VGZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50190664
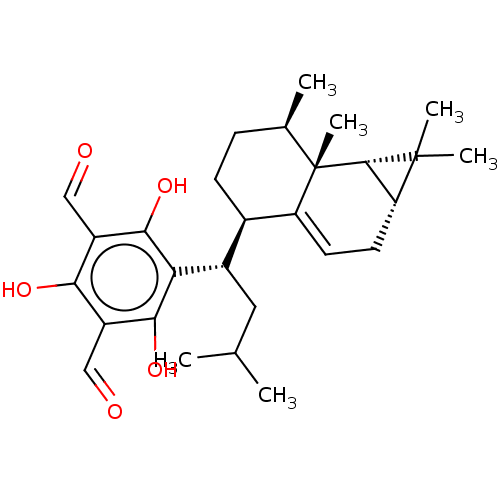
(CHEMBL3974642)Show SMILES [H][C@@]12CC=C3[C@]([H])(CC[C@@H](C)[C@]3(C)[C@]1([H])C2(C)C)[C@H](CC(C)C)c1c(O)c(C=O)c(O)c(C=O)c1O |r,t:3| Show InChI InChI=1S/C28H38O5/c1-14(2)11-17(22-24(32)18(12-29)23(31)19(13-30)25(22)33)16-8-7-15(3)28(6)20(16)9-10-21-26(28)27(21,4)5/h9,12-17,21,26,31-33H,7-8,10-11H2,1-6H3/t15-,16-,17+,21-,26+,28+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Sciences
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) using pNPP as substrate after 3 mins by colorimetric analysis |
J Nat Prod 79: 1365-72 (2016)
Article DOI: 10.1021/acs.jnatprod.6b00090
BindingDB Entry DOI: 10.7270/Q27W6F46 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50190671

(CHEMBL3935360)Show SMILES [H][C@@]12CC[C@@H](C)C3=C([C@]1([H])C2(C)C)[C@](C)(CC3)[C@@H](CC(C)C)c1c(O)c(C=O)c(O)c(C=O)c1O |r,c:6| Show InChI InChI=1S/C28H38O5/c1-14(2)11-20(21-25(32)17(12-29)24(31)18(13-30)26(21)33)28(6)10-9-16-15(3)7-8-19-23(22(16)28)27(19,4)5/h12-15,19-20,23,31-33H,7-11H2,1-6H3/t15-,19-,20+,23-,28-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Sciences
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) using pNPP as substrate after 3 mins by colorimetric analysis |
J Nat Prod 79: 1365-72 (2016)
Article DOI: 10.1021/acs.jnatprod.6b00090
BindingDB Entry DOI: 10.7270/Q27W6F46 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50190667

(CHEMBL3944321)Show SMILES [H][C@@]12CC[C@@H](C)C3=CC[C@](C)([C@@H](CC(C)C)c4c(O)c(C=O)c(O)c(C=O)c4O)[C@@]3([H])[C@]1([H])C2(C)C |r,t:6| Show InChI InChI=1S/C28H38O5/c1-14(2)11-20(21-25(32)17(12-29)24(31)18(13-30)26(21)33)28(6)10-9-16-15(3)7-8-19-23(22(16)28)27(19,4)5/h9,12-15,19-20,22-23,31-33H,7-8,10-11H2,1-6H3/t15-,19-,20+,22-,23-,28-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Sciences
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) using pNPP as substrate after 3 mins by colorimetric analysis |
J Nat Prod 79: 1365-72 (2016)
Article DOI: 10.1021/acs.jnatprod.6b00090
BindingDB Entry DOI: 10.7270/Q27W6F46 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50346601
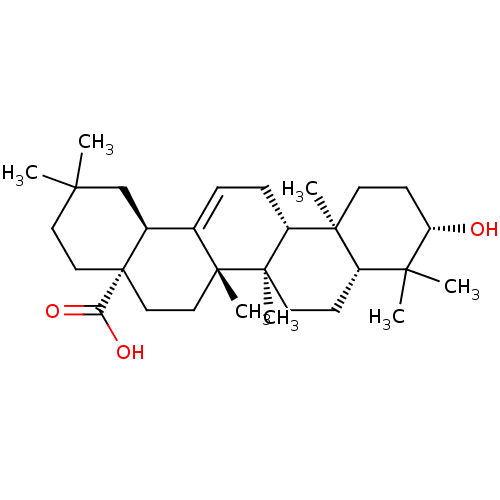
(NSC-114945 | OLEANOLIC_ACID | Oleanolic acid | Ole...)Show SMILES CC1(C)CC[C@@]2(CC[C@]3(C)C(=CC[C@@H]4[C@@]5(C)CC[C@H](O)C(C)(C)[C@@H]5CC[C@@]34C)[C@@H]2C1)C(O)=O |r,c:10| Show InChI InChI=1S/C30H48O3/c1-25(2)14-16-30(24(32)33)17-15-28(6)19(20(30)18-25)8-9-22-27(5)12-11-23(31)26(3,4)21(27)10-13-29(22,28)7/h8,20-23,31H,9-18H2,1-7H3,(H,32,33)/t20-,21-,22+,23-,27-,28+,29+,30-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Sciences
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) using pNPP as substrate after 3 mins by colorimetric analysis |
J Nat Prod 79: 1365-72 (2016)
Article DOI: 10.1021/acs.jnatprod.6b00090
BindingDB Entry DOI: 10.7270/Q27W6F46 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50459269

(CHEMBL4214417)Show SMILES [H][C@@]1([C@H](O)C[C@@]2(C)[C@]3([H])CC=C(C4=CC(=O)O[C@](O)(C[C@]12C)[C@]34C)C(C)(C)C(O)=O)[C@@](C)(O)C(=O)\C=C\C(C)(C)OC(C)=O |r,c:10,t:12| Show InChI InChI=1S/C32H44O10/c1-17(33)41-26(2,3)13-12-22(35)31(9,39)24-20(34)15-28(6)21-11-10-18(27(4,5)25(37)38)19-14-23(36)42-32(40,30(19,21)8)16-29(24,28)7/h10,12-14,20-21,24,34,39-40H,11,15-16H2,1-9H3,(H,37,38)/b13-12+/t20-,21+,24+,28+,29-,30+,31+,32-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Xinjiang Technical Institute of Physics and Chemistry
Curated by ChEMBL
| Assay Description
Inhibition of AChE (unknown origin) using acetylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition measured afte... |
J Nat Prod 81: 2115-2119 (2018)
Article DOI: 10.1021/acs.jnatprod.8b00461
BindingDB Entry DOI: 10.7270/Q25D8VGZ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50190670

(CHEMBL3931387)Show SMILES [H][C@]1(CC[C@H](C)C2=C[C@H](CC[C@@]12C)C(C)=C)[C@@H](CC(C)C)c1c(O)c(C=O)c(O)c(C=O)c1O |r,t:6| Show InChI InChI=1S/C28H38O5/c1-15(2)11-19(24-26(32)20(13-29)25(31)21(14-30)27(24)33)22-8-7-17(5)23-12-18(16(3)4)9-10-28(22,23)6/h12-15,17-19,22,31-33H,3,7-11H2,1-2,4-6H3/t17-,18-,19+,22-,28-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Sciences
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) using pNPP as substrate after 3 mins by colorimetric analysis |
J Nat Prod 79: 1365-72 (2016)
Article DOI: 10.1021/acs.jnatprod.6b00090
BindingDB Entry DOI: 10.7270/Q27W6F46 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50190662

(CHEMBL3916388)Show SMILES [H][C@@]12CC[C@@H](C)C3=CC[C@](C)([C@H](CC(C)C)c4c(O)c(C=O)c(O)c(C=O)c4O)[C@@]3([H])[C@]1([H])C2(C)C |r,t:6| Show InChI InChI=1S/C28H38O5/c1-14(2)11-20(21-25(32)17(12-29)24(31)18(13-30)26(21)33)28(6)10-9-16-15(3)7-8-19-23(22(16)28)27(19,4)5/h9,12-15,19-20,22-23,31-33H,7-8,10-11H2,1-6H3/t15-,19-,20-,22-,23-,28-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Sciences
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) using pNPP as substrate after 3 mins by colorimetric analysis |
J Nat Prod 79: 1365-72 (2016)
Article DOI: 10.1021/acs.jnatprod.6b00090
BindingDB Entry DOI: 10.7270/Q27W6F46 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50459270
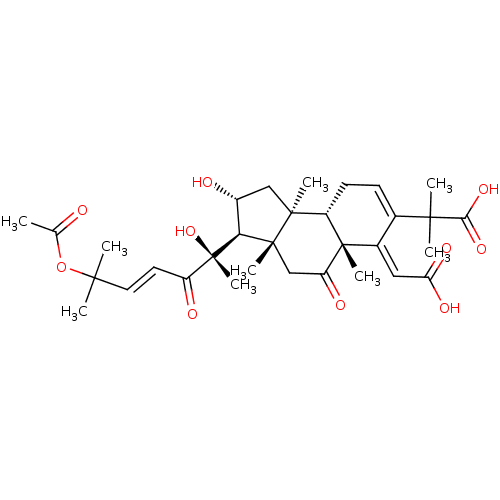
(CHEMBL4210949)Show SMILES [H][C@@]1([C@H](O)C[C@@]2(C)[C@]3([H])CC=C(\C(=C/C(O)=O)[C@]3(C)C(=O)C[C@]12C)C(C)(C)C(O)=O)[C@@](C)(O)C(=O)\C=C\C(C)(C)OC(C)=O |r,c:10| Show InChI InChI=1S/C32H44O10/c1-17(33)42-27(2,3)13-12-22(35)32(9,41)25-20(34)15-29(6)21-11-10-18(28(4,5)26(39)40)19(14-24(37)38)31(21,8)23(36)16-30(25,29)7/h10,12-14,20-21,25,34,41H,11,15-16H2,1-9H3,(H,37,38)(H,39,40)/b13-12+,19-14+/t20-,21+,25+,29+,30-,31+,32+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Xinjiang Technical Institute of Physics and Chemistry
Curated by ChEMBL
| Assay Description
Inhibition of AChE (unknown origin) using acetylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition measured afte... |
J Nat Prod 81: 2115-2119 (2018)
Article DOI: 10.1021/acs.jnatprod.8b00461
BindingDB Entry DOI: 10.7270/Q25D8VGZ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50190666

(CHEMBL3954985)Show SMILES [H][C@@]12CC[C@@](C)(OC)[C@]3([H])CC[C@](C)([C@@H](CC(C)C)c4c(O)c(C=O)c(O)c(C=O)c4O)[C@@]3([H])[C@]1([H])C2(C)C |r| Show InChI InChI=1S/C29H42O6/c1-15(2)12-20(21-25(33)16(13-30)24(32)17(14-31)26(21)34)28(5)10-8-19-23(28)22-18(27(22,3)4)9-11-29(19,6)35-7/h13-15,18-20,22-23,32-34H,8-12H2,1-7H3/t18-,19-,20+,22-,23-,28-,29-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Sciences
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) using pNPP as substrate after 3 mins by colorimetric analysis |
J Nat Prod 79: 1365-72 (2016)
Article DOI: 10.1021/acs.jnatprod.6b00090
BindingDB Entry DOI: 10.7270/Q27W6F46 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50190663

(CHEMBL3903731)Show SMILES [H][C@@]12CC=C3[C@]([H])(CC[C@@H](C)[C@]3(C)[C@]1([H])C2(C)C)[C@@H](CC(C)C)c1c(O)c(C=O)c(O)c(C=O)c1O |r,t:3| Show InChI InChI=1S/C28H38O5/c1-14(2)11-17(22-24(32)18(12-29)23(31)19(13-30)25(22)33)16-8-7-15(3)28(6)20(16)9-10-21-26(28)27(21,4)5/h9,12-17,21,26,31-33H,7-8,10-11H2,1-6H3/t15-,16-,17-,21-,26+,28+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Sciences
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) using pNPP as substrate after 3 mins by colorimetric analysis |
J Nat Prod 79: 1365-72 (2016)
Article DOI: 10.1021/acs.jnatprod.6b00090
BindingDB Entry DOI: 10.7270/Q27W6F46 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50190665

(CHEMBL3982631)Show SMILES [H][C@]12CC[C@](C)(Cc3c(O)c(C=O)c(O)c(C(=O)CC(C)C)c3O)C1=C[C@@H](CC[C@H]2C)C(C)(C)O |r,c:26| Show InChI InChI=1S/C28H40O6/c1-15(2)11-22(30)23-25(32)19(24(31)20(14-29)26(23)33)13-28(6)10-9-18-16(3)7-8-17(12-21(18)28)27(4,5)34/h12,14-18,31-34H,7-11,13H2,1-6H3/t16-,17-,18-,28-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Sciences
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) using pNPP as substrate after 3 mins by colorimetric analysis |
J Nat Prod 79: 1365-72 (2016)
Article DOI: 10.1021/acs.jnatprod.6b00090
BindingDB Entry DOI: 10.7270/Q27W6F46 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50190668

(CHEMBL3907420)Show SMILES [H][C@@]12CCC(C)=C3CC[C@](C)([C@@H](CC(C)C)c4c(O)c(C=O)c(O)c(C=O)c4O)[C@@]3([H])[C@]1([H])C2(C)C |r,t:5| Show InChI InChI=1S/C28H38O5/c1-14(2)11-20(21-25(32)17(12-29)24(31)18(13-30)26(21)33)28(6)10-9-16-15(3)7-8-19-23(22(16)28)27(19,4)5/h12-14,19-20,22-23,31-33H,7-11H2,1-6H3/t19-,20+,22-,23-,28-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Sciences
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) using pNPP as substrate after 3 mins by colorimetric analysis |
J Nat Prod 79: 1365-72 (2016)
Article DOI: 10.1021/acs.jnatprod.6b00090
BindingDB Entry DOI: 10.7270/Q27W6F46 |
More data for this
Ligand-Target Pair | |
High affinity cGMP-specific 3',5'-cyclic phosphodiesterase 9A
(Homo sapiens (Human)) | BDBM50041016
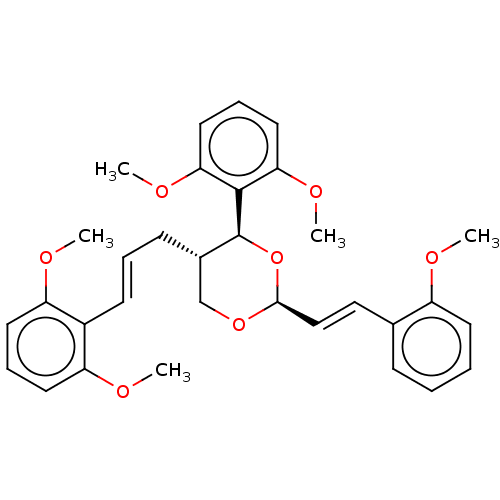
(CHEMBL3355396)Show SMILES COc1ccccc1\C=C\[C@H]1OC[C@H](C\C=C\c2c(OC)cccc2OC)[C@H](O1)c1c(OC)cccc1OC |r| Show InChI InChI=1S/C32H36O7/c1-33-25-14-7-6-11-22(25)19-20-30-38-21-23(12-8-13-24-26(34-2)15-9-16-27(24)35-3)32(39-30)31-28(36-4)17-10-18-29(31)37-5/h6-11,13-20,23,30,32H,12,21H2,1-5H3/b13-8+,20-19+/t23-,30-,32-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-sen University
Curated by ChEMBL
| Assay Description
Inhibition of recombinant PDE9A2 catalytic domain (unknown origin) expressed in Escherichia coli BL21 using [3H]cGMP as substrate after 15 mins by li... |
J Nat Prod 77: 2651-7 (2014)
Article DOI: 10.1021/np500528u
BindingDB Entry DOI: 10.7270/Q2N87CDV |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data