

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50370424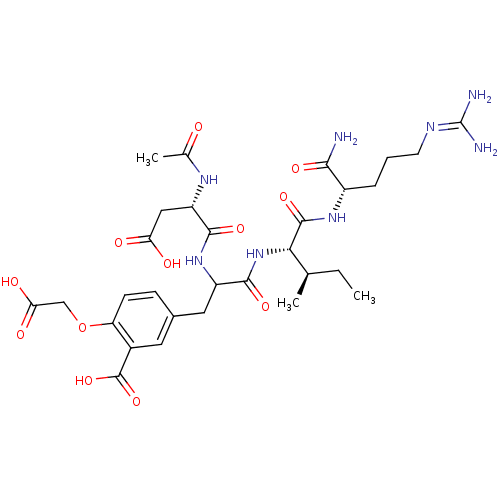 (CHEMBL1791292) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description In vitro inhibitory concentration against Protein-tyrosine phosphatase 1B hydrolysis of p-nitrophenol phosphate | Bioorg Med Chem Lett 14: 5081-3 (2004) Article DOI: 10.1016/j.bmcl.2004.07.078 BindingDB Entry DOI: 10.7270/Q2PV6M4Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50370425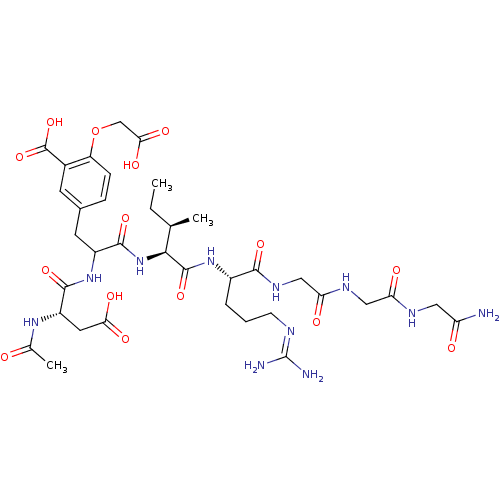 (CHEMBL1791291) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description In vitro inhibitory concentration against Protein-tyrosine phosphatase 1B hydrolysis of p-nitrophenol phosphate | Bioorg Med Chem Lett 14: 5081-3 (2004) Article DOI: 10.1016/j.bmcl.2004.07.078 BindingDB Entry DOI: 10.7270/Q2PV6M4Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50153248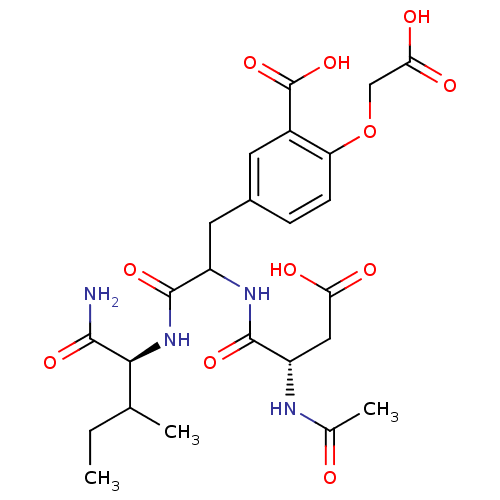 (CHEMBL183738 | N-acetyl-Asp-{5-(2-Amino-2-carboxy-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description In vitro inhibitory concentration against Protein-tyrosine phosphatase 1B hydrolysis of p-nitrophenol phosphate | Bioorg Med Chem Lett 14: 5081-3 (2004) Article DOI: 10.1016/j.bmcl.2004.07.078 BindingDB Entry DOI: 10.7270/Q2PV6M4Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pantetheinase (Homo sapiens (Human)) | BDBM50585537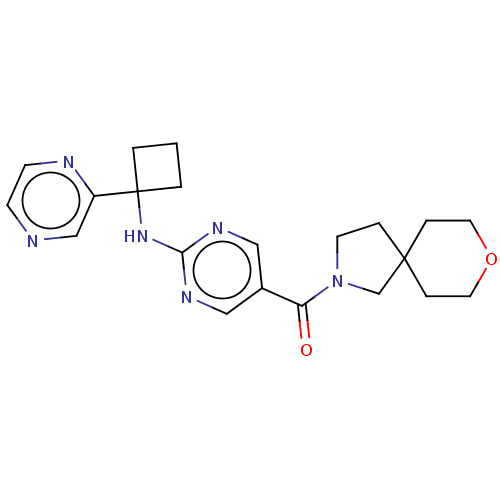 (CHEMBL5084938) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant N-terminal FLAG/His6-tagged human vanin-1 expressed in CHO cells using Pantetheine-7-amino-4-trifluoromethykournarin as sub... | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c01849 BindingDB Entry DOI: 10.7270/Q2XD15K6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pantetheinase (Homo sapiens (Human)) | BDBM480924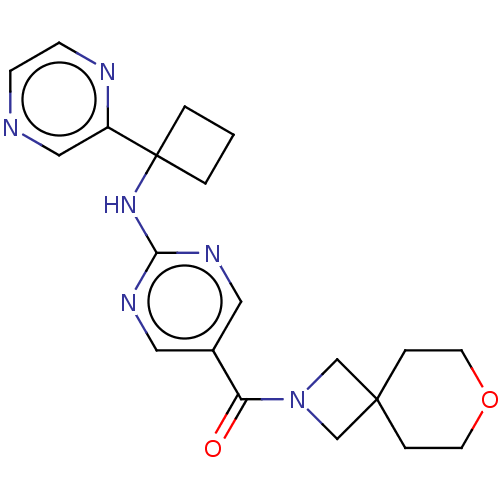 (US10906888, Example 138) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant N-terminal FLAG/His6-tagged human vanin-1 expressed in CHO cells using Pantetheine-7-amino-4-trifluoromethykournarin as sub... | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c01849 BindingDB Entry DOI: 10.7270/Q2XD15K6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pantetheinase (Homo sapiens (Human)) | BDBM480916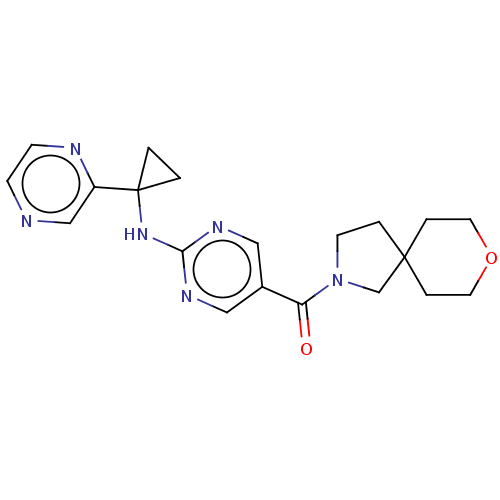 (8-oxa-2-azaspiro[4.5]dec-2-yl(2-{[1-(pyrazin-2-yl)...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | <0.0410 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description The test inhibitors were solubilized in DMSO to a stock concentration of 30 mM. On the day of the assay, dose response plates were prepared by diluti... | US Patent US10906888 (2021) BindingDB Entry DOI: 10.7270/Q2QN69WB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pantetheinase (Homo sapiens (Human)) | BDBM480924 (US10906888, Example 138) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.0430 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description The test inhibitors were solubilized in DMSO to a stock concentration of 30 mM. On the day of the assay, dose response plates were prepared by diluti... | US Patent US10906888 (2021) BindingDB Entry DOI: 10.7270/Q2QN69WB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pantetheinase (Homo sapiens (Human)) | BDBM480965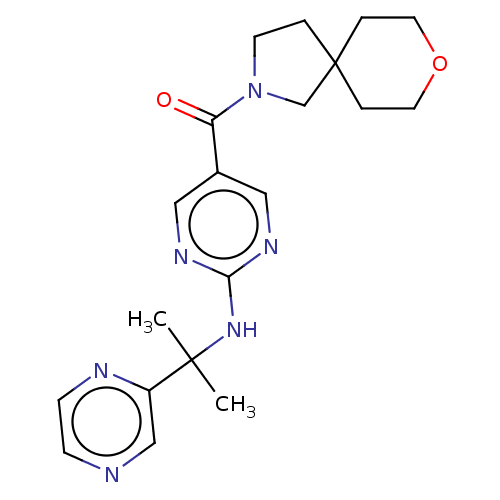 (8-oxa-2-azaspiro[4.5]dec-2-yl(2-{[2-(pyrazin-2-yl)...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant N-terminal FLAG/His6-tagged human vanin-1 expressed in CHO cells using Pantetheine-7-amino-4-trifluoromethykournarin as sub... | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c01849 BindingDB Entry DOI: 10.7270/Q2XD15K6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pantetheinase (Homo sapiens (Human)) | BDBM480965 (8-oxa-2-azaspiro[4.5]dec-2-yl(2-{[2-(pyrazin-2-yl)...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.0620 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description The test inhibitors were solubilized in DMSO to a stock concentration of 30 mM. On the day of the assay, dose response plates were prepared by diluti... | US Patent US10906888 (2021) BindingDB Entry DOI: 10.7270/Q2QN69WB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pantetheinase (Homo sapiens (Human)) | BDBM480929 (8-oxa-2-azaspiro[4.5]dec-2-yl(2-{[2-(pyrimidin-5-y...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant N-terminal FLAG/His6-tagged human vanin-1 expressed in CHO cells using Pantetheine-7-amino-4-trifluoromethykournarin as sub... | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c01849 BindingDB Entry DOI: 10.7270/Q2XD15K6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pantetheinase (Homo sapiens (Human)) | BDBM480929 (8-oxa-2-azaspiro[4.5]dec-2-yl(2-{[2-(pyrimidin-5-y...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.0820 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description The test inhibitors were solubilized in DMSO to a stock concentration of 30 mM. On the day of the assay, dose response plates were prepared by diluti... | US Patent US10906888 (2021) BindingDB Entry DOI: 10.7270/Q2QN69WB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pantetheinase (Homo sapiens (Human)) | BDBM480917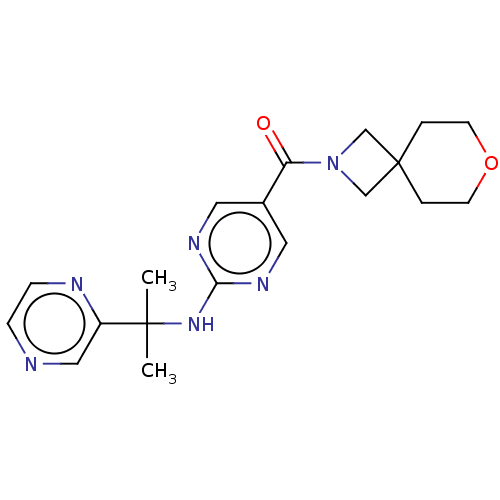 (7-oxa-2-azaspiro[3.5]non-2-yl(2-{[2-(pyrazin-2-yl)...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | <0.0940 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description The test inhibitors were solubilized in DMSO to a stock concentration of 30 mM. On the day of the assay, dose response plates were prepared by diluti... | US Patent US10906888 (2021) BindingDB Entry DOI: 10.7270/Q2QN69WB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM403204 (US10329302, Example 360 | US10793579, Example 360 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description Protocol B: To begin the assay, 45 μL of reaction mixture containing 20 mM HEPES pH=7.5, 5 mM MgCl2, 0.0025% Brij-35, 600 μM ATP, 228 μ... | US Patent US10793579 (2020) BindingDB Entry DOI: 10.7270/Q2H9988P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM403214 (US10329302, Example 370 | US10793579, Example 370 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
St. Jude Research Hospital | Assay Description This is an in vitro assay to measure IRAK4 enzymatic activity utilizing the DELFIA (Dissociation-Enhanced Lanthanide Fluorescent Immunoassay, Perkin-... | ACS Chem Biol 4: 834-43 (2009) BindingDB Entry DOI: 10.7270/Q2FT8PC5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM403204 (US10329302, Example 360 | US10793579, Example 360 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
St. Jude Research Hospital | Assay Description This is an in vitro assay to measure IRAK4 enzymatic activity utilizing the DELFIA (Dissociation-Enhanced Lanthanide Fluorescent Immunoassay, Perkin-... | ACS Chem Biol 4: 834-43 (2009) BindingDB Entry DOI: 10.7270/Q2FT8PC5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM403214 (US10329302, Example 370 | US10793579, Example 370 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description Protocol B: To begin the assay, 45 μL of reaction mixture containing 20 mM HEPES pH=7.5, 5 mM MgCl2, 0.0025% Brij-35, 600 μM ATP, 228 μ... | US Patent US10793579 (2020) BindingDB Entry DOI: 10.7270/Q2H9988P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239499 (CHEMBL4081711 | US10329302, Example 344 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB US Patent | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description Protocol B: To begin the assay, 45 μL of reaction mixture containing 20 mM HEPES pH=7.5, 5 mM MgCl2, 0.0025% Brij-35, 600 μM ATP, 228 μ... | US Patent US10793579 (2020) BindingDB Entry DOI: 10.7270/Q2H9988P | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239499 (CHEMBL4081711 | US10329302, Example 344 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB US Patent | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
St. Jude Research Hospital | Assay Description This is an in vitro assay to measure IRAK4 enzymatic activity utilizing the DELFIA (Dissociation-Enhanced Lanthanide Fluorescent Immunoassay, Perkin-... | ACS Chem Biol 4: 834-43 (2009) BindingDB Entry DOI: 10.7270/Q2FT8PC5 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase kinase kinase 5 (Homo sapiens (Human)) | BDBM50464991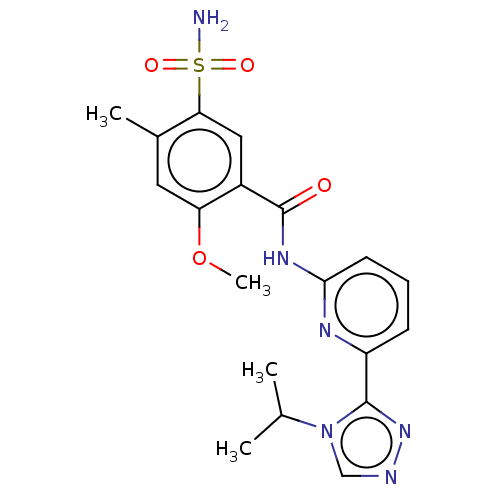 (CHEMBL4283739) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of recombinant full length human GST-tagged ASK1 expressed in baculovirus expression system using STK3 peptide substrate preincubated for ... | Eur J Med Chem 145: 606-621 (2018) Article DOI: 10.1016/j.ejmech.2017.12.041 BindingDB Entry DOI: 10.7270/Q25H7JXQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239500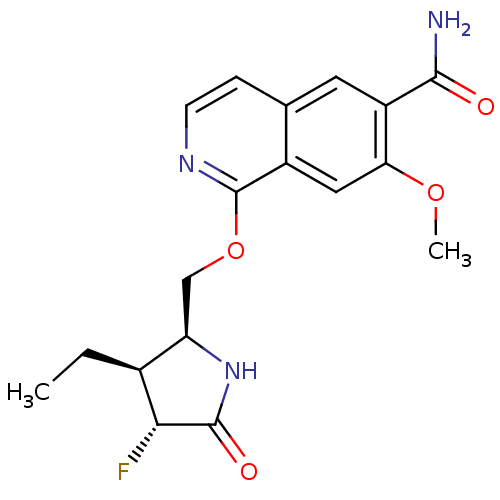 (CHEMBL4066705 | US10329302, Example 337 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of N-terminal His6-tagged human full length IRAK4 preincubated for 20 mins followed by biotinylated-AGAGRDKYKTLRQIR substrate addition in ... | J Med Chem 60: 5521-5542 (2017) Article DOI: 10.1021/acs.jmedchem.7b00231 BindingDB Entry DOI: 10.7270/Q26D5W42 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239499 (CHEMBL4081711 | US10329302, Example 344 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Citation and Details BindingDB Entry DOI: 10.7270/Q27D308N | ||||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239500 (CHEMBL4066705 | US10329302, Example 337 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details BindingDB Entry DOI: 10.7270/Q27D308N | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM403204 (US10329302, Example 360 | US10793579, Example 360 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details BindingDB Entry DOI: 10.7270/Q27D308N | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM403214 (US10329302, Example 370 | US10793579, Example 370 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details BindingDB Entry DOI: 10.7270/Q27D308N | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM319575 (5-(((2S,3S,4S)-3-ethyl-4-fluoro-5-oxopyrrolidin-2-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description This is an in vitro assay to measure IRAK4 enzymatic activity utilizing the DELFIA (Dissociation-Enhanced Lanthanide Fluorescent Immunoassay, Perkin-... | US Patent US10174000 (2019) BindingDB Entry DOI: 10.7270/Q27S7QWQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239500 (CHEMBL4066705 | US10329302, Example 337 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description Protocol B: To begin the assay, 45 μL of reaction mixture containing 20 mM HEPES pH=7.5, 5 mM MgCl2, 0.0025% Brij-35, 600 μM ATP, 228 μ... | US Patent US10793579 (2020) BindingDB Entry DOI: 10.7270/Q2H9988P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239500 (CHEMBL4066705 | US10329302, Example 337 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
St. Jude Research Hospital | Assay Description This is an in vitro assay to measure IRAK4 enzymatic activity utilizing the DELFIA (Dissociation-Enhanced Lanthanide Fluorescent Immunoassay, Perkin-... | ACS Chem Biol 4: 834-43 (2009) BindingDB Entry DOI: 10.7270/Q2FT8PC5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pantetheinase (Homo sapiens (Human)) | BDBM480917 (7-oxa-2-azaspiro[3.5]non-2-yl(2-{[2-(pyrazin-2-yl)...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant N-terminal FLAG/His6-tagged human vanin-1 expressed in CHO cells using Pantetheine-7-amino-4-trifluoromethykournarin as sub... | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c01849 BindingDB Entry DOI: 10.7270/Q2XD15K6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pantetheinase (Homo sapiens (Human)) | BDBM480956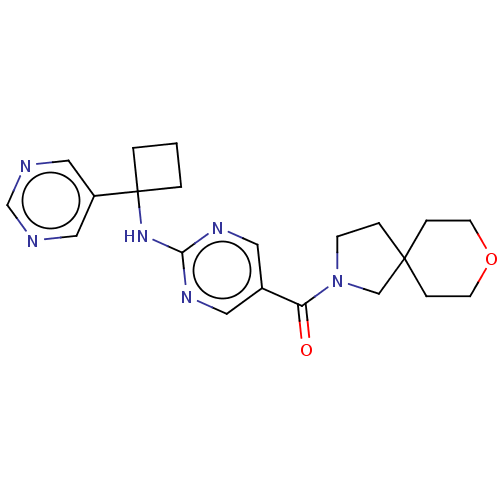 (US10906888, Example 173) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.116 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description The test inhibitors were solubilized in DMSO to a stock concentration of 30 mM. On the day of the assay, dose response plates were prepared by diluti... | US Patent US10906888 (2021) BindingDB Entry DOI: 10.7270/Q2QN69WB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pantetheinase (Homo sapiens (Human)) | BDBM480956 (US10906888, Example 173) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant N-terminal FLAG/His6-tagged human vanin-1 expressed in CHO cells using Pantetheine-7-amino-4-trifluoromethykournarin as sub... | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c01849 BindingDB Entry DOI: 10.7270/Q2XD15K6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pantetheinase (Homo sapiens (Human)) | BDBM480963 (US10906888, Example 180) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.196 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description The test inhibitors were solubilized in DMSO to a stock concentration of 30 mM. On the day of the assay, dose response plates were prepared by diluti... | US Patent US10906888 (2021) BindingDB Entry DOI: 10.7270/Q2QN69WB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM403035 (US10329302, Example 183 | US10793579, Example 183 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details BindingDB Entry DOI: 10.7270/Q27D308N | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM402999 (US10329302, Example 146 | US10793579, Example 146 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details BindingDB Entry DOI: 10.7270/Q27D308N | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239499 (CHEMBL4081711 | US10329302, Example 344 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Citation and Details BindingDB Entry DOI: 10.7270/Q27D308N | ||||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM403189 (US10329302, Example 345 | US10793579, Example 345 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details BindingDB Entry DOI: 10.7270/Q27D308N | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239499 (CHEMBL4081711 | US10329302, Example 344 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Citation and Details BindingDB Entry DOI: 10.7270/Q27D308N | ||||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239499 (CHEMBL4081711 | US10329302, Example 344 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Citation and Details BindingDB Entry DOI: 10.7270/Q27D308N | ||||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM403169 (US10329302, Example 323 | US10793579, Example 323 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details BindingDB Entry DOI: 10.7270/Q27D308N | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM403035 (US10329302, Example 183 | US10793579, Example 183 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description Protocol B: To begin the assay, 45 μL of reaction mixture containing 20 mM HEPES pH=7.5, 5 mM MgCl2, 0.0025% Brij-35, 600 μM ATP, 228 μ... | US Patent US10793579 (2020) BindingDB Entry DOI: 10.7270/Q2H9988P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239499 (CHEMBL4081711 | US10329302, Example 344 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB US Patent | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description Protocol B: To begin the assay, 45 μL of reaction mixture containing 20 mM HEPES pH=7.5, 5 mM MgCl2, 0.0025% Brij-35, 600 μM ATP, 228 μ... | US Patent US10793579 (2020) BindingDB Entry DOI: 10.7270/Q2H9988P | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM403189 (US10329302, Example 345 | US10793579, Example 345 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description Protocol B: To begin the assay, 45 μL of reaction mixture containing 20 mM HEPES pH=7.5, 5 mM MgCl2, 0.0025% Brij-35, 600 μM ATP, 228 μ... | US Patent US10793579 (2020) BindingDB Entry DOI: 10.7270/Q2H9988P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239499 (CHEMBL4081711 | US10329302, Example 344 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB US Patent | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description Protocol B: To begin the assay, 45 μL of reaction mixture containing 20 mM HEPES pH=7.5, 5 mM MgCl2, 0.0025% Brij-35, 600 μM ATP, 228 μ... | US Patent US10793579 (2020) BindingDB Entry DOI: 10.7270/Q2H9988P | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239499 (CHEMBL4081711 | US10329302, Example 344 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB US Patent | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description Protocol B: To begin the assay, 45 μL of reaction mixture containing 20 mM HEPES pH=7.5, 5 mM MgCl2, 0.0025% Brij-35, 600 μM ATP, 228 μ... | US Patent US10793579 (2020) BindingDB Entry DOI: 10.7270/Q2H9988P | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM403169 (US10329302, Example 323 | US10793579, Example 323 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description Protocol B: To begin the assay, 45 μL of reaction mixture containing 20 mM HEPES pH=7.5, 5 mM MgCl2, 0.0025% Brij-35, 600 μM ATP, 228 μ... | US Patent US10793579 (2020) BindingDB Entry DOI: 10.7270/Q2H9988P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM402999 (US10329302, Example 146 | US10793579, Example 146 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. US Patent | Assay Description Protocol B: To begin the assay, 45 μL of reaction mixture containing 20 mM HEPES pH=7.5, 5 mM MgCl2, 0.0025% Brij-35, 600 μM ATP, 228 μ... | US Patent US10793579 (2020) BindingDB Entry DOI: 10.7270/Q2H9988P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239499 (CHEMBL4081711 | US10329302, Example 344 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of N-terminal His6-tagged human full length IRAK4 preincubated for 20 mins followed by biotinylated-AGAGRDKYKTLRQIR substrate addition in ... | J Med Chem 60: 5521-5542 (2017) Article DOI: 10.1021/acs.jmedchem.7b00231 BindingDB Entry DOI: 10.7270/Q26D5W42 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM403035 (US10329302, Example 183 | US10793579, Example 183 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
St. Jude Research Hospital | Assay Description This is an in vitro assay to measure IRAK4 enzymatic activity utilizing the DELFIA (Dissociation-Enhanced Lanthanide Fluorescent Immunoassay, Perkin-... | ACS Chem Biol 4: 834-43 (2009) BindingDB Entry DOI: 10.7270/Q2FT8PC5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM402999 (US10329302, Example 146 | US10793579, Example 146 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
St. Jude Research Hospital | Assay Description This is an in vitro assay to measure IRAK4 enzymatic activity utilizing the DELFIA (Dissociation-Enhanced Lanthanide Fluorescent Immunoassay, Perkin-... | ACS Chem Biol 4: 834-43 (2009) BindingDB Entry DOI: 10.7270/Q2FT8PC5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM403189 (US10329302, Example 345 | US10793579, Example 345 ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
St. Jude Research Hospital | Assay Description This is an in vitro assay to measure IRAK4 enzymatic activity utilizing the DELFIA (Dissociation-Enhanced Lanthanide Fluorescent Immunoassay, Perkin-... | ACS Chem Biol 4: 834-43 (2009) BindingDB Entry DOI: 10.7270/Q2FT8PC5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interleukin-1 receptor-associated kinase 4 (Homo sapiens (Human)) | BDBM50239499 (CHEMBL4081711 | US10329302, Example 344 | US107935...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB US Patent | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
St. Jude Research Hospital | Assay Description This is an in vitro assay to measure IRAK4 enzymatic activity utilizing the DELFIA (Dissociation-Enhanced Lanthanide Fluorescent Immunoassay, Perkin-... | ACS Chem Biol 4: 834-43 (2009) BindingDB Entry DOI: 10.7270/Q2FT8PC5 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Displayed 1 to 50 (of 2321 total ) | Next | Last >> |