

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50032163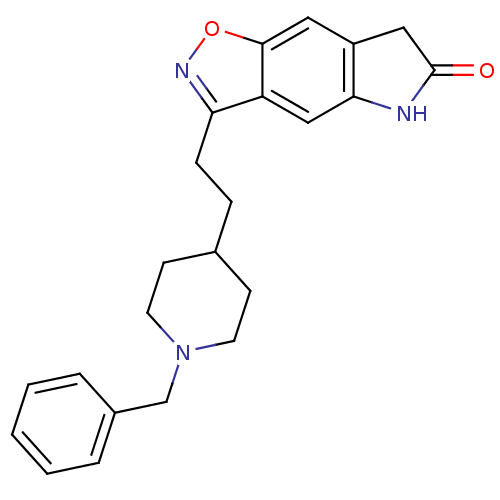 (3-(2-(1-benzylpiperidin-4-yl)ethyl)-5H-isoxazolo[5...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 0.950 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc Curated by ChEMBL | Assay Description In vitro inhibition of Acetylcholinesterase from human erythrocytes | J Med Chem 38: 2802-8 (1995) BindingDB Entry DOI: 10.7270/Q2Q52NN0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50032163 (3-(2-(1-benzylpiperidin-4-yl)ethyl)-5H-isoxazolo[5...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.950 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Kansas Curated by ChEMBL | Assay Description Inhibitory activity against Acetylcholinesterase enzyme using human AChE assay | J Med Chem 47: 5492-500 (2004) Article DOI: 10.1021/jm049695v BindingDB Entry DOI: 10.7270/Q2765G3W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50032163 (3-(2-(1-benzylpiperidin-4-yl)ethyl)-5H-isoxazolo[5...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.950 | n/a | n/a | n/a | n/a | n/a | n/a |
Sichuan University Curated by ChEMBL | Assay Description Inhibition of AChE | Eur J Med Chem 45: 1167-72 (2010) Article DOI: 10.1016/j.ejmech.2009.12.038 BindingDB Entry DOI: 10.7270/Q25H7GFM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50032163 (3-(2-(1-benzylpiperidin-4-yl)ethyl)-5H-isoxazolo[5...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.955 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade Federal do Rio de Janeiro (UFRJ) Curated by ChEMBL | Assay Description Inhibition of human acetylcholinesterase | Eur J Med Chem 46: 39-51 (2010) Article DOI: 10.1016/j.ejmech.2010.10.009 BindingDB Entry DOI: 10.7270/Q2SX6FG0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||