 Found 911 hits with Last Name = 'henderson' and Initial = 'mj'
Found 911 hits with Last Name = 'henderson' and Initial = 'mj' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Sigma intracellular receptor 2
(Homo sapiens (Human)) | BDBM50606398
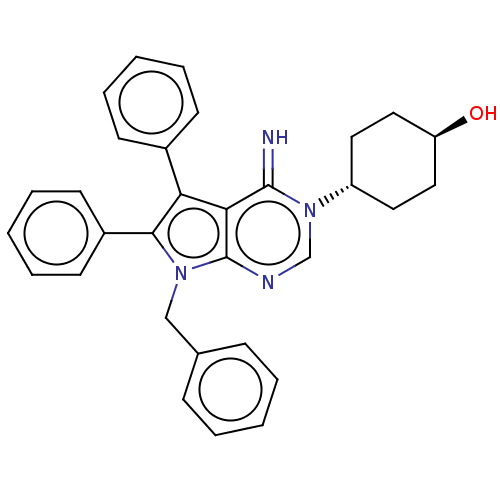
(ML-246 | Metarrestin | Ml-246)Show SMILES O[C@H]1CC[C@@H](CC1)n1cnc2n(Cc3ccccc3)c(c(-c3ccccc3)c2c1=N)-c1ccccc1 |wU:4.7,wD:1.0,(20.99,-11.86,;22.33,-11.09,;23.66,-11.86,;25,-11.09,;25,-9.55,;23.66,-8.78,;22.33,-9.55,;26.33,-8.78,;26.33,-7.24,;27.66,-6.47,;29,-7.24,;30.46,-6.76,;30.94,-5.3,;29.91,-4.15,;30.38,-2.69,;29.35,-1.55,;27.85,-1.87,;27.37,-3.33,;28.4,-4.47,;31.37,-8.01,;30.46,-9.26,;30.94,-10.72,;32.44,-11.04,;32.92,-12.5,;31.89,-13.65,;30.38,-13.33,;29.91,-11.86,;29,-8.78,;27.66,-9.55,;27.66,-11.09,;32.91,-8.01,;33.68,-6.68,;35.22,-6.68,;35.99,-8.01,;35.22,-9.34,;33.68,-9.34,)| | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 244 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00204
BindingDB Entry DOI: 10.7270/Q2FN1B9R |
More data for this
Ligand-Target Pair | |
Lysine-specific demethylase 5A
(Homo sapiens (Human)) | BDBM50468050

(CHEMBL4287599)Show SMILES CN(C)C\C=C\C(=O)Nc1cccc(c1)C(OCCN1CCCCC1)c1cc2nccc(C(O)=O)c2s1 Show InChI InChI=1S/C28H34N4O4S/c1-31(2)13-7-10-25(33)30-21-9-6-8-20(18-21)26(36-17-16-32-14-4-3-5-15-32)24-19-23-27(37-24)22(28(34)35)11-12-29-23/h6-12,18-19,26H,3-5,13-17H2,1-2H3,(H,30,33)(H,34,35)/b10-7+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Texas M.D. Anderson Cancer Center
Curated by ChEMBL
| Assay Description
Irreversible inhibition of KDM5A ARID/PhD1 domain deletion mutant (1 to 739 residues) (unknown origin) |
J Med Chem 61: 10588-10601 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01219
BindingDB Entry DOI: 10.7270/Q2R49TGF |
More data for this
Ligand-Target Pair | |
Alpha-2C adrenergic receptor
(Homo sapiens (Human)) | BDBM50606398

(ML-246 | Metarrestin | Ml-246)Show SMILES O[C@H]1CC[C@@H](CC1)n1cnc2n(Cc3ccccc3)c(c(-c3ccccc3)c2c1=N)-c1ccccc1 |wU:4.7,wD:1.0,(20.99,-11.86,;22.33,-11.09,;23.66,-11.86,;25,-11.09,;25,-9.55,;23.66,-8.78,;22.33,-9.55,;26.33,-8.78,;26.33,-7.24,;27.66,-6.47,;29,-7.24,;30.46,-6.76,;30.94,-5.3,;29.91,-4.15,;30.38,-2.69,;29.35,-1.55,;27.85,-1.87,;27.37,-3.33,;28.4,-4.47,;31.37,-8.01,;30.46,-9.26,;30.94,-10.72,;32.44,-11.04,;32.92,-12.5,;31.89,-13.65,;30.38,-13.33,;29.91,-11.86,;29,-8.78,;27.66,-9.55,;27.66,-11.09,;32.91,-8.01,;33.68,-6.68,;35.22,-6.68,;35.99,-8.01,;35.22,-9.34,;33.68,-9.34,)| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 1.22E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00204
BindingDB Entry DOI: 10.7270/Q2FN1B9R |
More data for this
Ligand-Target Pair | |
Alpha-1D adrenergic receptor
(Homo sapiens (Human)) | BDBM50606398

(ML-246 | Metarrestin | Ml-246)Show SMILES O[C@H]1CC[C@@H](CC1)n1cnc2n(Cc3ccccc3)c(c(-c3ccccc3)c2c1=N)-c1ccccc1 |wU:4.7,wD:1.0,(20.99,-11.86,;22.33,-11.09,;23.66,-11.86,;25,-11.09,;25,-9.55,;23.66,-8.78,;22.33,-9.55,;26.33,-8.78,;26.33,-7.24,;27.66,-6.47,;29,-7.24,;30.46,-6.76,;30.94,-5.3,;29.91,-4.15,;30.38,-2.69,;29.35,-1.55,;27.85,-1.87,;27.37,-3.33,;28.4,-4.47,;31.37,-8.01,;30.46,-9.26,;30.94,-10.72,;32.44,-11.04,;32.92,-12.5,;31.89,-13.65,;30.38,-13.33,;29.91,-11.86,;29,-8.78,;27.66,-9.55,;27.66,-11.09,;32.91,-8.01,;33.68,-6.68,;35.22,-6.68,;35.99,-8.01,;35.22,-9.34,;33.68,-9.34,)| | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 3.26E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00204
BindingDB Entry DOI: 10.7270/Q2FN1B9R |
More data for this
Ligand-Target Pair | |
Sodium-dependent dopamine transporter
(Homo sapiens (Human)) | BDBM50606398

(ML-246 | Metarrestin | Ml-246)Show SMILES O[C@H]1CC[C@@H](CC1)n1cnc2n(Cc3ccccc3)c(c(-c3ccccc3)c2c1=N)-c1ccccc1 |wU:4.7,wD:1.0,(20.99,-11.86,;22.33,-11.09,;23.66,-11.86,;25,-11.09,;25,-9.55,;23.66,-8.78,;22.33,-9.55,;26.33,-8.78,;26.33,-7.24,;27.66,-6.47,;29,-7.24,;30.46,-6.76,;30.94,-5.3,;29.91,-4.15,;30.38,-2.69,;29.35,-1.55,;27.85,-1.87,;27.37,-3.33,;28.4,-4.47,;31.37,-8.01,;30.46,-9.26,;30.94,-10.72,;32.44,-11.04,;32.92,-12.5,;31.89,-13.65,;30.38,-13.33,;29.91,-11.86,;29,-8.78,;27.66,-9.55,;27.66,-11.09,;32.91,-8.01,;33.68,-6.68,;35.22,-6.68,;35.99,-8.01,;35.22,-9.34,;33.68,-9.34,)| | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 3.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00204
BindingDB Entry DOI: 10.7270/Q2FN1B9R |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Homo sapiens (Human)) | BDBM50606398

(ML-246 | Metarrestin | Ml-246)Show SMILES O[C@H]1CC[C@@H](CC1)n1cnc2n(Cc3ccccc3)c(c(-c3ccccc3)c2c1=N)-c1ccccc1 |wU:4.7,wD:1.0,(20.99,-11.86,;22.33,-11.09,;23.66,-11.86,;25,-11.09,;25,-9.55,;23.66,-8.78,;22.33,-9.55,;26.33,-8.78,;26.33,-7.24,;27.66,-6.47,;29,-7.24,;30.46,-6.76,;30.94,-5.3,;29.91,-4.15,;30.38,-2.69,;29.35,-1.55,;27.85,-1.87,;27.37,-3.33,;28.4,-4.47,;31.37,-8.01,;30.46,-9.26,;30.94,-10.72,;32.44,-11.04,;32.92,-12.5,;31.89,-13.65,;30.38,-13.33,;29.91,-11.86,;29,-8.78,;27.66,-9.55,;27.66,-11.09,;32.91,-8.01,;33.68,-6.68,;35.22,-6.68,;35.99,-8.01,;35.22,-9.34,;33.68,-9.34,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 3.56E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00204
BindingDB Entry DOI: 10.7270/Q2FN1B9R |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50527623

(CHEMBL4443378)Show SMILES CC[C@H](Nc1nc(N)nc(C)c1C#N)c1nc2cccc(CCCCCC(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C29H32N8O3/c1-3-22(33-26-21(17-30)18(2)32-29(31)35-26)27-34-23-15-10-12-19(11-6-4-9-16-24(38)36-40)25(23)28(39)37(27)20-13-7-5-8-14-20/h5,7-8,10,12-15,22,40H,3-4,6,9,11,16H2,1-2H3,(H,36,38)(H3,31,32,33,35)/t22-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50527619
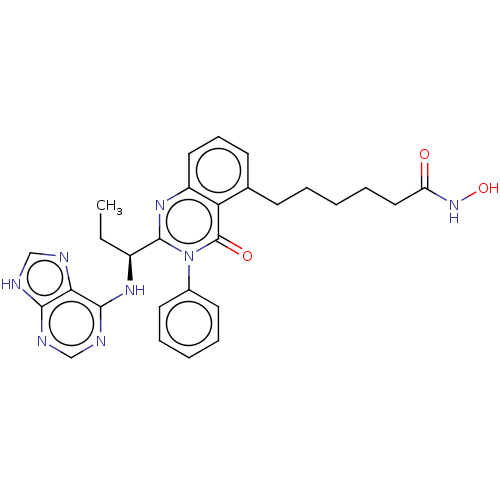
(CHEMBL4451623)Show SMILES CC[C@H](Nc1ncnc2[nH]cnc12)c1nc2cccc(CCCCCC(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C28H30N8O3/c1-2-20(33-26-24-25(30-16-29-24)31-17-32-26)27-34-21-14-9-11-18(10-5-3-8-15-22(37)35-39)23(21)28(38)36(27)19-12-6-4-7-13-19/h4,6-7,9,11-14,16-17,20,39H,2-3,5,8,10,15H2,1H3,(H,35,37)(H2,29,30,31,32,33)/t20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50527624
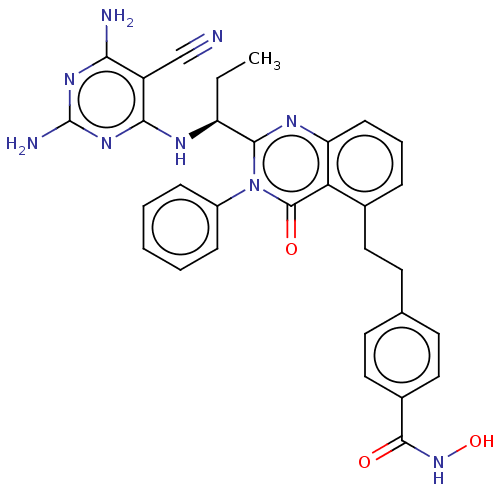
(CHEMBL4450613)Show SMILES CC[C@H](Nc1nc(N)nc(N)c1C#N)c1nc2cccc(CCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C31H29N9O3/c1-2-23(35-27-22(17-32)26(33)37-31(34)38-27)28-36-24-10-6-7-19(14-11-18-12-15-20(16-13-18)29(41)39-43)25(24)30(42)40(28)21-8-4-3-5-9-21/h3-10,12-13,15-16,23,43H,2,11,14H2,1H3,(H,39,41)(H5,33,34,35,37,38)/t23-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50527616

(CHEMBL4464003)Show SMILES CC[C@H](Nc1ncnc2[nH]cnc12)c1nc2cccc(CNC(=O)c3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C31H27N9O4/c1-2-22(37-27-25-26(34-16-33-25)35-17-36-27)28-38-23-10-6-7-20(24(23)31(43)40(28)21-8-4-3-5-9-21)15-32-29(41)18-11-13-19(14-12-18)30(42)39-44/h3-14,16-17,22,44H,2,15H2,1H3,(H,32,41)(H,39,42)(H2,33,34,35,36,37)/t22-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50527622

(CHEMBL4550648)Show SMILES CC[C@H](Nc1nc(N)nc(N)c1C#N)c1nc2cccc(CCCCCC(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C28H31N9O3/c1-2-20(32-25-19(16-29)24(30)34-28(31)35-25)26-33-21-14-9-11-17(10-5-3-8-15-22(38)36-40)23(21)27(39)37(26)18-12-6-4-7-13-18/h4,6-7,9,11-14,20,40H,2-3,5,8,10,15H2,1H3,(H,36,38)(H5,30,31,32,34,35)/t20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50527617

(CHEMBL4522666)Show SMILES C[C@H](Nc1nc(N)nc(N)c1C#N)c1nc2cccc(CNC(=O)c3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C30H26N10O4/c1-16(35-25-21(14-31)24(32)37-30(33)38-25)26-36-22-9-5-6-19(23(22)29(43)40(26)20-7-3-2-4-8-20)15-34-27(41)17-10-12-18(13-11-17)28(42)39-44/h2-13,16,44H,15H2,1H3,(H,34,41)(H,39,42)(H5,32,33,35,37,38)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50527620

(CHEMBL4563838)Show SMILES CC[C@H](Nc1ncnc2[nH]cnc12)c1nc2cccc(-c3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C29H24N8O3/c1-2-21(34-26-24-25(31-15-30-24)32-16-33-26)27-35-22-10-6-9-20(17-11-13-18(14-12-17)28(38)36-40)23(22)29(39)37(27)19-7-4-3-5-8-19/h3-16,21,40H,2H2,1H3,(H,36,38)(H2,30,31,32,33,34)/t21-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
L-lactate dehydrogenase A chain
(Homo sapiens (Human)) | BDBM50546980

(CHEMBL4792513)Show SMILES NS(=O)(=O)c1ccc(Cc2c(CC3CC3)n(nc2-c2ccc(F)c(c2)C#CC(F)C2CCOC2)-c2nc(cs2)C(O)=O)cc1F | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human LDHA using sodium pyruvate as substrate preincubated for 5 mins followed by diaphorase/resazurin addition and measure... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00916
BindingDB Entry DOI: 10.7270/Q2057KJ6 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50527623

(CHEMBL4443378)Show SMILES CC[C@H](Nc1nc(N)nc(C)c1C#N)c1nc2cccc(CCCCCC(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C29H32N8O3/c1-3-22(33-26-21(17-30)18(2)32-29(31)35-26)27-34-23-15-10-12-19(11-6-4-9-16-24(38)36-40)25(23)28(39)37(27)20-13-7-5-8-14-20/h5,7-8,10,12-15,22,40H,3-4,6,9,11,16H2,1-2H3,(H,36,38)(H3,31,32,33,35)/t22-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50527605

(CHEMBL4526673)Show SMILES C[C@H](Nc1nc(N)nc(C)c1C#N)c1nc2cccc(NCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C30H27N9O3/c1-17-22(15-31)26(37-30(32)35-17)34-18(2)27-36-24-10-6-9-23(25(24)29(41)39(27)21-7-4-3-5-8-21)33-16-19-11-13-20(14-12-19)28(40)38-42/h3-14,18,33,42H,16H2,1-2H3,(H,38,40)(H3,32,34,35,37)/t18-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50527624

(CHEMBL4450613)Show SMILES CC[C@H](Nc1nc(N)nc(N)c1C#N)c1nc2cccc(CCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C31H29N9O3/c1-2-23(35-27-22(17-32)26(33)37-31(34)38-27)28-36-24-10-6-7-19(14-11-18-12-15-20(16-13-18)29(41)39-43)25(24)30(42)40(28)21-8-4-3-5-9-21/h3-10,12-13,15-16,23,43H,2,11,14H2,1H3,(H,39,41)(H5,33,34,35,37,38)/t23-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Histone deacetylase 6
(Homo sapiens (Human)) | BDBM50005711
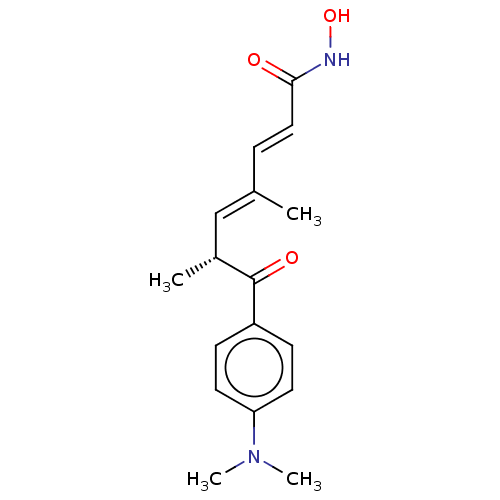
(CHEBI:46024 | GNF-Pf-1011 | TRICHOSTATIN | Trichos...)Show SMILES C[C@H](\C=C(/C)\C=C\C(=O)NO)C(=O)c1ccc(cc1)N(C)C |r| Show InChI InChI=1S/C17H22N2O3/c1-12(5-10-16(20)18-22)11-13(2)17(21)14-6-8-15(9-7-14)19(3)4/h5-11,13,22H,1-4H3,(H,18,20)/b10-5+,12-11+/t13-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal GST-tagged HDAC6 (1 to 1125 residues) expressed in baculovirus infected insect cells using fluorogenic pep... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50527622

(CHEMBL4550648)Show SMILES CC[C@H](Nc1nc(N)nc(N)c1C#N)c1nc2cccc(CCCCCC(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C28H31N9O3/c1-2-20(32-25-19(16-29)24(30)34-28(31)35-25)26-33-21-14-9-11-17(10-5-3-8-15-22(38)36-40)23(21)27(39)37(26)18-12-6-4-7-13-18/h4,6-7,9,11-14,20,40H,2-3,5,8,10,15H2,1H3,(H,36,38)(H5,30,31,32,34,35)/t20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50527610
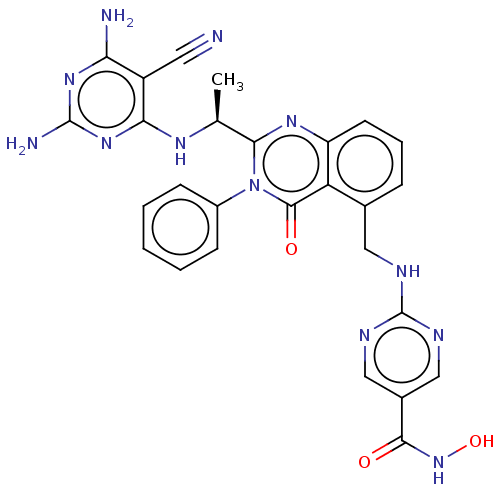
(CHEMBL4583074)Show SMILES C[C@H](Nc1nc(N)nc(N)c1C#N)c1nc2cccc(CNc3ncc(cn3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C27H24N12O3/c1-14(34-22-18(10-28)21(29)36-26(30)37-22)23-35-19-9-5-6-15(11-31-27-32-12-16(13-33-27)24(40)38-42)20(19)25(41)39(23)17-7-3-2-4-8-17/h2-9,12-14,42H,11H2,1H3,(H,38,40)(H,31,32,33)(H5,29,30,34,36,37)/t14-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
L-lactate dehydrogenase A chain
(Homo sapiens (Human)) | BDBM50546969
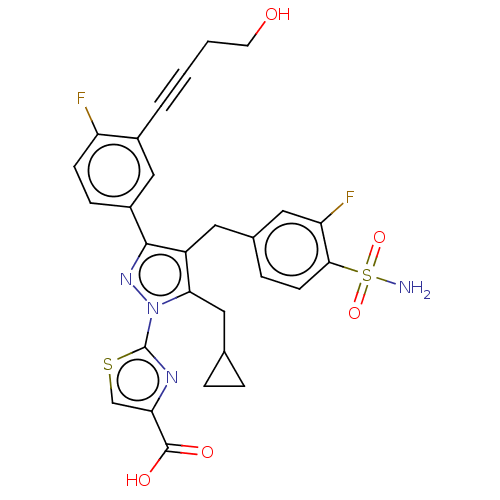
(CHEMBL4786682 | US11247971, Cmpd ID 409)Show SMILES NS(=O)(=O)c1ccc(Cc2c(CC3CC3)n(nc2-c2ccc(F)c(c2)C#CCCO)-c2nc(cs2)C(O)=O)cc1F | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human LDHA using sodium pyruvate as substrate preincubated for 5 mins followed by diaphorase/resazurin addition and measure... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00916
BindingDB Entry DOI: 10.7270/Q2057KJ6 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM25045
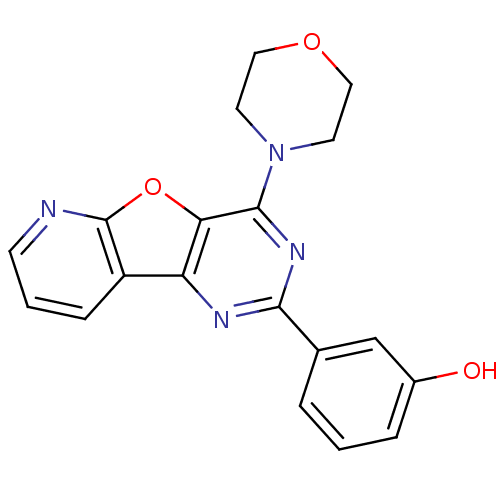
(3-(4-morpholin-4-ylpyrido[2,3]furo[2,4-b]pyrimidin...)Show InChI InChI=1S/C19H16N4O3/c24-13-4-1-3-12(11-13)17-21-15-14-5-2-6-20-19(14)26-16(15)18(22-17)23-7-9-25-10-8-23/h1-6,11,24H,7-10H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Histone deacetylase 6
(Homo sapiens (Human)) | BDBM50527634

(CHEMBL4456827)Show SMILES CC[C@H](Nc1ncnc(N)c1Cl)c1nc2cccc(NCc3ccc(cn3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C28H26ClN9O3/c1-2-19(35-25-23(29)24(30)33-15-34-25)26-36-21-10-6-9-20(22(21)28(40)38(26)18-7-4-3-5-8-18)32-14-17-12-11-16(13-31-17)27(39)37-41/h3-13,15,19,32,41H,2,14H2,1H3,(H,37,39)(H3,30,33,34,35)/t19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal GST-tagged HDAC6 (1 to 1125 residues) expressed in baculovirus infected insect cells using fluorogenic pep... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Histone deacetylase 6
(Homo sapiens (Human)) | BDBM50527610

(CHEMBL4583074)Show SMILES C[C@H](Nc1nc(N)nc(N)c1C#N)c1nc2cccc(CNc3ncc(cn3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C27H24N12O3/c1-14(34-22-18(10-28)21(29)36-26(30)37-22)23-35-19-9-5-6-15(11-31-27-32-12-16(13-33-27)24(40)38-42)20(19)25(41)39(23)17-7-3-2-4-8-17/h2-9,12-14,42H,11H2,1H3,(H,38,40)(H,31,32,33)(H5,29,30,34,36,37)/t14-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal GST-tagged HDAC6 (1 to 1125 residues) expressed in baculovirus infected insect cells using fluorogenic pep... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
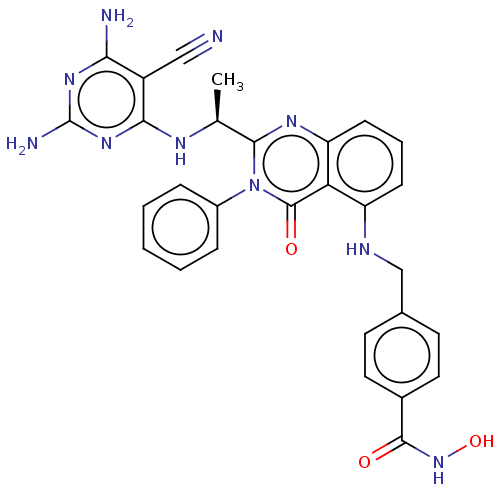
(Homo sapiens (Human)) | BDBM50527613
(CHEMBL4460447)Show SMILES C[C@H](Nc1nc(N)nc(N)c1C#N)c1nc2cccc(NCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C29H26N10O3/c1-16(34-25-20(14-30)24(31)36-29(32)37-25)26-35-22-9-5-8-21(23(22)28(41)39(26)19-6-3-2-4-7-19)33-15-17-10-12-18(13-11-17)27(40)38-42/h2-13,16,33,42H,15H2,1H3,(H,38,40)(H5,31,32,34,36,37)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50527605

(CHEMBL4526673)Show SMILES C[C@H](Nc1nc(N)nc(C)c1C#N)c1nc2cccc(NCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C30H27N9O3/c1-17-22(15-31)26(37-30(32)35-17)34-18(2)27-36-24-10-6-9-23(25(24)29(41)39(27)21-7-4-3-5-8-21)33-16-19-11-13-20(14-12-19)28(40)38-42/h3-14,18,33,42H,16H2,1-2H3,(H,38,40)(H3,32,34,35,37)/t18-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Histone deacetylase 6
(Homo sapiens (Human)) | BDBM50527629

(CHEMBL4459679)Show SMILES CC[C@H](Nc1cc(C)nc(N)n1)c1nc2cccc(NCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C30H30N8O3/c1-3-22(34-25-16-18(2)33-30(31)36-25)27-35-24-11-7-10-23(26(24)29(40)38(27)21-8-5-4-6-9-21)32-17-19-12-14-20(15-13-19)28(39)37-41/h4-16,22,32,41H,3,17H2,1-2H3,(H,37,39)(H3,31,33,34,36)/t22-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal GST-tagged HDAC6 (1 to 1125 residues) expressed in baculovirus infected insect cells using fluorogenic pep... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Histone deacetylase 6
(Homo sapiens (Human)) | BDBM50527604

(CHEMBL4578757)Show SMILES CC[C@H](Nc1ncnc(N)c1C#N)c1nc2cccc(NCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C30H27N9O3/c1-2-22(36-27-21(15-31)26(32)34-17-35-27)28-37-24-10-6-9-23(25(24)30(41)39(28)20-7-4-3-5-8-20)33-16-18-11-13-19(14-12-18)29(40)38-42/h3-14,17,22,33,42H,2,16H2,1H3,(H,38,40)(H3,32,34,35,36)/t22-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal GST-tagged HDAC6 (1 to 1125 residues) expressed in baculovirus infected insect cells using fluorogenic pep... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50527613

(CHEMBL4460447)Show SMILES C[C@H](Nc1nc(N)nc(N)c1C#N)c1nc2cccc(NCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C29H26N10O3/c1-16(34-25-20(14-30)24(31)36-29(32)37-25)26-35-22-9-5-8-21(23(22)28(41)39(26)19-6-3-2-4-7-19)33-15-17-10-12-18(13-11-17)27(40)38-42/h2-13,16,33,42H,15H2,1H3,(H,38,40)(H5,31,32,34,36,37)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Histone deacetylase 6
(Homo sapiens (Human)) | BDBM50527633
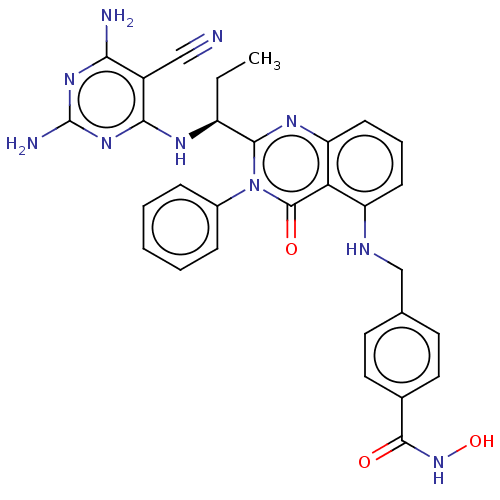
(CHEMBL4464812)Show SMILES CC[C@H](Nc1nc(N)nc(N)c1C#N)c1nc2cccc(NCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C30H28N10O3/c1-2-21(35-26-20(15-31)25(32)37-30(33)38-26)27-36-23-10-6-9-22(24(23)29(42)40(27)19-7-4-3-5-8-19)34-16-17-11-13-18(14-12-17)28(41)39-43/h3-14,21,34,43H,2,16H2,1H3,(H,39,41)(H5,32,33,35,37,38)/t21-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal GST-tagged HDAC6 (1 to 1125 residues) expressed in baculovirus infected insect cells using fluorogenic pep... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM25045

(3-(4-morpholin-4-ylpyrido[2,3]furo[2,4-b]pyrimidin...)Show InChI InChI=1S/C19H16N4O3/c24-13-4-1-3-12(11-13)17-21-15-14-5-2-6-20-19(14)26-16(15)18(22-17)23-7-9-25-10-8-23/h1-6,11,24H,7-10H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone deacetylase 6
(Homo sapiens (Human)) | BDBM50527625
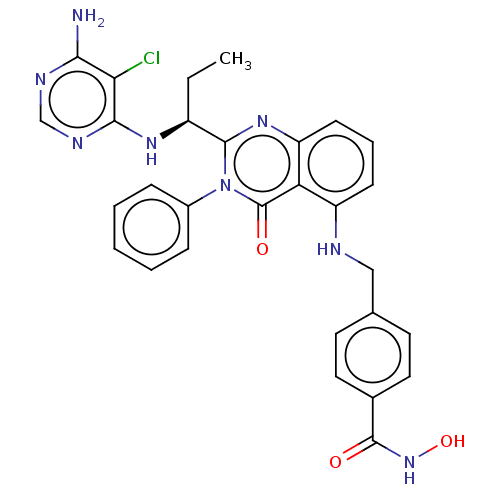
(CHEMBL4467927)Show SMILES CC[C@H](Nc1ncnc(N)c1Cl)c1nc2cccc(NCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C29H27ClN8O3/c1-2-20(35-26-24(30)25(31)33-16-34-26)27-36-22-10-6-9-21(23(22)29(40)38(27)19-7-4-3-5-8-19)32-15-17-11-13-18(14-12-17)28(39)37-41/h3-14,16,20,32,41H,2,15H2,1H3,(H,37,39)(H3,31,33,34,35)/t20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal GST-tagged HDAC6 (1 to 1125 residues) expressed in baculovirus infected insect cells using fluorogenic pep... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Histone deacetylase 6
(Homo sapiens (Human)) | BDBM50527619

(CHEMBL4451623)Show SMILES CC[C@H](Nc1ncnc2[nH]cnc12)c1nc2cccc(CCCCCC(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C28H30N8O3/c1-2-20(33-26-24-25(30-16-29-24)31-17-32-26)27-34-21-14-9-11-18(10-5-3-8-15-22(37)35-39)23(21)28(38)36(27)19-12-6-4-7-13-19/h4,6-7,9,11-14,16-17,20,39H,2-3,5,8,10,15H2,1H3,(H,35,37)(H2,29,30,31,32,33)/t20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal GST-tagged HDAC6 (1 to 1125 residues) expressed in baculovirus infected insect cells using fluorogenic pep... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50527608

(CHEMBL4445342)Show SMILES CC[C@H](Nc1ncnc2[nH]cnc12)c1nc2cccc(CCCCC(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C27H28N8O3/c1-2-19(32-25-23-24(29-15-28-23)30-16-31-25)26-33-20-13-8-10-17(9-6-7-14-21(36)34-38)22(20)27(37)35(26)18-11-4-3-5-12-18/h3-5,8,10-13,15-16,19,38H,2,6-7,9,14H2,1H3,(H,34,36)(H2,28,29,30,31,32)/t19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | <5 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
L-lactate dehydrogenase A chain
(Homo sapiens (Human)) | BDBM50546981

(CHEMBL4797357)Show SMILES NS(=O)(=O)c1ccc(Cc2c(CC3CC3)n(nc2-c2ccc(F)c(c2)C#CC(O)C2CCOC2)-c2nc(cs2)C(O)=O)cc1F | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human LDHA using sodium pyruvate as substrate preincubated for 5 mins followed by diaphorase/resazurin addition and measure... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00916
BindingDB Entry DOI: 10.7270/Q2057KJ6 |
More data for this
Ligand-Target Pair | |
Histone deacetylase 6
(Homo sapiens (Human)) | BDBM50527627

(CHEMBL4516095)Show SMILES CC[C@H](Nc1ncnc2[nH]cnc12)c1nc2cccc(NCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C30H27N9O3/c1-2-21(36-27-25-26(33-16-32-25)34-17-35-27)28-37-23-10-6-9-22(24(23)30(41)39(28)20-7-4-3-5-8-20)31-15-18-11-13-19(14-12-18)29(40)38-42/h3-14,16-17,21,31,42H,2,15H2,1H3,(H,38,40)(H2,32,33,34,35,36)/t21-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal GST-tagged HDAC6 (1 to 1125 residues) expressed in baculovirus infected insect cells using fluorogenic pep... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50527628

(CHEMBL4555432)Show SMILES CC[C@H](Nc1ncnc2[nH]cnc12)c1nc2cccc(CCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C31H28N8O3/c1-2-23(36-28-26-27(33-17-32-26)34-18-35-28)29-37-24-10-6-7-20(14-11-19-12-15-21(16-13-19)30(40)38-42)25(24)31(41)39(29)22-8-4-3-5-9-22/h3-10,12-13,15-18,23,42H,2,11,14H2,1H3,(H,38,40)(H2,32,33,34,35,36)/t23-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50527611

(CHEMBL4544187)Show SMILES CC[C@H](Nc1nc(N)nc(C)c1C#N)c1nc2cccc(NCCCCC(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C28H31N9O3/c1-3-20(33-25-19(16-29)17(2)32-28(30)35-25)26-34-22-13-9-12-21(31-15-8-7-14-23(38)36-40)24(22)27(39)37(26)18-10-5-4-6-11-18/h4-6,9-13,20,31,40H,3,7-8,14-15H2,1-2H3,(H,36,38)(H3,30,32,33,35)/t20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Histone deacetylase 6
(Homo sapiens (Human)) | BDBM50527602

(CHEMBL4581057)Show SMILES CC[C@H](Nc1ncnc2[nH]cnc12)c1nc2cccc(CNc3ncc(cn3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C28H25N11O3/c1-2-19(36-24-22-23(33-14-32-22)34-15-35-24)25-37-20-10-6-7-16(11-29-28-30-12-17(13-31-28)26(40)38-42)21(20)27(41)39(25)18-8-4-3-5-9-18/h3-10,12-15,19,42H,2,11H2,1H3,(H,38,40)(H,29,30,31)(H2,32,33,34,35,36)/t19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal GST-tagged HDAC6 (1 to 1125 residues) expressed in baculovirus infected insect cells using fluorogenic pep... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
L-lactate dehydrogenase A chain
(Homo sapiens (Human)) | BDBM50546978
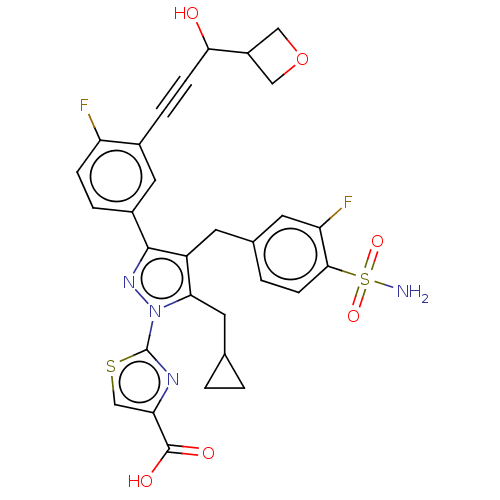
(CHEMBL4752940)Show SMILES NS(=O)(=O)c1ccc(Cc2c(CC3CC3)n(nc2-c2ccc(F)c(c2)C#CC(O)C2COC2)-c2nc(cs2)C(O)=O)cc1F | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human LDHA using sodium pyruvate as substrate preincubated for 5 mins followed by diaphorase/resazurin addition and measure... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00916
BindingDB Entry DOI: 10.7270/Q2057KJ6 |
More data for this
Ligand-Target Pair | |
Histone deacetylase 6
(Homo sapiens (Human)) | BDBM50527613

(CHEMBL4460447)Show SMILES C[C@H](Nc1nc(N)nc(N)c1C#N)c1nc2cccc(NCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C29H26N10O3/c1-16(34-25-20(14-30)24(31)36-29(32)37-25)26-35-22-9-5-8-21(23(22)28(41)39(26)19-6-3-2-4-7-19)33-15-17-10-12-18(13-11-17)27(40)38-42/h2-13,16,33,42H,15H2,1H3,(H,38,40)(H5,31,32,34,36,37)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal GST-tagged HDAC6 (1 to 1125 residues) expressed in baculovirus infected insect cells using fluorogenic pep... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM25045

(3-(4-morpholin-4-ylpyrido[2,3]furo[2,4-b]pyrimidin...)Show InChI InChI=1S/C19H16N4O3/c24-13-4-1-3-12(11-13)17-21-15-14-5-2-6-20-19(14)26-16(15)18(22-17)23-7-9-25-10-8-23/h1-6,11,24H,7-10H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kbeta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex u... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50527615
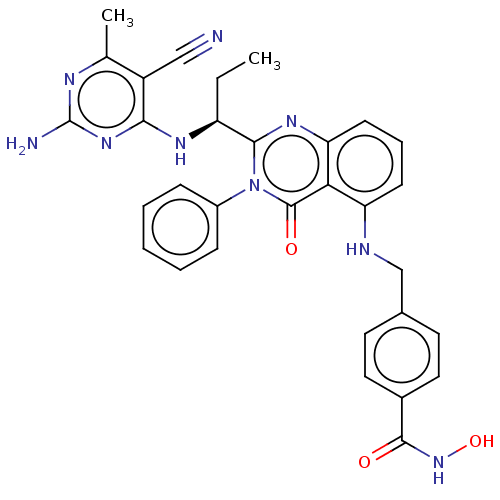
(CHEMBL4455923)Show SMILES CC[C@H](Nc1nc(N)nc(C)c1C#N)c1nc2cccc(NCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C31H29N9O3/c1-3-23(36-27-22(16-32)18(2)35-31(33)38-27)28-37-25-11-7-10-24(26(25)30(42)40(28)21-8-5-4-6-9-21)34-17-19-12-14-20(15-13-19)29(41)39-43/h4-15,23,34,43H,3,17H2,1-2H3,(H,39,41)(H3,33,35,36,38)/t23-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
L-lactate dehydrogenase A chain
(Homo sapiens (Human)) | BDBM50546979
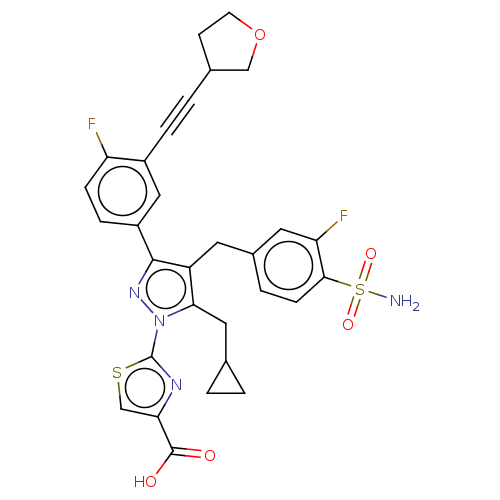
(CHEMBL4747300 | US11247971, Cmpd ID 423)Show SMILES NS(=O)(=O)c1ccc(Cc2c(CC3CC3)n(nc2-c2ccc(F)c(c2)C#CC2CCOC2)-c2nc(cs2)C(O)=O)cc1F | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human LDHA using sodium pyruvate as substrate preincubated for 5 mins followed by diaphorase/resazurin addition and measure... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00916
BindingDB Entry DOI: 10.7270/Q2057KJ6 |
More data for this
Ligand-Target Pair | |
L-lactate dehydrogenase A chain
(Homo sapiens (Human)) | BDBM50546977
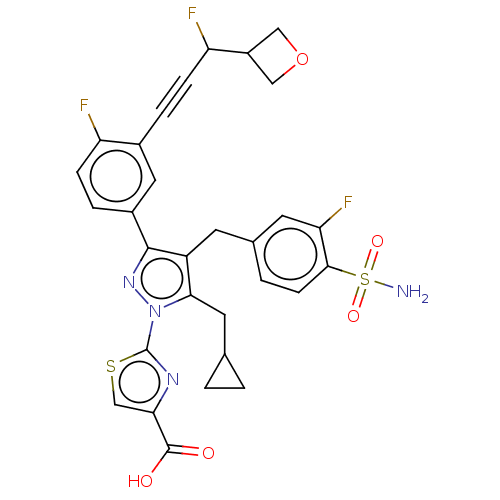
(CHEMBL4759378)Show SMILES NS(=O)(=O)c1ccc(Cc2c(CC3CC3)n(nc2-c2ccc(F)c(c2)C#CC(F)C2COC2)-c2nc(cs2)C(O)=O)cc1F | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human LDHA using sodium pyruvate as substrate preincubated for 5 mins followed by diaphorase/resazurin addition and measure... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00916
BindingDB Entry DOI: 10.7270/Q2057KJ6 |
More data for this
Ligand-Target Pair | |
L-lactate dehydrogenase A chain
(Homo sapiens (Human)) | BDBM50546975
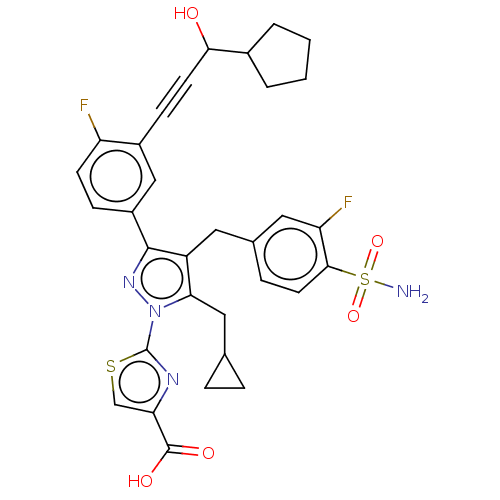
(CHEMBL4749903)Show SMILES NS(=O)(=O)c1ccc(Cc2c(CC3CC3)n(nc2-c2ccc(F)c(c2)C#CC(O)C2CCCC2)-c2nc(cs2)C(O)=O)cc1F | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human LDHA using sodium pyruvate as substrate preincubated for 5 mins followed by diaphorase/resazurin addition and measure... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00916
BindingDB Entry DOI: 10.7270/Q2057KJ6 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50527611

(CHEMBL4544187)Show SMILES CC[C@H](Nc1nc(N)nc(C)c1C#N)c1nc2cccc(NCCCCC(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C28H31N9O3/c1-3-20(33-25-19(16-29)17(2)32-28(30)35-25)26-34-22-13-9-12-21(31-15-8-7-14-23(38)36-40)24(22)27(39)37(26)18-10-5-4-6-11-18/h4-6,9-13,20,31,40H,3,7-8,14-15H2,1-2H3,(H,36,38)(H3,30,32,33,35)/t20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma assessed as reduction in PIP3 product complex formation by measuring displacement of biotin-labelled PIP3 from complex ... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |
L-lactate dehydrogenase A chain
(Homo sapiens (Human)) | BDBM50546970
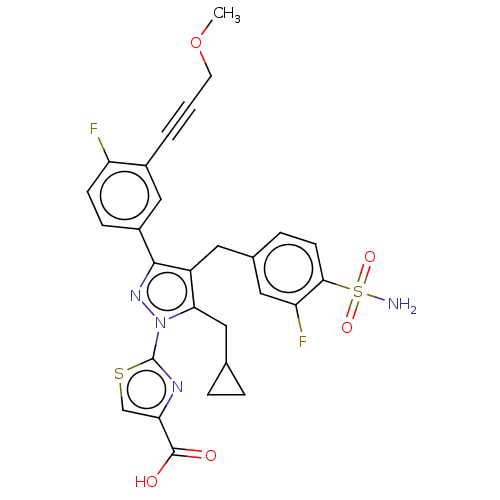
(CHEMBL4783945 | US11247971, Cmpd ID 404)Show SMILES COCC#Cc1cc(ccc1F)-c1nn(c(CC2CC2)c1Cc1ccc(c(F)c1)S(N)(=O)=O)-c1nc(cs1)C(O)=O | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human LDHA using sodium pyruvate as substrate preincubated for 5 mins followed by diaphorase/resazurin addition and measure... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00916
BindingDB Entry DOI: 10.7270/Q2057KJ6 |
More data for this
Ligand-Target Pair | |
L-lactate dehydrogenase A chain
(Homo sapiens (Human)) | BDBM50546998

(CHEMBL4790159 | US11247971, Cmpd ID 405)Show SMILES Cn1cncc1C#Cc1cc(ccc1F)-c1nn(c(CC2CC2)c1Cc1ccc(c(F)c1)S(N)(=O)=O)-c1nc(cs1)C(O)=O | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human LDHA using sodium pyruvate as substrate preincubated for 5 mins followed by diaphorase/resazurin addition and measure... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00916
BindingDB Entry DOI: 10.7270/Q2057KJ6 |
More data for this
Ligand-Target Pair | |
Histone deacetylase 6
(Homo sapiens (Human)) | BDBM50527605

(CHEMBL4526673)Show SMILES C[C@H](Nc1nc(N)nc(C)c1C#N)c1nc2cccc(NCc3ccc(cc3)C(=O)NO)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C30H27N9O3/c1-17-22(15-31)26(37-30(32)35-17)34-18(2)27-36-24-10-6-9-23(25(24)29(41)39(27)21-7-4-3-5-8-21)33-16-19-11-13-20(14-12-19)28(40)38-42/h3-14,18,33,42H,16H2,1-2H3,(H,38,40)(H3,32,34,35,37)/t18-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
National Center for Advancing Translational Sciences
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal GST-tagged HDAC6 (1 to 1125 residues) expressed in baculovirus infected insect cells using fluorogenic pep... |
J Med Chem 63: 4256-4292 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00193
BindingDB Entry DOI: 10.7270/Q2KS6W0V |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data