 Found 8 hits in this display
Found 8 hits in this display Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Arginase-1
(Homo sapiens (Human)) | BDBM50509011

(CHEMBL4538713)Show SMILES N[C@H](CN1C[C@H](CCCB(O)O)[C@@](N)(C1)C(O)=O)Cc1ccccc1 |r| Show InChI InChI=1S/C17H28BN3O4/c19-15(9-13-5-2-1-3-6-13)11-21-10-14(7-4-8-18(24)25)17(20,12-21)16(22)23/h1-3,5-6,14-15,24-25H,4,7-12,19-20H2,(H,22,23)/t14-,15-,17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
New England Discovery Partners
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant arginase 1 expressed in Escherichia coli BL21 (DE3) assessed as reduction in urea production using L-arginine as subs... |
J Med Chem 62: 8164-8177 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00931
BindingDB Entry DOI: 10.7270/Q2X92FM9 |
More data for this
Ligand-Target Pair | |
Arginase-1
(Homo sapiens (Human)) | BDBM50509018
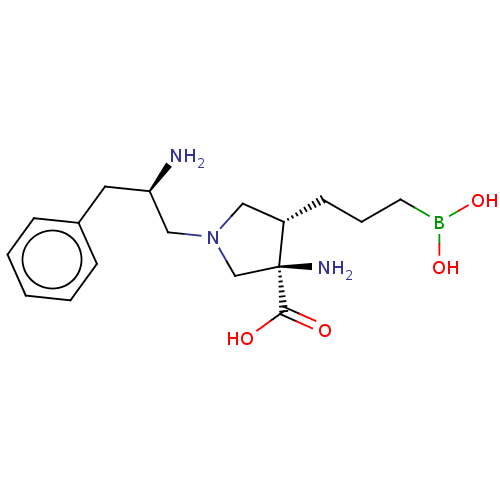
(CHEMBL4461464)Show SMILES N[C@@H](CN1C[C@H](CCCB(O)O)[C@@](N)(C1)C(O)=O)Cc1ccccc1 |r| Show InChI InChI=1S/C17H28BN3O4/c19-15(9-13-5-2-1-3-6-13)11-21-10-14(7-4-8-18(24)25)17(20,12-21)16(22)23/h1-3,5-6,14-15,24-25H,4,7-12,19-20H2,(H,22,23)/t14-,15+,17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
New England Discovery Partners
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant arginase 1 expressed in Escherichia coli BL21 (DE3) assessed as reduction in urea production using L-arginine as subs... |
J Med Chem 62: 8164-8177 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00931
BindingDB Entry DOI: 10.7270/Q2X92FM9 |
More data for this
Ligand-Target Pair | |
Arginase-1
(Homo sapiens (Human)) | BDBM290421
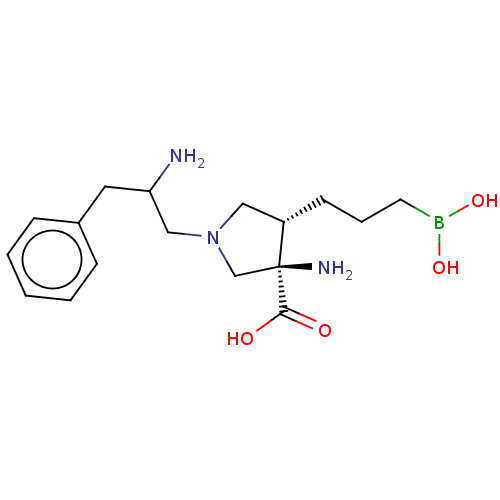
((3R,4S)-3-amino-1-(2-amino-3-phenylpropyl)-4-(3- b...)Show SMILES NC(CN1C[C@H](CCCB(O)O)[C@@](N)(C1)C(O)=O)Cc1ccccc1 |r| Show InChI InChI=1S/C17H28BN3O4/c19-15(9-13-5-2-1-3-6-13)11-21-10-14(7-4-8-18(24)25)17(20,12-21)16(22)23/h1-3,5-6,14-15,24-25H,4,7-12,19-20H2,(H,22,23)/t14-,15?,17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 125 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of arginase I (ARG I) and arginase II (ARG II) by Formula I or Formula II compounds is followed spectrophotometrically at 530 nm. The comp... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q289193N |
More data for this
Ligand-Target Pair | |
Arginase-2, mitochondrial
(Homo sapiens (Human)) | BDBM290421

((3R,4S)-3-amino-1-(2-amino-3-phenylpropyl)-4-(3- b...)Show SMILES NC(CN1C[C@H](CCCB(O)O)[C@@](N)(C1)C(O)=O)Cc1ccccc1 |r| Show InChI InChI=1S/C17H28BN3O4/c19-15(9-13-5-2-1-3-6-13)11-21-10-14(7-4-8-18(24)25)17(20,12-21)16(22)23/h1-3,5-6,14-15,24-25H,4,7-12,19-20H2,(H,22,23)/t14-,15?,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 125 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q289193N |
More data for this
Ligand-Target Pair | |
Arginase-2, mitochondrial
(Homo sapiens (Human)) | BDBM290421

((3R,4S)-3-amino-1-(2-amino-3-phenylpropyl)-4-(3- b...)Show SMILES NC(CN1C[C@H](CCCB(O)O)[C@@](N)(C1)C(O)=O)Cc1ccccc1 |r| Show InChI InChI=1S/C17H28BN3O4/c19-15(9-13-5-2-1-3-6-13)11-21-10-14(7-4-8-18(24)25)17(20,12-21)16(22)23/h1-3,5-6,14-15,24-25H,4,7-12,19-20H2,(H,22,23)/t14-,15?,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <250 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Mars, Incorporated
US Patent
| Assay Description
Inhibition of arginase I (ARG I) and arginase II (ARG II) by Formula I or Formula II compounds is followed spectrophotometrically at 530 nm. The comp... |
US Patent US10098902 (2018)
BindingDB Entry DOI: 10.7270/Q2319XXK |
More data for this
Ligand-Target Pair | |
Arginase-1
(Homo sapiens (Human)) | BDBM290421

((3R,4S)-3-amino-1-(2-amino-3-phenylpropyl)-4-(3- b...)Show SMILES NC(CN1C[C@H](CCCB(O)O)[C@@](N)(C1)C(O)=O)Cc1ccccc1 |r| Show InChI InChI=1S/C17H28BN3O4/c19-15(9-13-5-2-1-3-6-13)11-21-10-14(7-4-8-18(24)25)17(20,12-21)16(22)23/h1-3,5-6,14-15,24-25H,4,7-12,19-20H2,(H,22,23)/t14-,15?,17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <250 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Mars, Incorporated
US Patent
| Assay Description
Inhibition of arginase I (ARG I) and arginase II (ARG II) by Formula I or Formula II compounds is followed spectrophotometrically at 530 nm. The comp... |
US Patent US10098902 (2018)
BindingDB Entry DOI: 10.7270/Q2319XXK |
More data for this
Ligand-Target Pair | |
Arginase-1
(Homo sapiens (Human)) | BDBM290421

((3R,4S)-3-amino-1-(2-amino-3-phenylpropyl)-4-(3- b...)Show SMILES NC(CN1C[C@H](CCCB(O)O)[C@@](N)(C1)C(O)=O)Cc1ccccc1 |r| Show InChI InChI=1S/C17H28BN3O4/c19-15(9-13-5-2-1-3-6-13)11-21-10-14(7-4-8-18(24)25)17(20,12-21)16(22)23/h1-3,5-6,14-15,24-25H,4,7-12,19-20H2,(H,22,23)/t14-,15?,17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Mars, Incorporated
US Patent
| Assay Description
Inhibition of arginase I (ARG I) and arginase II (ARG II) by Formula I or Formula II compounds is followed spectrophotometrically at 530 nm. The comp... |
US Patent US10603330 (2020)
BindingDB Entry DOI: 10.7270/Q2WD43K6 |
More data for this
Ligand-Target Pair | |
Arginase-2, mitochondrial
(Homo sapiens (Human)) | BDBM290421

((3R,4S)-3-amino-1-(2-amino-3-phenylpropyl)-4-(3- b...)Show SMILES NC(CN1C[C@H](CCCB(O)O)[C@@](N)(C1)C(O)=O)Cc1ccccc1 |r| Show InChI InChI=1S/C17H28BN3O4/c19-15(9-13-5-2-1-3-6-13)11-21-10-14(7-4-8-18(24)25)17(20,12-21)16(22)23/h1-3,5-6,14-15,24-25H,4,7-12,19-20H2,(H,22,23)/t14-,15?,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Mars, Incorporated
US Patent
| Assay Description
Inhibition of arginase I (ARG I) and arginase II (ARG II) by Formula I or Formula II compounds is followed spectrophotometrically at 530 nm. The comp... |
US Patent US10603330 (2020)
BindingDB Entry DOI: 10.7270/Q2WD43K6 |
More data for this
Ligand-Target Pair | |