

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aurora kinase A (Homo sapiens (Human)) | BDBM50337135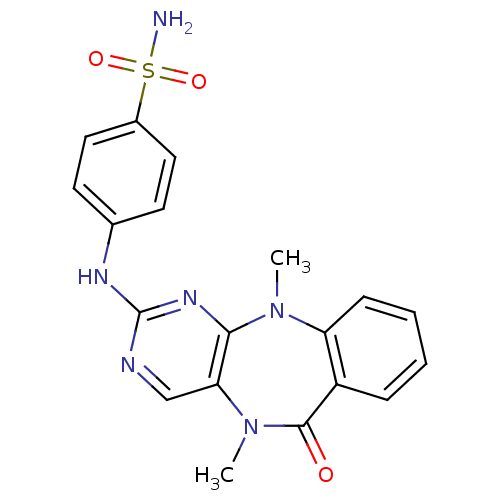 (4-((5,11-dimethyl-6-oxo-6,11-dihydro-5H-benzo[e]py...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School | Assay Description In vitro kinase assay of Aurora A, B, and C using Z-LYTE technology (Invitrogen) and ATP at Km apparent for each kinase. | ACS Chem Biol 7: 185-96 (2012) Article DOI: 10.1021/cb200305u BindingDB Entry DOI: 10.7270/Q2PV6HZG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aurora kinase A (Homo sapiens (Human)) | BDBM50337135 (4-((5,11-dimethyl-6-oxo-6,11-dihydro-5H-benzo[e]py...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute Curated by ChEMBL | Assay Description Inhibition of human Aurora kinase A (E122 to K401 residues) expressed in mammalian expression system by Z'LYTE assay | ACS Med Chem Lett 7: 908-912 (2016) Article DOI: 10.1021/acsmedchemlett.6b00209 BindingDB Entry DOI: 10.7270/Q27P91B0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aurora kinase A (Homo sapiens (Human)) | BDBM50337135 (4-((5,11-dimethyl-6-oxo-6,11-dihydro-5H-benzo[e]py...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 4.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description The enzymatic activities against PI3K-α, PI3K-β, PI3K-γ and PI3K-δ were tested in ADAPTA assays. Activity against AURKB and AURKB... | Citation and Details BindingDB Entry DOI: 10.7270/Q20868G7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Leucine-rich repeat serine/threonine-protein kinase 2 (Homo sapiens (Human)) | BDBM50337135 (4-((5,11-dimethyl-6-oxo-6,11-dihydro-5H-benzo[e]py...) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Inhibition of recombinant human GST-tagged LRRK2 catalytic domain (970 to 2527 residues) expressed in baculovirus expression system using LRRKtide as... | J Med Chem 63: 7817-7826 (2020) Article DOI: 10.1021/acs.jmedchem.0c00596 BindingDB Entry DOI: 10.7270/Q2SB4981 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aurora kinase B (Homo sapiens (Human)) | BDBM50337135 (4-((5,11-dimethyl-6-oxo-6,11-dihydro-5H-benzo[e]py...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 8.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School | Assay Description In vitro kinase assay of Aurora A, B, and C using Z-LYTE technology (Invitrogen) and ATP at Km apparent for each kinase. | ACS Chem Biol 7: 185-96 (2012) Article DOI: 10.1021/cb200305u BindingDB Entry DOI: 10.7270/Q2PV6HZG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aurora kinase B (Homo sapiens (Human)) | BDBM50337135 (4-((5,11-dimethyl-6-oxo-6,11-dihydro-5H-benzo[e]py...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute Curated by ChEMBL | Assay Description Inhibition of human Aurora kinase B (D25 to A303 residues) expressed in mammalian expression system by Z'LYTE assay | ACS Med Chem Lett 7: 908-912 (2016) Article DOI: 10.1021/acsmedchemlett.6b00209 BindingDB Entry DOI: 10.7270/Q27P91B0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aurora kinase B (Homo sapiens (Human)) | BDBM50337135 (4-((5,11-dimethyl-6-oxo-6,11-dihydro-5H-benzo[e]py...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 13.3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description The enzymatic activities against PI3K-α, PI3K-β, PI3K-γ and PI3K-δ were tested in ADAPTA assays. Activity against AURKB and AURKB... | Citation and Details BindingDB Entry DOI: 10.7270/Q20868G7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform (Homo sapiens (Human)) | BDBM50337135 (4-((5,11-dimethyl-6-oxo-6,11-dihydro-5H-benzo[e]py...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 14.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description The enzymatic activities against PI3K-α, PI3K-β, PI3K-γ and PI3K-δ were tested in ADAPTA assays. Activity against AURKB and AURKB... | Citation and Details BindingDB Entry DOI: 10.7270/Q20868G7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform (Homo sapiens (Human)) | BDBM50337135 (4-((5,11-dimethyl-6-oxo-6,11-dihydro-5H-benzo[e]py...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute Curated by ChEMBL | Assay Description Inhibition of human PI3Kgamma (S144 to A1102 residues) expressed in mammalian expression system incubated for 60 mins by ADAPTA assay | ACS Med Chem Lett 7: 908-912 (2016) Article DOI: 10.1021/acsmedchemlett.6b00209 BindingDB Entry DOI: 10.7270/Q27P91B0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Phosphatidylinositol 3-kinase C2 domain-containing subunit gamma (Homo sapiens (Human)) | BDBM50337135 (4-((5,11-dimethyl-6-oxo-6,11-dihydro-5H-benzo[e]py...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 49 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description The enzymatic activities against PI3K-α, PI3K-β, PI3K-γ and PI3K-δ were tested in ADAPTA assays. Activity against AURKB and AURKB... | Citation and Details BindingDB Entry DOI: 10.7270/Q20868G7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform (Homo sapiens (Human)) | BDBM50337135 (4-((5,11-dimethyl-6-oxo-6,11-dihydro-5H-benzo[e]py...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 49 | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute Curated by ChEMBL | Assay Description Inhibition of human PI3Kdelta (R108 to Q1044 residues) expressed in mammalian expression system incubated for 60 mins by ADAPTA assay | ACS Med Chem Lett 7: 908-912 (2016) Article DOI: 10.1021/acsmedchemlett.6b00209 BindingDB Entry DOI: 10.7270/Q27P91B0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine/threonine-protein kinase DCLK1 (Homo sapiens (Human)) | BDBM50337135 (4-((5,11-dimethyl-6-oxo-6,11-dihydro-5H-benzo[e]py...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 187 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal His6-tagged DCLK1 (G351 to H689 residues) expressed in Escherichia coli BL21 DE3 using 5-FAM-KKLRRTLSVA-CO... | J Med Chem 63: 7817-7826 (2020) Article DOI: 10.1021/acs.jmedchem.0c00596 BindingDB Entry DOI: 10.7270/Q2SB4981 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 7 (Homo sapiens (Human)) | BDBM50337135 (4-((5,11-dimethyl-6-oxo-6,11-dihydro-5H-benzo[e]py...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 310 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of EGF-induced BMK1 autophosphorylation in human HeLa cells by SDS-PAGE analysis | ACS Med Chem Lett 2: 195-200 (2011) Article DOI: 10.1021/ml100304b BindingDB Entry DOI: 10.7270/Q2222VSB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||