

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50194649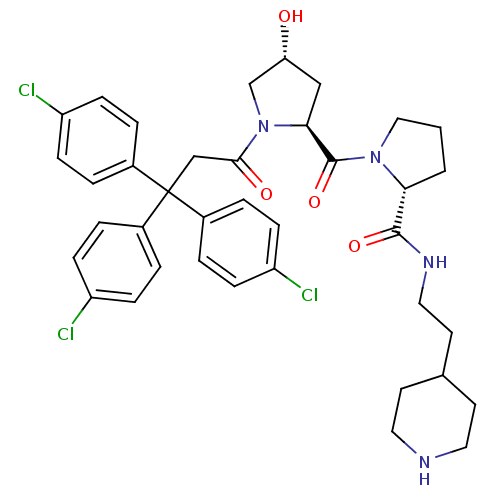 ((2R)-1-((2S,4R)-4-hydroxy-1-[3,3,3-tris(4-chloroph...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute Curated by ChEMBL | Assay Description Inhibition of [3H]NMS binding to human cloned M3 receptor expressed in CHO cells | J Med Chem 49: 5653-63 (2006) Article DOI: 10.1021/jm051205r BindingDB Entry DOI: 10.7270/Q2F76C6K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M4 (Homo sapiens (Human)) | BDBM50194649 ((2R)-1-((2S,4R)-4-hydroxy-1-[3,3,3-tris(4-chloroph...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute Curated by ChEMBL | Assay Description Inhibition of [3H]NMS binding to human cloned M4 receptor expressed in CHO cells | J Med Chem 49: 5653-63 (2006) Article DOI: 10.1021/jm051205r BindingDB Entry DOI: 10.7270/Q2F76C6K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM50194649 ((2R)-1-((2S,4R)-4-hydroxy-1-[3,3,3-tris(4-chloroph...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 94 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute Curated by ChEMBL | Assay Description Inhibition of [3H]NMS binding to human cloned M2 receptor expressed in CHO cells | J Med Chem 49: 5653-63 (2006) Article DOI: 10.1021/jm051205r BindingDB Entry DOI: 10.7270/Q2F76C6K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50194649 ((2R)-1-((2S,4R)-4-hydroxy-1-[3,3,3-tris(4-chloroph...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute Curated by ChEMBL | Assay Description Inhibition of [3H]NMS binding to human cloned M1 receptor expressed in CHO cells | J Med Chem 49: 5653-63 (2006) Article DOI: 10.1021/jm051205r BindingDB Entry DOI: 10.7270/Q2F76C6K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M5 (Homo sapiens (Human)) | BDBM50194649 ((2R)-1-((2S,4R)-4-hydroxy-1-[3,3,3-tris(4-chloroph...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 950 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute Curated by ChEMBL | Assay Description Inhibition of [3H]NMS binding to human cloned M5 receptor expressed in CHO cells | J Med Chem 49: 5653-63 (2006) Article DOI: 10.1021/jm051205r BindingDB Entry DOI: 10.7270/Q2F76C6K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||