

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Protease (Human immunodeficiency virus 1 (HIV-1)) | BDBM50067593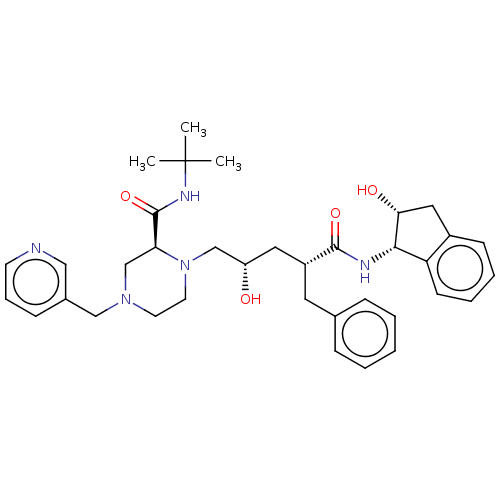 (CHEBI:44032 | Crixivan | Indinavir | L-735524 | MK...) | PDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | Purchase DrugBank MCE KEGG PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | <10 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Biotechnology Curated by ChEMBL | Assay Description Inhibition of HIV1 recombinant protease | Eur J Med Chem 45: 4096-103 (2010) Article DOI: 10.1016/j.ejmech.2010.05.070 BindingDB Entry DOI: 10.7270/Q2C53PP4 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16034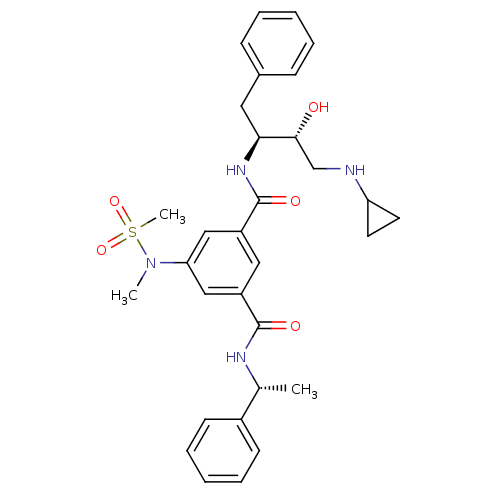 (1-N-[(2S,3R)-4-(cyclopropylamino)-3-hydroxy-1-phen...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Pharmacology and Toxicology Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 proteolytic activity | Bioorg Med Chem Lett 22: 1408-14 (2012) Article DOI: 10.1016/j.bmcl.2011.12.034 BindingDB Entry DOI: 10.7270/Q24Q7VGC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16034 (1-N-[(2S,3R)-4-(cyclopropylamino)-3-hydroxy-1-phen...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Pharmacology and Toxicology Curated by ChEMBL | Assay Description Inhibition of BACE1 | Bioorg Med Chem Lett 22: 1408-14 (2012) Article DOI: 10.1016/j.bmcl.2011.12.034 BindingDB Entry DOI: 10.7270/Q24Q7VGC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16034 (1-N-[(2S,3R)-4-(cyclopropylamino)-3-hydroxy-1-phen...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 19.9 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Pharmacology and Toxicology Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 using Eu-CEVNLDAEFK-Qsy7 as substrate by HTRF assay | Bioorg Med Chem Lett 22: 1408-14 (2012) Article DOI: 10.1016/j.bmcl.2011.12.034 BindingDB Entry DOI: 10.7270/Q24Q7VGC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16034 (1-N-[(2S,3R)-4-(cyclopropylamino)-3-hydroxy-1-phen...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Pharmacology and Toxicology Curated by ChEMBL | Assay Description Inhibition of BACE1 expressed in HEK293T cells co-transfected APP with NFEV mutation at proteolytic site by sAPP_NF cell based assay | Bioorg Med Chem Lett 22: 1408-14 (2012) Article DOI: 10.1016/j.bmcl.2011.12.034 BindingDB Entry DOI: 10.7270/Q24Q7VGC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16034 (1-N-[(2S,3R)-4-(cyclopropylamino)-3-hydroxy-1-phen...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 45 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Pharmacology and Toxicology Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 expressed in CHO cells co-transfected with human APP with Swedish mutation assessed as amyloid beta1-40 secreti... | Bioorg Med Chem Lett 22: 1408-14 (2012) Article DOI: 10.1016/j.bmcl.2011.12.034 BindingDB Entry DOI: 10.7270/Q24Q7VGC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50070942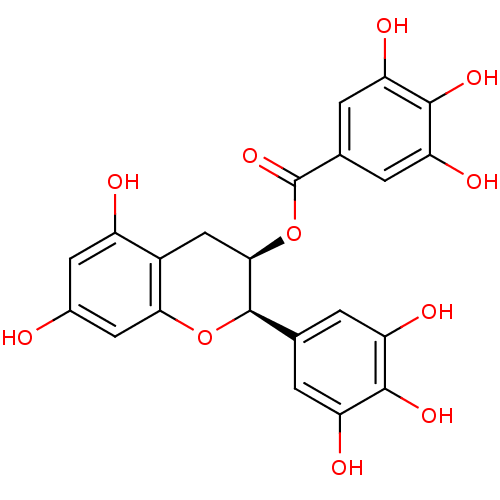 ((-)-Epigallocatechin gallate | (-)-Epigallocatechi...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 757 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Pharmacology and Toxicology Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 using Eu-CEVNLDAEFK-Qsy7 as substrate by HTRF assay | Bioorg Med Chem Lett 22: 1408-14 (2012) Article DOI: 10.1016/j.bmcl.2011.12.034 BindingDB Entry DOI: 10.7270/Q24Q7VGC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50070942 ((-)-Epigallocatechin gallate | (-)-Epigallocatechi...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Pharmacology and Toxicology Curated by ChEMBL | Assay Description Inhibition of BACE1 using Rh-EVNLDAEFK-Quencher as substrate | Bioorg Med Chem Lett 22: 1408-14 (2012) Article DOI: 10.1016/j.bmcl.2011.12.034 BindingDB Entry DOI: 10.7270/Q24Q7VGC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50364303 (CHEMBL1952315) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.27E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Pharmacology and Toxicology Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 using Eu-CEVNLDAEFK-Qsy7 as substrate by HTRF assay | Bioorg Med Chem Lett 22: 1408-14 (2012) Article DOI: 10.1016/j.bmcl.2011.12.034 BindingDB Entry DOI: 10.7270/Q24Q7VGC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50364304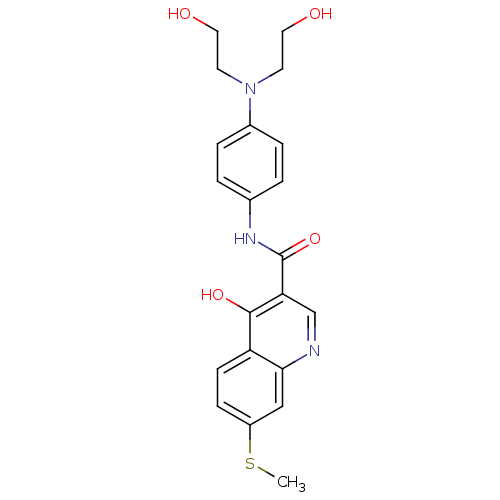 (CHEMBL1952316) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.81E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Pharmacology and Toxicology Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 using Eu-CEVNLDAEFK-Qsy7 as substrate by HTRF assay | Bioorg Med Chem Lett 22: 1408-14 (2012) Article DOI: 10.1016/j.bmcl.2011.12.034 BindingDB Entry DOI: 10.7270/Q24Q7VGC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50364305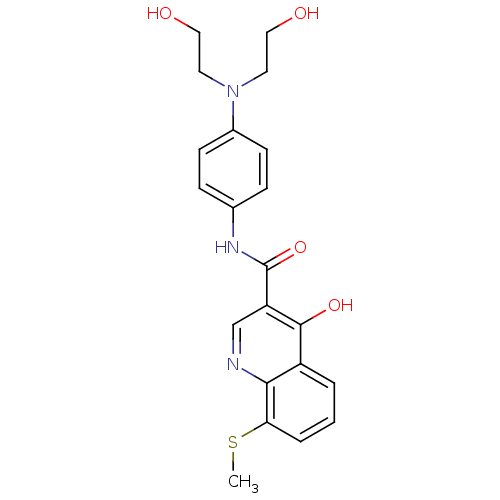 (CHEMBL1952317) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.84E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Pharmacology and Toxicology Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 using Eu-CEVNLDAEFK-Qsy7 as substrate by HTRF assay | Bioorg Med Chem Lett 22: 1408-14 (2012) Article DOI: 10.1016/j.bmcl.2011.12.034 BindingDB Entry DOI: 10.7270/Q24Q7VGC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase (Human immunodeficiency virus 1) | BDBM1434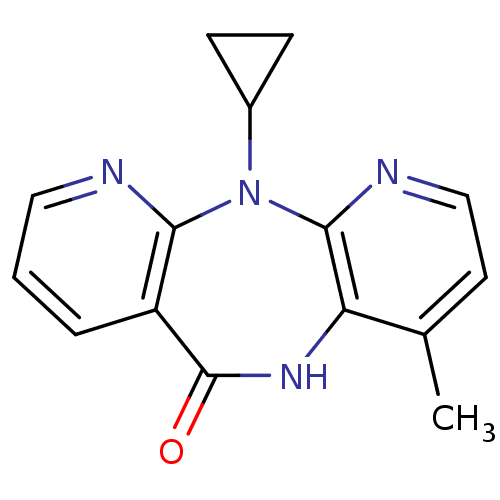 (11-cyclopropyl-5,11-dihydro-4-methyl-6H-dipyrido[3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.32E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Biotechnology Curated by ChEMBL | Assay Description Inhibition of HIV1 reverse transcriptase after 1 hr by ELISA | Eur J Med Chem 45: 4096-103 (2010) Article DOI: 10.1016/j.ejmech.2010.05.070 BindingDB Entry DOI: 10.7270/Q2C53PP4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protease (Human immunodeficiency virus 1 (HIV-1)) | BDBM50482685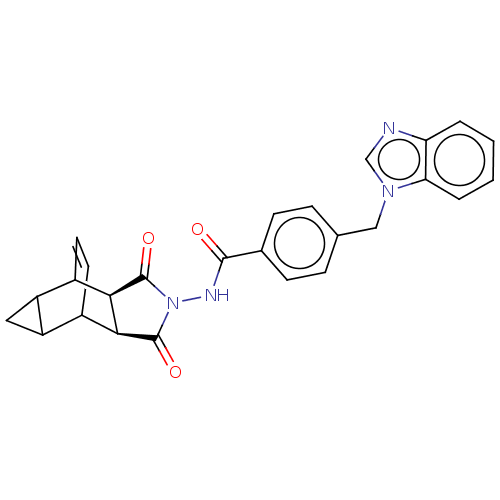 (CHEMBL1241554) | PDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | >1.14E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Biotechnology Curated by ChEMBL | Assay Description Inhibition of HIV1 recombinant protease | Eur J Med Chem 45: 4096-103 (2010) Article DOI: 10.1016/j.ejmech.2010.05.070 BindingDB Entry DOI: 10.7270/Q2C53PP4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50482685 (CHEMBL1241554) | PDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | >2.28E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Biotechnology Curated by ChEMBL | Assay Description Inhibition of HIV1 integrase after 1 hr | Eur J Med Chem 45: 4096-103 (2010) Article DOI: 10.1016/j.ejmech.2010.05.070 BindingDB Entry DOI: 10.7270/Q2C53PP4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase (Human immunodeficiency virus 1) | BDBM50482685 (CHEMBL1241554) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | >4.57E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Beijing Institute of Biotechnology Curated by ChEMBL | Assay Description Inhibition of HIV1 reverse transcriptase after 1 hr by ELISA | Eur J Med Chem 45: 4096-103 (2010) Article DOI: 10.1016/j.ejmech.2010.05.070 BindingDB Entry DOI: 10.7270/Q2C53PP4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16034 (1-N-[(2S,3R)-4-(cyclopropylamino)-3-hydroxy-1-phen...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 43 | n/a | n/a | n/a | n/a |
Beijing Institute of Pharmacology and Toxicology Curated by ChEMBL | Assay Description Inhibition of BACE1 expressed in HEK293T cells co-transfected APP with NFEV mutation at proteolytic site assessed as amyloid EV40 secretion | Bioorg Med Chem Lett 22: 1408-14 (2012) Article DOI: 10.1016/j.bmcl.2011.12.034 BindingDB Entry DOI: 10.7270/Q24Q7VGC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||