

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50116878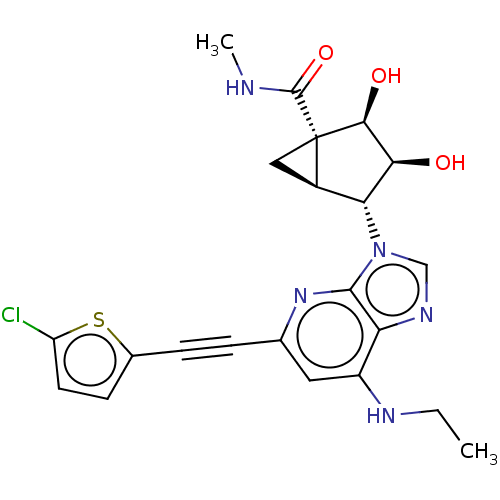 (CHEMBL3612940) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Displacement of [125I]N6-(4-amino-3-iodobenzyl)adenosine-5'-N-methyluronamide from human adenosine A3 receptor expressed in CHO cell membranes | ACS Med Chem Lett 6: 804-8 (2015) Article DOI: 10.1021/acsmedchemlett.5b00150 BindingDB Entry DOI: 10.7270/Q23R0VPF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Mus musculus) | BDBM50116878 (CHEMBL3612940) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Binding affinity to mouse adenosine A3 receptor expressed in HEK293 cell membranes | ACS Med Chem Lett 6: 804-8 (2015) Article DOI: 10.1021/acsmedchemlett.5b00150 BindingDB Entry DOI: 10.7270/Q23R0VPF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Mus musculus) | BDBM50116878 (CHEMBL3612940) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Concentration required to inhibit binding of ICAM-1 to LFA-1 (Leukocyte function-associated antigen-1), evaluated ELISA | J Med Chem 60: 3109-3123 (2017) Article DOI: 10.1021/acs.jmedchem.7b00141 BindingDB Entry DOI: 10.7270/Q2Z60RBZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Canis lupus familiaris) | BDBM50116878 (CHEMBL3612940) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 49 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Binding affinity to canine adenosine A3 receptor expressed in HEK293 cell membranes | ACS Med Chem Lett 6: 804-8 (2015) Article DOI: 10.1021/acsmedchemlett.5b00150 BindingDB Entry DOI: 10.7270/Q23R0VPF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50116878 (CHEMBL3612940) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Agonist activity at human adenosine A3 receptor expressed in CHO cells assessed as inhibition of cAMP production | ACS Med Chem Lett 6: 804-8 (2015) Article DOI: 10.1021/acsmedchemlett.5b00150 BindingDB Entry DOI: 10.7270/Q23R0VPF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||