 Found 9 hits in this display
Found 9 hits in this display Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Neuromedin-K receptor
(Homo sapiens (Human)) | BDBM251922
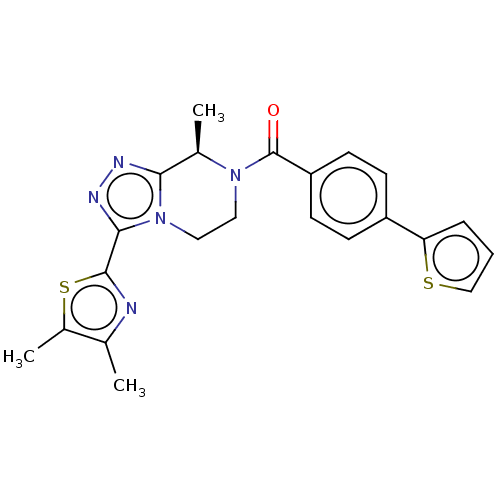
(US10065961, Compound 22 | US10683295, Compound 22 ...)Show SMILES C[C@H]1N(CCn2c1nnc2-c1nc(C)c(C)s1)C(=O)c1ccc(cc1)-c1cccs1 |r| Show InChI InChI=1S/C22H21N5OS2/c1-13-15(3)30-21(23-13)20-25-24-19-14(2)26(10-11-27(19)20)22(28)17-8-6-16(7-9-17)18-5-4-12-29-18/h4-9,12,14H,10-11H2,1-3H3/t14-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Euroscreen S.A.
US Patent
| Assay Description
The ability of compounds of the invention to inhibit the binding of the NK-3 receptor selective antagonist 3H-SB222200 was assessed by an in vitro ra... |
US Patent US9475814 (2016)
BindingDB Entry DOI: 10.7270/Q2B8571Q |
More data for this
Ligand-Target Pair | |
Neuromedin-K receptor
(Homo sapiens (Human)) | BDBM251922

(US10065961, Compound 22 | US10683295, Compound 22 ...)Show SMILES C[C@H]1N(CCn2c1nnc2-c1nc(C)c(C)s1)C(=O)c1ccc(cc1)-c1cccs1 |r| Show InChI InChI=1S/C22H21N5OS2/c1-13-15(3)30-21(23-13)20-25-24-19-14(2)26(10-11-27(19)20)22(28)17-8-6-16(7-9-17)18-5-4-12-29-18/h4-9,12,14H,10-11H2,1-3H3/t14-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ogeda SA
US Patent
| Assay Description
NK3: The ability of compounds of the invention to inhibit the binding of the NK-3 receptor selective antagonist 3H-SB222200 was assessed by an in vit... |
US Patent US10683295 (2020)
BindingDB Entry DOI: 10.7270/Q2S75KC1 |
More data for this
Ligand-Target Pair | |
Neuromedin-K receptor
(Homo sapiens (Human)) | BDBM251922

(US10065961, Compound 22 | US10683295, Compound 22 ...)Show SMILES C[C@H]1N(CCn2c1nnc2-c1nc(C)c(C)s1)C(=O)c1ccc(cc1)-c1cccs1 |r| Show InChI InChI=1S/C22H21N5OS2/c1-13-15(3)30-21(23-13)20-25-24-19-14(2)26(10-11-27(19)20)22(28)17-8-6-16(7-9-17)18-5-4-12-29-18/h4-9,12,14H,10-11H2,1-3H3/t14-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ogeda SA.
US Patent
| Assay Description
The antagonist activity of compounds of the invention is measured following pre-incubation (3 minutes) of the compound with the cells, followed by ad... |
US Patent US10065961 (2018)
BindingDB Entry DOI: 10.7270/Q2MW2K4R |
More data for this
Ligand-Target Pair | |
Neuromedin-K receptor
(Homo sapiens (Human)) | BDBM251922

(US10065961, Compound 22 | US10683295, Compound 22 ...)Show SMILES C[C@H]1N(CCn2c1nnc2-c1nc(C)c(C)s1)C(=O)c1ccc(cc1)-c1cccs1 |r| Show InChI InChI=1S/C22H21N5OS2/c1-13-15(3)30-21(23-13)20-25-24-19-14(2)26(10-11-27(19)20)22(28)17-8-6-16(7-9-17)18-5-4-12-29-18/h4-9,12,14H,10-11H2,1-3H3/t14-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ogeda SA
US Patent
| Assay Description
NK3: The ability of compounds of the invention to inhibit the binding of the NK-3 receptor selective antagonist 3H-SB222200 was assessed by an in vit... |
US Patent US10941151 (2021)
BindingDB Entry DOI: 10.7270/Q2PC35G3 |
More data for this
Ligand-Target Pair | |
Neuromedin-K receptor
(Homo sapiens (Human)) | BDBM251922

(US10065961, Compound 22 | US10683295, Compound 22 ...)Show SMILES C[C@H]1N(CCn2c1nnc2-c1nc(C)c(C)s1)C(=O)c1ccc(cc1)-c1cccs1 |r| Show InChI InChI=1S/C22H21N5OS2/c1-13-15(3)30-21(23-13)20-25-24-19-14(2)26(10-11-27(19)20)22(28)17-8-6-16(7-9-17)18-5-4-12-29-18/h4-9,12,14H,10-11H2,1-3H3/t14-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Euroscreen S.A.
US Patent
| Assay Description
Changes in intracellular calcium levels are a recognized indicator of G protein-coupled receptor activity. The efficacy of compounds of the invention... |
US Patent US9475814 (2016)
BindingDB Entry DOI: 10.7270/Q2B8571Q |
More data for this
Ligand-Target Pair | |
Neuromedin-K receptor
(Homo sapiens (Human)) | BDBM251922

(US10065961, Compound 22 | US10683295, Compound 22 ...)Show SMILES C[C@H]1N(CCn2c1nnc2-c1nc(C)c(C)s1)C(=O)c1ccc(cc1)-c1cccs1 |r| Show InChI InChI=1S/C22H21N5OS2/c1-13-15(3)30-21(23-13)20-25-24-19-14(2)26(10-11-27(19)20)22(28)17-8-6-16(7-9-17)18-5-4-12-29-18/h4-9,12,14H,10-11H2,1-3H3/t14-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Ogeda SA.
US Patent
| Assay Description
The antagonist activity of compounds of the invention is measured following pre-incubation (3 minutes) of the compound with the cells, followed by ad... |
US Patent US10065961 (2018)
BindingDB Entry DOI: 10.7270/Q2MW2K4R |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM251922

(US10065961, Compound 22 | US10683295, Compound 22 ...)Show SMILES C[C@H]1N(CCn2c1nnc2-c1nc(C)c(C)s1)C(=O)c1ccc(cc1)-c1cccs1 |r| Show InChI InChI=1S/C22H21N5OS2/c1-13-15(3)30-21(23-13)20-25-24-19-14(2)26(10-11-27(19)20)22(28)17-8-6-16(7-9-17)18-5-4-12-29-18/h4-9,12,14H,10-11H2,1-3H3/t14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Ogeda SA
US Patent
| Assay Description
hERG: The hERG inhibition study aims at quantifying the in vitro effects of compounds of the invention on the potassium-selective IKr current generat... |
US Patent US10683295 (2020)
BindingDB Entry DOI: 10.7270/Q2S75KC1 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM251922

(US10065961, Compound 22 | US10683295, Compound 22 ...)Show SMILES C[C@H]1N(CCn2c1nnc2-c1nc(C)c(C)s1)C(=O)c1ccc(cc1)-c1cccs1 |r| Show InChI InChI=1S/C22H21N5OS2/c1-13-15(3)30-21(23-13)20-25-24-19-14(2)26(10-11-27(19)20)22(28)17-8-6-16(7-9-17)18-5-4-12-29-18/h4-9,12,14H,10-11H2,1-3H3/t14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Euroscreen S.A.
US Patent
| Assay Description
The hERG inhibition study aims at quantifying the in vitro effects of compounds of the invention on the potassium-selective IKf current generated in ... |
US Patent US9475814 (2016)
BindingDB Entry DOI: 10.7270/Q2B8571Q |
More data for this
Ligand-Target Pair | |
Transcriptional regulator ERG
(Homo sapiens (Human)) | BDBM251922

(US10065961, Compound 22 | US10683295, Compound 22 ...)Show SMILES C[C@H]1N(CCn2c1nnc2-c1nc(C)c(C)s1)C(=O)c1ccc(cc1)-c1cccs1 |r| Show InChI InChI=1S/C22H21N5OS2/c1-13-15(3)30-21(23-13)20-25-24-19-14(2)26(10-11-27(19)20)22(28)17-8-6-16(7-9-17)18-5-4-12-29-18/h4-9,12,14H,10-11H2,1-3H3/t14-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Ogeda SA.
US Patent
| Assay Description
The human Ether-a-go-go Related Gene (hERG) encodes the inward rectifying voltage gated potassium channel in the heart (IKr) which is involved in car... |
US Patent US10065961 (2018)
BindingDB Entry DOI: 10.7270/Q2MW2K4R |
More data for this
Ligand-Target Pair | |