 Found 74 hits with Last Name = 'woggon' and Initial = 'wd'
Found 74 hits with Last Name = 'woggon' and Initial = 'wd' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50134247
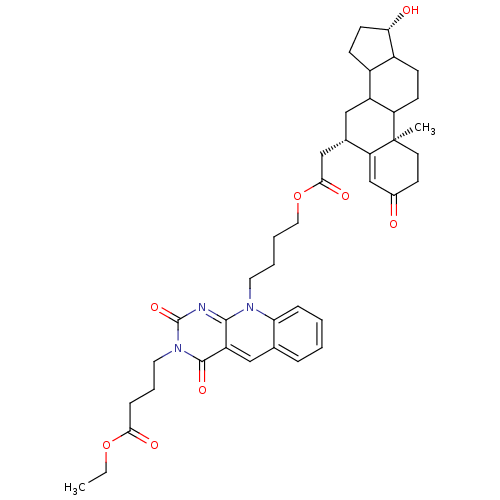
(4-(10-{4-[2-((6S,10R,17S)-17-Hydroxy-10-methyl-3-o...)Show SMILES CCOC(=O)CCCn1c(=O)nc2n(CCCCOC(=O)C[C@@H]3CC4C5CC[C@H](O)C5CCC4[C@@]4(C)CCC(=O)C=C34)c3ccccc3cc2c1=O |t:42| Show InChI InChI=1S/C41H51N3O8/c1-3-51-36(47)11-8-19-44-39(49)31-21-25-9-4-5-10-34(25)43(38(31)42-40(44)50)18-6-7-20-52-37(48)23-26-22-30-28-13-15-35(46)29(28)12-14-32(30)41(2)17-16-27(45)24-33(26)41/h4-5,9-10,21,24,26,28-30,32,35,46H,3,6-8,11-20,22-23H2,1-2H3/t26-,28?,29?,30?,32?,35-,41+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Basel
Curated by ChEMBL
| Assay Description
Inhibition of Cytochrome P450 3A4 with midazolam (4-OH) |
Bioorg Med Chem Lett 13: 3643-5 (2003)
BindingDB Entry DOI: 10.7270/Q2XG9RP9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50134247

(4-(10-{4-[2-((6S,10R,17S)-17-Hydroxy-10-methyl-3-o...)Show SMILES CCOC(=O)CCCn1c(=O)nc2n(CCCCOC(=O)C[C@@H]3CC4C5CC[C@H](O)C5CCC4[C@@]4(C)CCC(=O)C=C34)c3ccccc3cc2c1=O |t:42| Show InChI InChI=1S/C41H51N3O8/c1-3-51-36(47)11-8-19-44-39(49)31-21-25-9-4-5-10-34(25)43(38(31)42-40(44)50)18-6-7-20-52-37(48)23-26-22-30-28-13-15-35(46)29(28)12-14-32(30)41(2)17-16-27(45)24-33(26)41/h4-5,9-10,21,24,26,28-30,32,35,46H,3,6-8,11-20,22-23H2,1-2H3/t26-,28?,29?,30?,32?,35-,41+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Basel
Curated by ChEMBL
| Assay Description
Inhibition of Cytochrome P450 3A4 with midazolam (4-OH) |
Bioorg Med Chem Lett 13: 3643-5 (2003)
BindingDB Entry DOI: 10.7270/Q2XG9RP9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84668
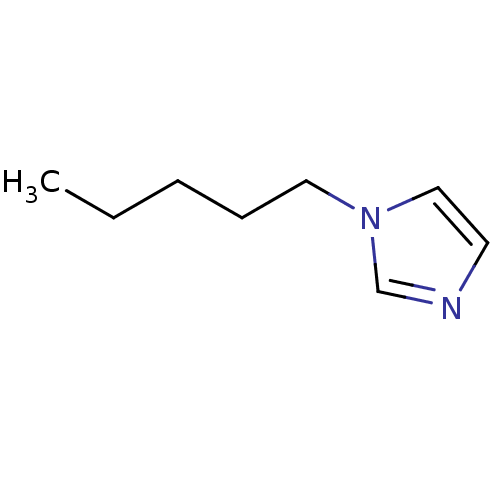
(1-Subsituted 1H-imidazole, 3)Show InChI InChI=1S/C8H14N2/c1-2-3-4-6-10-7-5-9-8-10/h5,7-8H,2-4,6H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84669
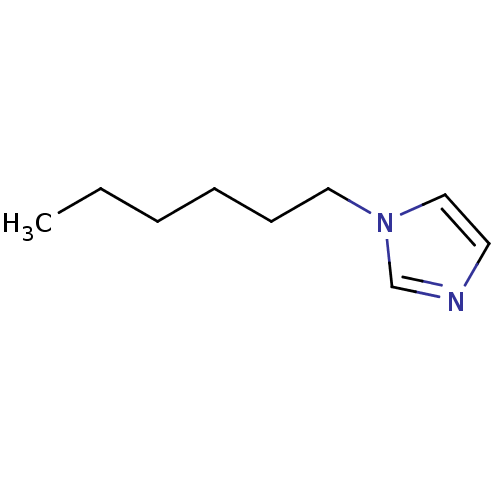
(1-Subsituted 1H-imidazole, 4)Show InChI InChI=1S/C9H16N2/c1-2-3-4-5-7-11-8-6-10-9-11/h6,8-9H,2-5,7H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84676
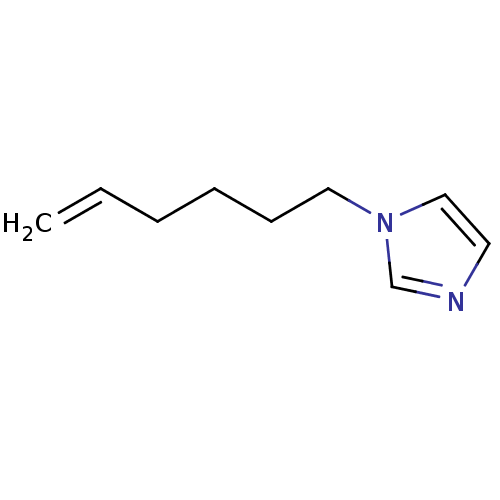
(1-Subsituted 1H-imidazole, 13)Show InChI InChI=1S/C9H14N2/c1-2-3-4-5-7-11-8-6-10-9-11/h2,6,8-9H,1,3-5,7H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50134247

(4-(10-{4-[2-((6S,10R,17S)-17-Hydroxy-10-methyl-3-o...)Show SMILES CCOC(=O)CCCn1c(=O)nc2n(CCCCOC(=O)C[C@@H]3CC4C5CC[C@H](O)C5CCC4[C@@]4(C)CCC(=O)C=C34)c3ccccc3cc2c1=O |t:42| Show InChI InChI=1S/C41H51N3O8/c1-3-51-36(47)11-8-19-44-39(49)31-21-25-9-4-5-10-34(25)43(38(31)42-40(44)50)18-6-7-20-52-37(48)23-26-22-30-28-13-15-35(46)29(28)12-14-32(30)41(2)17-16-27(45)24-33(26)41/h4-5,9-10,21,24,26,28-30,32,35,46H,3,6-8,11-20,22-23H2,1-2H3/t26-,28?,29?,30?,32?,35-,41+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Basel
Curated by ChEMBL
| Assay Description
Inhibition of Cytochrome P450 3A4 with testosterone |
Bioorg Med Chem Lett 13: 3643-5 (2003)
BindingDB Entry DOI: 10.7270/Q2XG9RP9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84669

(1-Subsituted 1H-imidazole, 4)Show InChI InChI=1S/C9H16N2/c1-2-3-4-5-7-11-8-6-10-9-11/h6,8-9H,2-5,7H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84671
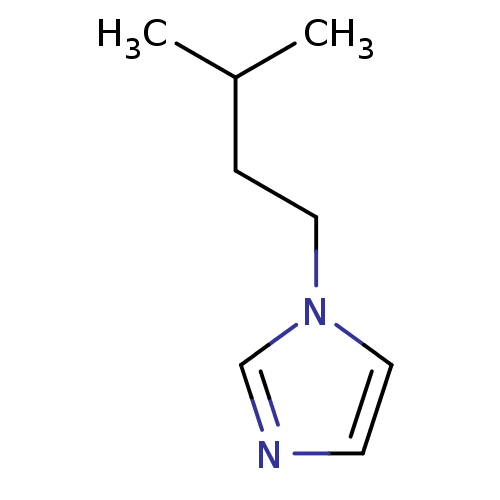
(1-Subsituted 1H-imidazole, 7)Show InChI InChI=1S/C8H14N2/c1-8(2)3-5-10-6-4-9-7-10/h4,6-8H,3,5H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.10E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84670
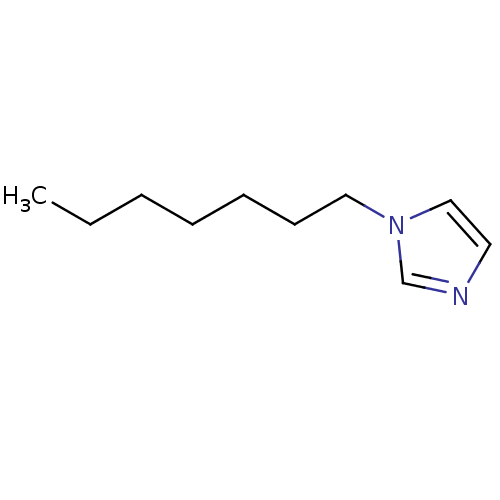
(1-Subsituted 1H-imidazole, 5)Show InChI InChI=1S/C10H18N2/c1-2-3-4-5-6-8-12-9-7-11-10-12/h7,9-10H,2-6,8H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.10E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50134247

(4-(10-{4-[2-((6S,10R,17S)-17-Hydroxy-10-methyl-3-o...)Show SMILES CCOC(=O)CCCn1c(=O)nc2n(CCCCOC(=O)C[C@@H]3CC4C5CC[C@H](O)C5CCC4[C@@]4(C)CCC(=O)C=C34)c3ccccc3cc2c1=O |t:42| Show InChI InChI=1S/C41H51N3O8/c1-3-51-36(47)11-8-19-44-39(49)31-21-25-9-4-5-10-34(25)43(38(31)42-40(44)50)18-6-7-20-52-37(48)23-26-22-30-28-13-15-35(46)29(28)12-14-32(30)41(2)17-16-27(45)24-33(26)41/h4-5,9-10,21,24,26,28-30,32,35,46H,3,6-8,11-20,22-23H2,1-2H3/t26-,28?,29?,30?,32?,35-,41+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 4.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Basel
Curated by ChEMBL
| Assay Description
Inhibition of Cytochrome P450 3A4 with midazolam (1'-OH) |
Bioorg Med Chem Lett 13: 3643-5 (2003)
BindingDB Entry DOI: 10.7270/Q2XG9RP9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84668

(1-Subsituted 1H-imidazole, 3)Show InChI InChI=1S/C8H14N2/c1-2-3-4-6-10-7-5-9-8-10/h5,7-8H,2-4,6H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.30E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84674
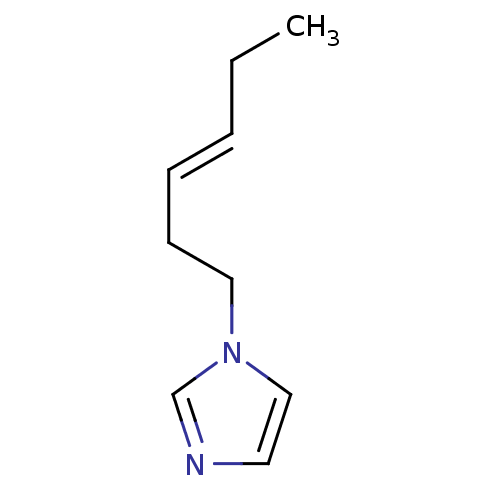
(1-Subsituted 1H-imidazole, 11 | 1-Subsituted 1H-im...)Show InChI InChI=1S/C9H14N2/c1-2-3-4-5-7-11-8-6-10-9-11/h3-4,6,8-9H,2,5,7H2,1H3/b4-3+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.50E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84671

(1-Subsituted 1H-imidazole, 7)Show InChI InChI=1S/C8H14N2/c1-8(2)3-5-10-6-4-9-7-10/h4,6-8H,3,5H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.60E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84672
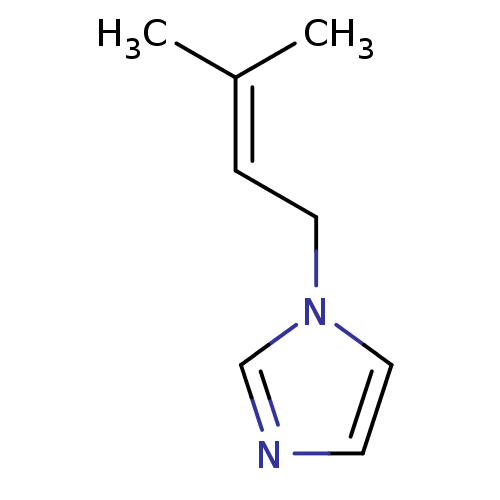
(1-Subsituted 1H-imidazole, 9)Show InChI InChI=1S/C8H12N2/c1-8(2)3-5-10-6-4-9-7-10/h3-4,6-7H,5H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.70E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84677
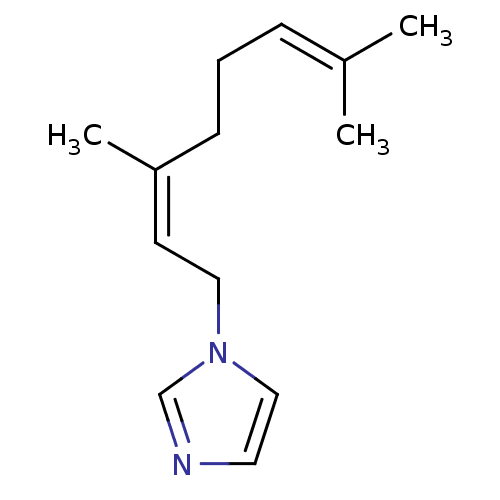
(1-Subsituted 1H-imidazole, 14)Show SMILES [#6]\[#6](-[#6])=[#6]\[#6]-[#6]\[#6](-[#6])=[#6]/[#6]-n1ccnc1 Show InChI InChI=1S/C13H20N2/c1-12(2)5-4-6-13(3)7-9-15-10-8-14-11-15/h5,7-8,10-11H,4,6,9H2,1-3H3/b13-7- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 5.70E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84677

(1-Subsituted 1H-imidazole, 14)Show SMILES [#6]\[#6](-[#6])=[#6]\[#6]-[#6]\[#6](-[#6])=[#6]/[#6]-n1ccnc1 Show InChI InChI=1S/C13H20N2/c1-12(2)5-4-6-13(3)7-9-15-10-8-14-11-15/h5,7-8,10-11H,4,6,9H2,1-3H3/b13-7- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 6.50E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84673
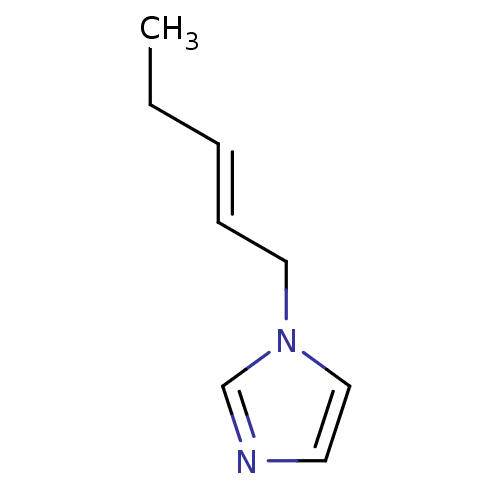
(1-Subsituted 1H-imidazole, 10)Show InChI InChI=1S/C8H12N2/c1-2-3-4-6-10-7-5-9-8-10/h3-5,7-8H,2,6H2,1H3/b4-3+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.70E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84674

(1-Subsituted 1H-imidazole, 11 | 1-Subsituted 1H-im...)Show InChI InChI=1S/C9H14N2/c1-2-3-4-5-7-11-8-6-10-9-11/h3-4,6,8-9H,2,5,7H2,1H3/b4-3+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.80E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84674

(1-Subsituted 1H-imidazole, 11 | 1-Subsituted 1H-im...)Show InChI InChI=1S/C9H14N2/c1-2-3-4-5-7-11-8-6-10-9-11/h3-4,6,8-9H,2,5,7H2,1H3/b4-3+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7.10E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84672

(1-Subsituted 1H-imidazole, 9)Show InChI InChI=1S/C8H12N2/c1-8(2)3-5-10-6-4-9-7-10/h3-4,6-7H,5H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7.70E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84694
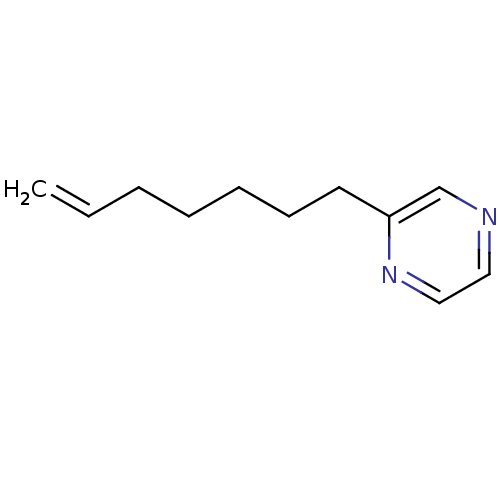
(2-Subsituted pyrazine, 34)Show InChI InChI=1S/C11H16N2/c1-2-3-4-5-6-7-11-10-12-8-9-13-11/h2,8-10H,1,3-7H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9.80E+3 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50134248
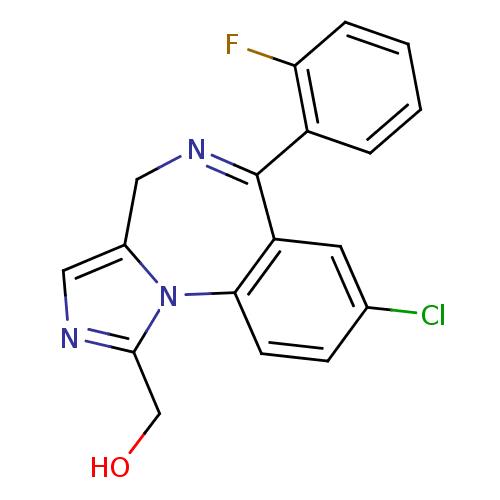
(Alpha-Hydroxy-Midazolam | CHEMBL1188 | US9333197, ...)Show SMILES OCc1ncc2CN=C(c3ccccc3F)c3cc(Cl)ccc3-n12 |t:7| Show InChI InChI=1S/C18H13ClFN3O/c19-11-5-6-16-14(7-11)18(13-3-1-2-4-15(13)20)22-9-12-8-21-17(10-24)23(12)16/h1-8,24H,9-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Basel
Curated by ChEMBL
| Assay Description
Binding affinity for Cytochrome P450 3A4; Range = 1-10 uM |
Bioorg Med Chem Lett 13: 3643-5 (2003)
BindingDB Entry DOI: 10.7270/Q2XG9RP9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50336640

((nifedipine) 2,6-Dimethyl-4-(2-nitro-phenyl)-1,4-d...)Show SMILES COC(=O)C1C(C(C(=O)OC)=C(C)N=C1C)c1ccccc1[N+]([O-])=O |c:13,t:10| Show InChI InChI=1S/C17H18N2O6/c1-9-13(16(20)24-3)15(14(10(2)18-9)17(21)25-4)11-7-5-6-8-12(11)19(22)23/h5-8,13,15H,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Basel
Curated by ChEMBL
| Assay Description
Binding affinity for Cytochrome P450 3A4; Range = 1-10 uM |
Bioorg Med Chem Lett 13: 3643-5 (2003)
BindingDB Entry DOI: 10.7270/Q2XG9RP9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50134246
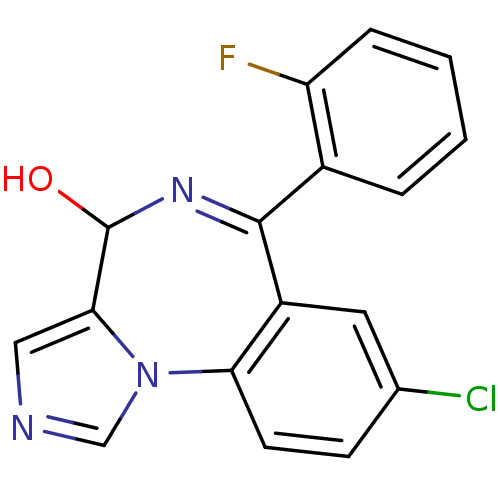
(8-Chloro-6-(2-fluoro-phenyl)-4H-2,5,10b-triaza-ben...)Show SMILES OC1N=C(c2ccccc2F)c2cc(Cl)ccc2-n2cncc12 |t:2| Show InChI InChI=1S/C17H11ClFN3O/c18-10-5-6-14-12(7-10)16(11-3-1-2-4-13(11)19)21-17(23)15-8-20-9-22(14)15/h1-9,17,23H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Basel
Curated by ChEMBL
| Assay Description
Binding affinity for Cytochrome P450 3A4; Range = 1-10 uM |
Bioorg Med Chem Lett 13: 3643-5 (2003)
BindingDB Entry DOI: 10.7270/Q2XG9RP9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM8885

((1S,2R,10R,11S,14S,15S)-14-hydroxy-2,15-dimethylte...)Show SMILES [H][C@@]12CC[C@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])CCC2=CC(=O)CC[C@]12C |r,t:18| Show InChI InChI=1S/C19H28O2/c1-18-9-7-13(20)11-12(18)3-4-14-15-5-6-17(21)19(15,2)10-8-16(14)18/h11,14-17,21H,3-10H2,1-2H3/t14-,15-,16-,17-,18-,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
PubMed
| n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Basel
Curated by ChEMBL
| Assay Description
Binding affinity for Cytochrome P450 3A4; Range = 1-10 uM |
Bioorg Med Chem Lett 13: 3643-5 (2003)
BindingDB Entry DOI: 10.7270/Q2XG9RP9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84695
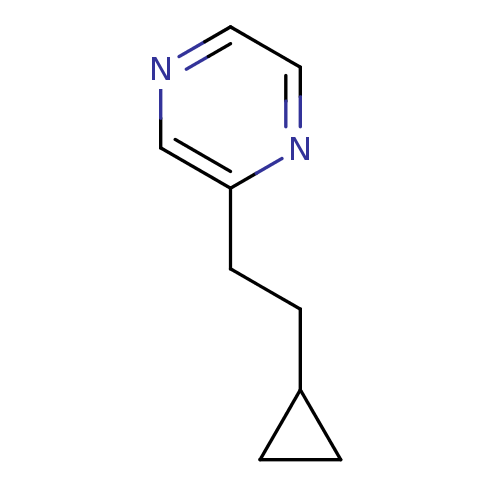
(2-Subsituted pyrazine, 35)Show InChI InChI=1S/C9H12N2/c1-2-8(1)3-4-9-7-10-5-6-11-9/h5-8H,1-4H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.03E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84686
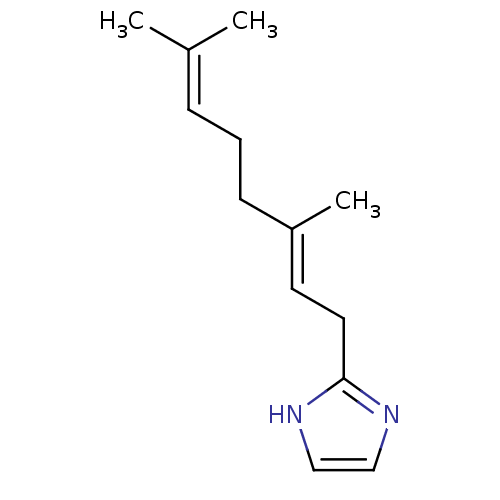
(2-Subsituted 1H-imidazole, 23)Show SMILES CC(C)=CCC\C(C)=C\Cc1ncc[nH]1 |(-3.96,12.47,;-5.29,11.7,;-6.63,12.47,;-5.29,10.16,;-6.63,9.39,;-6.63,7.85,;-7.96,7.08,;-9.3,7.85,;-7.96,5.54,;-9.3,4.77,;-9.3,3.23,;-10.54,2.33,;-10.07,.87,;-8.53,.87,;-8.05,2.33,)| Show InChI InChI=1S/C13H20N2/c1-11(2)5-4-6-12(3)7-8-13-14-9-10-15-13/h5,7,9-10H,4,6,8H2,1-3H3,(H,14,15)/b12-7+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.07E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84670

(1-Subsituted 1H-imidazole, 5)Show InChI InChI=1S/C10H18N2/c1-2-3-4-5-6-8-12-9-7-11-10-12/h7,9-10H,2-6,8H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84693
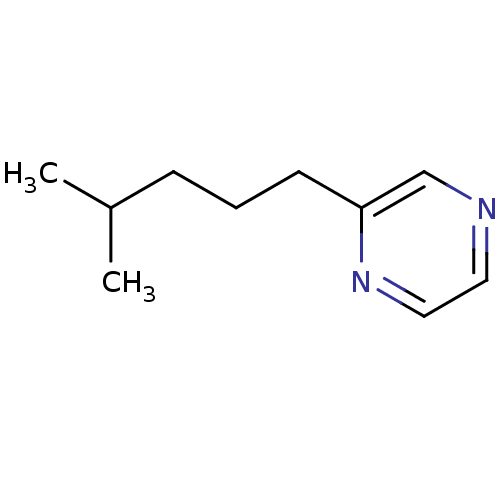
(2-Subsituted pyrazine, 33)Show InChI InChI=1S/C10H16N2/c1-9(2)4-3-5-10-8-11-6-7-12-10/h6-9H,3-5H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.12E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84673

(1-Subsituted 1H-imidazole, 10)Show InChI InChI=1S/C8H12N2/c1-2-3-4-6-10-7-5-9-8-10/h3-5,7-8H,2,6H2,1H3/b4-3+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.14E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84674

(1-Subsituted 1H-imidazole, 11 | 1-Subsituted 1H-im...)Show InChI InChI=1S/C9H14N2/c1-2-3-4-5-7-11-8-6-10-9-11/h3-4,6,8-9H,2,5,7H2,1H3/b4-3+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.15E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84676

(1-Subsituted 1H-imidazole, 13)Show InChI InChI=1S/C9H14N2/c1-2-3-4-5-7-11-8-6-10-9-11/h2,6,8-9H,1,3-5,7H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.40E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84685
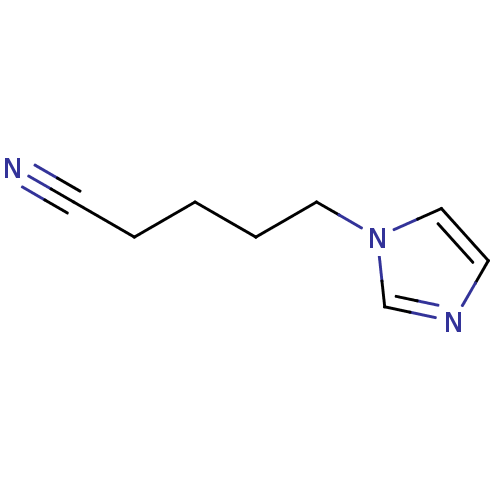
(1-Subsituted 1H-imidazole, 22)Show InChI InChI=1S/C8H11N3/c9-4-2-1-3-6-11-7-5-10-8-11/h5,7-8H,1-3,6H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.91E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84693

(2-Subsituted pyrazine, 33)Show InChI InChI=1S/C10H16N2/c1-9(2)4-3-5-10-8-11-6-7-12-10/h6-9H,3-5H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.16E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84685

(1-Subsituted 1H-imidazole, 22)Show InChI InChI=1S/C8H11N3/c9-4-2-1-3-6-11-7-5-10-8-11/h5,7-8H,1-3,6H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.24E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84694

(2-Subsituted pyrazine, 34)Show InChI InChI=1S/C11H16N2/c1-2-3-4-5-6-7-11-10-12-8-9-13-11/h2,8-10H,1,3-7H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.38E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84696
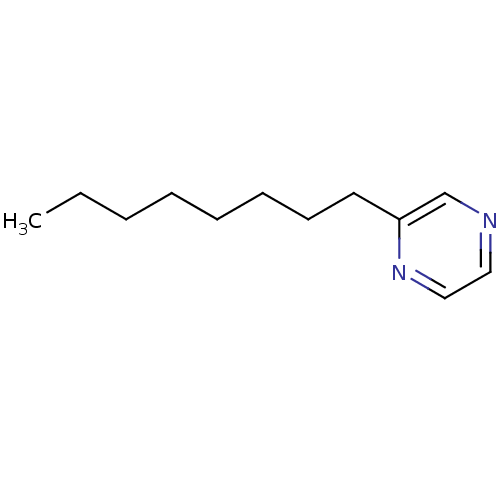
(2-Subsituted pyrazine, 36)Show InChI InChI=1S/C12H20N2/c1-2-3-4-5-6-7-8-12-11-13-9-10-14-12/h9-11H,2-8H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.30E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84695

(2-Subsituted pyrazine, 35)Show InChI InChI=1S/C9H12N2/c1-2-8(1)3-4-9-7-10-5-6-11-9/h5-8H,1-4H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.90E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM36010
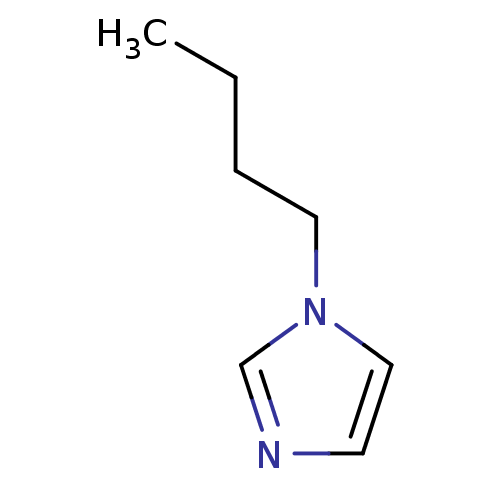
(1-Subsituted 1H-imidazole, 2 | 1-butylimidazole)Show InChI InChI=1S/C7H12N2/c1-2-3-5-9-6-4-8-7-9/h4,6-7H,2-3,5H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.40E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84690
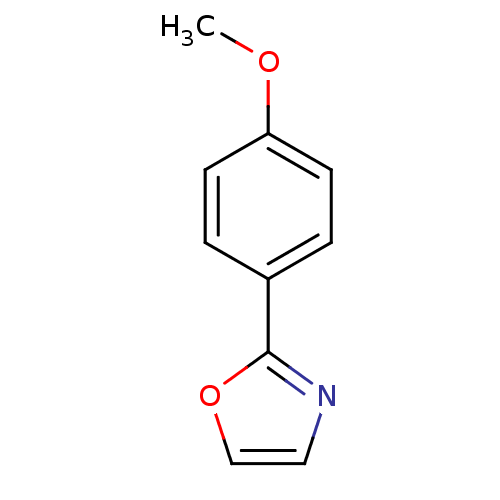
(2-Subsituted oxazole, 29)Show InChI InChI=1S/C10H9NO2/c1-12-9-4-2-8(3-5-9)10-11-6-7-13-10/h2-7H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 4.90E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM50149347
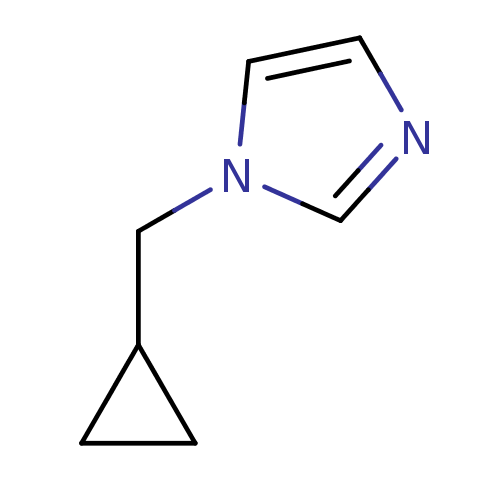
(1-Cyclopropylmethyl-1H-imidazole | 1-Subsituted 1H...)Show InChI InChI=1S/C7H10N2/c1-2-7(1)5-9-4-3-8-6-9/h3-4,6-7H,1-2,5H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.40E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84678
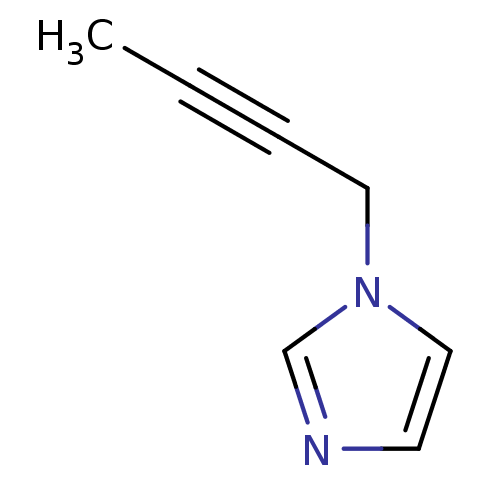
(1-Subsituted 1H-imidazole, 15)Show InChI InChI=1S/C7H8N2/c1-2-3-5-9-6-4-8-7-9/h4,6-7H,5H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.40E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84684

(1-Subsituted 1H-imidazole, 21 | CHEMBL543494)Show InChI InChI=1S/C8H12N2O2/c11-8(12)3-1-2-5-10-6-4-9-7-10/h4,6-7H,1-3,5H2,(H,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8.20E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM36010

(1-Subsituted 1H-imidazole, 2 | 1-butylimidazole)Show InChI InChI=1S/C7H12N2/c1-2-3-5-9-6-4-8-7-9/h4,6-7H,2-3,5H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8.60E+4 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84667
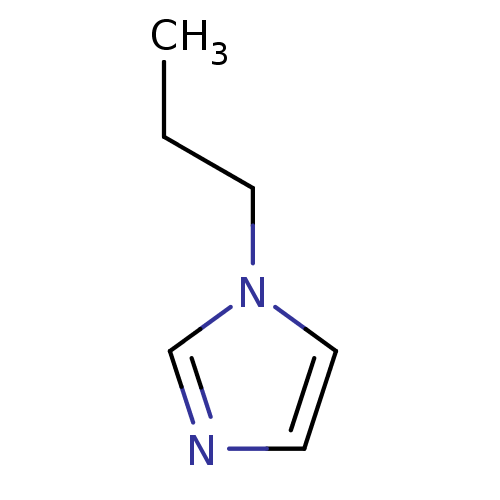
(1-Subsituted 1H-imidazole, 1)Show InChI InChI=1S/C6H10N2/c1-2-4-8-5-3-7-6-8/h3,5-6H,2,4H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.10E+5 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84696

(2-Subsituted pyrazine, 36)Show InChI InChI=1S/C12H20N2/c1-2-3-4-5-6-7-8-12-11-13-9-10-14-12/h9-11H,2-8H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.15E+5 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84683
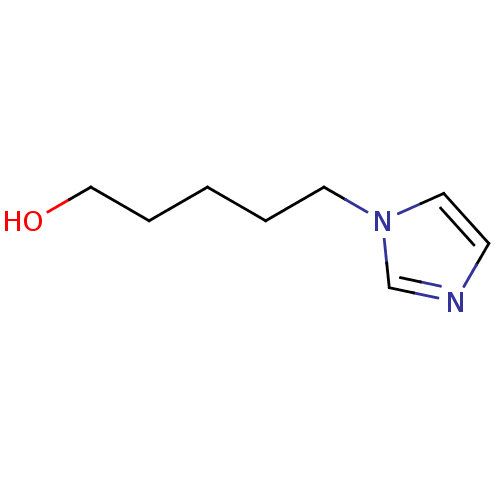
(1-Subsituted 1H-imidazole, 20)Show InChI InChI=1S/C8H14N2O/c11-7-3-1-2-5-10-6-4-9-8-10/h4,6,8,11H,1-3,5,7H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.22E+5 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84681
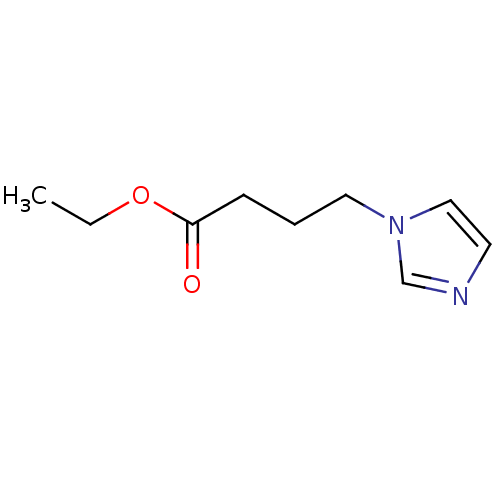
(1-Subsituted 1H-imidazole, 18)Show InChI InChI=1S/C9H14N2O2/c1-2-13-9(12)4-3-6-11-7-5-10-8-11/h5,7-8H,2-4,6H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.23E+5 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM84679
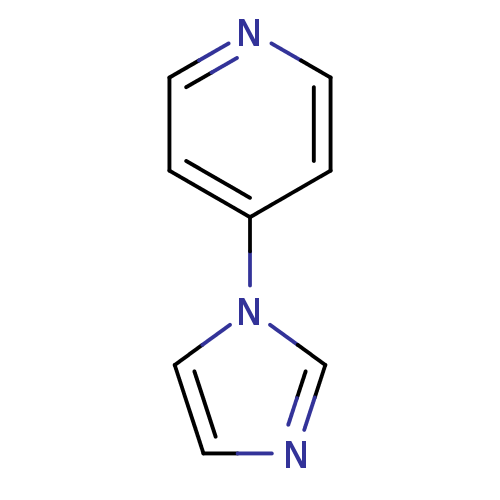
(1-Subsituted 1H-imidazole, 16)Show InChI InChI=1S/C8H7N3/c1-3-9-4-2-8(1)11-6-5-10-7-11/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 1.36E+5 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A13
(Homo sapiens (Human)) | BDBM84689
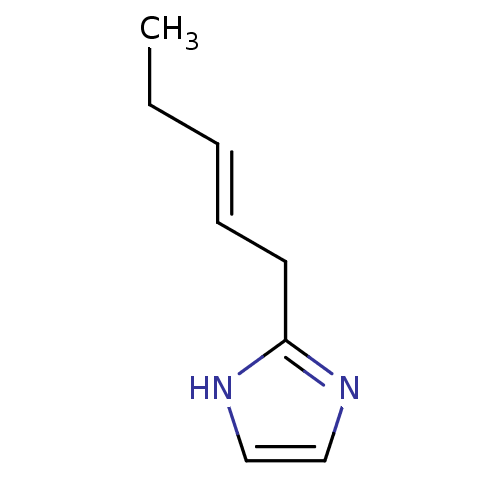
(2-Subsituted 1H-imidazole, 28)Show InChI InChI=1S/C8H12N2/c1-2-3-4-5-8-9-6-7-10-8/h3-4,6-7H,2,5H2,1H3,(H,9,10)/b4-3+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.43E+5 | n/a | n/a | n/a | n/a | 7.6 | 37 |
University of Basel
| Assay Description
Inhibition assay using cytochrome P450 enzyme CYP2A6 and CYP2A13. |
Chembiochem 10: 1562-7 (2009)
Article DOI: 10.1002/cbic.200800712
BindingDB Entry DOI: 10.7270/Q2R78CR9 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data