 Found 19 hits of ic50 for monomerid = 17289
Found 19 hits of ic50 for monomerid = 17289 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Estrogen receptor
(Homo sapiens (Human)) | BDBM17289
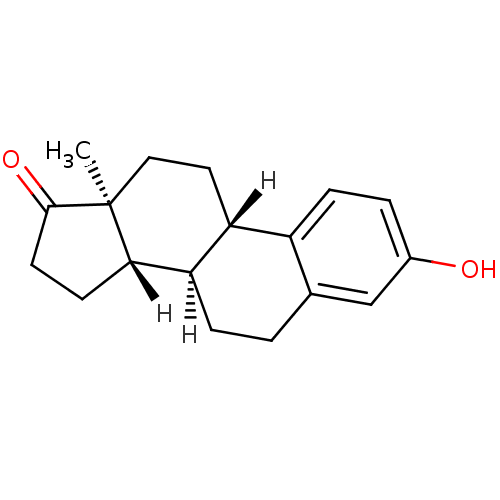
((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 29 | n/a | 166 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
| Assay Description
Ligand binding was determined using a scintillation proximity assay with streptavidin-coated SPA beads (Amersham) and biotinylated receptor. Receptor... |
J Med Chem 49: 2440-55 (2006)
Article DOI: 10.1021/jm0509389
BindingDB Entry DOI: 10.7270/Q2WH2N82 |
More data for this
Ligand-Target Pair | |
Estrogen receptor beta
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 31 | n/a | 26 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
| Assay Description
Ligand binding was determined using a scintillation proximity assay with streptavidin-coated SPA beads (Amersham) and biotinylated receptor. Receptor... |
J Med Chem 49: 2440-55 (2006)
Article DOI: 10.1021/jm0509389
BindingDB Entry DOI: 10.7270/Q2WH2N82 |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 96 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Experimental Genetics
Curated by ChEMBL
| Assay Description
Displacement of [3H]17beta-estradiol from human ERalpha expressed in SF9 cells |
Bioorg Med Chem Lett 19: 6740-4 (2009)
Article DOI: 10.1016/j.bmcl.2009.09.113
BindingDB Entry DOI: 10.7270/Q20K29HP |
More data for this
Ligand-Target Pair | |
17-beta-hydroxysteroid dehydrogenase type 1
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 109 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Experimental Genetics
Curated by ChEMBL
| Assay Description
Inhibition of His-tagged human 17beta-HSD1 expressed in Escherichia coli by scintillation counting |
Bioorg Med Chem Lett 19: 6740-4 (2009)
Article DOI: 10.1016/j.bmcl.2009.09.113
BindingDB Entry DOI: 10.7270/Q20K29HP |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Estrogen receptor
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity towards estrogen receptor alpha by [3H]17-beta-estradiol displacement. |
Bioorg Med Chem Lett 14: 2327-30 (2004)
Article DOI: 10.1016/j.bmcl.2004.01.099
BindingDB Entry DOI: 10.7270/Q2HT2PVS |
More data for this
Ligand-Target Pair | |
17-beta-hydroxysteroid dehydrogenase type 1
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 218 | n/a | n/a | n/a | n/a | n/a | n/a |
CHUQ-Pavillon CHUL and Universit£ Laval
Curated by ChEMBL
| Assay Description
Inhibition of 17beta-HSD1 in human T47D cells assessed as inhibition of transformation of [14C]-estrone into [14C]estrogen |
Bioorg Med Chem 16: 1849-60 (2008)
Article DOI: 10.1016/j.bmc.2007.11.007
BindingDB Entry DOI: 10.7270/Q2KP8302 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
17-beta-hydroxysteroid dehydrogenase type 1
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 330 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bath
Curated by ChEMBL
| Assay Description
Inhibition of 17-beta HSD1 in T47D cells |
J Med Chem 49: 1325-45 (2006)
Article DOI: 10.1021/jm050830t
BindingDB Entry DOI: 10.7270/Q2C24X7J |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Estrogen receptor
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 480 | n/a | 0.700 | n/a | n/a | 7.4 | 22 |
Novartis Pharmaceuticals
| Assay Description
Radioligand binding assay was performed by using 96-well microtiterplates containing ER, 17beta-estradiol, and the test compound to be tested and SPA... |
J Med Chem 45: 1399-401 (2002)
Article DOI: 10.1021/jm015577l
BindingDB Entry DOI: 10.7270/Q27M0665 |
More data for this
Ligand-Target Pair | |
Estrogen receptor beta
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 540 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity towards estrogen receptor beta by [3H]17-beta-estradiol displacement. |
Bioorg Med Chem Lett 14: 2327-30 (2004)
Article DOI: 10.1016/j.bmcl.2004.01.099
BindingDB Entry DOI: 10.7270/Q2HT2PVS |
More data for this
Ligand-Target Pair | |
17-beta-hydroxysteroid dehydrogenase type 1
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 600 | n/a | n/a | n/a | n/a | n/a | n/a |
CHUQ - Pavillon CHUL and Université Laval
Curated by ChEMBL
| Assay Description
Inhibition of (unknown origin) 17beta-HSD1 expressed in HEK 293 cells assessed as conversion of [14C]estrone to [14C]estradiol |
Eur J Med Chem 43: 2298-306 (2008)
Article DOI: 10.1016/j.ejmech.2008.01.044
BindingDB Entry DOI: 10.7270/Q2P84BP6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
17-beta-hydroxysteroid dehydrogenase type 1
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 810 | n/a | n/a | n/a | n/a | n/a | n/a |
CHUQ - Pavillon CHUL and Université Laval
Curated by ChEMBL
| Assay Description
Inhibition of (unknown origin) 17beta-HSD1 expressed in HEK 293 cells assessed as conversion of [14C]estrone to [14C]estradiol using NADH |
Eur J Med Chem 43: 2298-306 (2008)
Article DOI: 10.1016/j.ejmech.2008.01.044
BindingDB Entry DOI: 10.7270/Q2P84BP6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Estrogen receptor beta
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.14E+3 | n/a | 2.10 | n/a | n/a | 7.4 | 22 |
Novartis Pharmaceuticals
| Assay Description
Radioligand binding assay was performed by using 96-well microtiterplates containing ER, 17beta-estradiol, and the test compound to be tested and SPA... |
J Med Chem 45: 1399-401 (2002)
Article DOI: 10.1021/jm015577l
BindingDB Entry DOI: 10.7270/Q27M0665 |
More data for this
Ligand-Target Pair | |
Broad substrate specificity ATP-binding cassette transporter ABCG2
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Japanese Foundation for Cancer Research
Curated by ChEMBL
| Assay Description
TP_TRANSPORTER: drug resistance(Mitoxantrone) in BCRP-expressing K562 cells |
Mol Cancer Ther 2: 105-12 (2003)
BindingDB Entry DOI: 10.7270/Q2J104FD |
More data for this
Ligand-Target Pair | |
Broad substrate specificity ATP-binding cassette transporter ABCG2
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Japanese Foundation for Cancer Research
Curated by ChEMBL
| Assay Description
TP_TRANSPORTER: drug resistance(SN-38) in BCRP-expressing K562 cells |
Mol Cancer Ther 2: 105-12 (2003)
BindingDB Entry DOI: 10.7270/Q2J104FD |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1B1
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
CHU de Qu£bec - Research Center (CHUL
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human CYP1B1 expressed in bacterial microsomes co-expressing P450 reductase using 7-ethyl-O-resorufin as substrate after 45... |
Bioorg Med Chem Lett 26: 5272-5276 (2016)
Article DOI: 10.1016/j.bmcl.2016.09.046
BindingDB Entry DOI: 10.7270/Q2V98B2J |
More data for this
Ligand-Target Pair | |
Steryl-sulfatase
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of human steroid sulfatase using 4-methylumbelliferyl sulfate substrate after 10 mins by fluorescence assay |
Bioorg Med Chem 19: 5999-6005 (2011)
Article DOI: 10.1016/j.bmc.2011.08.046
BindingDB Entry DOI: 10.7270/Q2FX79VJ |
More data for this
Ligand-Target Pair | |
Alpha-synuclein
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9.31E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of alpha-synuclein aggregation (unknown origin) incubated for 8 days by thioflavin S based fluorescence assay |
Citation and Details
Article DOI: 10.1016/j.ejmech.2019.01.045
BindingDB Entry DOI: 10.7270/Q2H998V9 |
More data for this
Ligand-Target Pair | |
Ghrelin O-acyltransferase
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | 7.0 | 25 |
Syracuse University
| Assay Description
Assays were performed with ~100 μg of membrane protein, as determined by a Bradford assay. The membrane fraction was preincubated with 1 μM met... |
Biochemistry 56: 919-931 (2017)
Article DOI: 10.1021/acs.biochem.6b01008
BindingDB Entry DOI: 10.7270/Q270808D |
More data for this
Ligand-Target Pair | |
Bile salt export pump
(Homo sapiens (Human)) | BDBM17289

((1S,10R,11S,15S)-5-hydroxy-15-methyltetracyclo[8.7...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@]1([H])c3ccc(O)cc3CC[C@@]21[H] Show InChI InChI=1S/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14-,15-,16+,18+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of [3H]haloperidol binding for Dopamine receptor D2 in rat striatal membranes. |
Drug Metab Dispos 40: 2332-41 (2012)
Article DOI: 10.1124/dmd.112.047068
BindingDB Entry DOI: 10.7270/Q2ZP488M |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data