 Found 2 hits Enz. Inhib. hit(s) with Target = 'Beta-2 adrenergic receptor' and Ligand = 'BDBM221902'
Found 2 hits Enz. Inhib. hit(s) with Target = 'Beta-2 adrenergic receptor' and Ligand = 'BDBM221902' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM221902
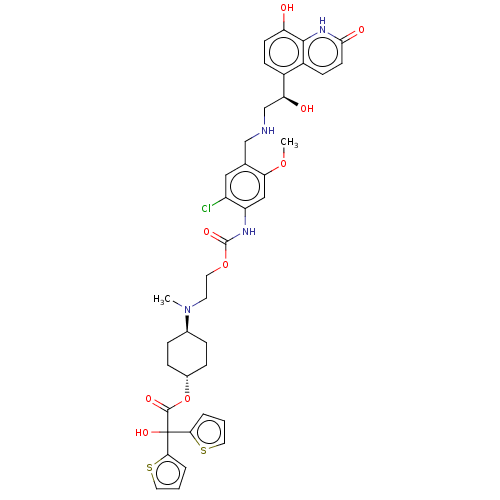
(US9315463, 12)Show SMILES COc1cc(NC(=O)OCCN(C)[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:41.45,13.12,wD:16.19,(-5.85,-3.32,;-4.52,-2.55,;-4.52,-1.01,;-3.19,-.24,;-3.19,1.3,;-1.85,2.07,;-.52,1.3,;-.52,-.24,;.82,2.07,;2.15,1.3,;3.48,2.07,;4.82,1.3,;4.82,-.24,;6.15,2.07,;6.15,3.61,;7.48,4.38,;8.82,3.61,;8.82,2.07,;7.48,1.3,;10.15,4.38,;11.49,3.61,;11.49,2.07,;12.82,4.38,;12.05,5.72,;13.59,3.05,;15.13,3.05,;15.6,1.58,;14.36,.68,;13.11,1.58,;14.15,5.15,;14.15,6.69,;15.62,7.17,;16.52,5.92,;15.62,4.68,;-4.52,2.07,;-4.52,3.61,;-5.85,1.3,;-5.85,-.24,;-7.19,-1.01,;-8.52,-.24,;-9.85,-1.01,;-11.19,-.24,;-11.19,1.3,;-12.52,-1.01,;-13.85,-.24,;-15.19,-1.01,;-15.19,-2.55,;-16.52,-3.32,;-13.85,-3.32,;-13.85,-4.86,;-12.52,-5.63,;-12.52,-7.17,;-11.19,-4.86,;-11.19,-3.32,;-12.52,-2.55,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | 25 |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human adrenergic beta1 and beta2 receptors was performed using commercial membranes prepared from Sf9 cells where they are ov... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM221902

(US9315463, 12)Show SMILES COc1cc(NC(=O)OCCN(C)[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:41.45,13.12,wD:16.19,(-5.85,-3.32,;-4.52,-2.55,;-4.52,-1.01,;-3.19,-.24,;-3.19,1.3,;-1.85,2.07,;-.52,1.3,;-.52,-.24,;.82,2.07,;2.15,1.3,;3.48,2.07,;4.82,1.3,;4.82,-.24,;6.15,2.07,;6.15,3.61,;7.48,4.38,;8.82,3.61,;8.82,2.07,;7.48,1.3,;10.15,4.38,;11.49,3.61,;11.49,2.07,;12.82,4.38,;12.05,5.72,;13.59,3.05,;15.13,3.05,;15.6,1.58,;14.36,.68,;13.11,1.58,;14.15,5.15,;14.15,6.69,;15.62,7.17,;16.52,5.92,;15.62,4.68,;-4.52,2.07,;-4.52,3.61,;-5.85,1.3,;-5.85,-.24,;-7.19,-1.01,;-8.52,-.24,;-9.85,-1.01,;-11.19,-.24,;-11.19,1.3,;-12.52,-1.01,;-13.85,-.24,;-15.19,-1.01,;-15.19,-2.55,;-16.52,-3.32,;-13.85,-3.32,;-13.85,-4.86,;-12.52,-5.63,;-12.52,-7.17,;-11.19,-4.86,;-11.19,-3.32,;-12.52,-2.55,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.251 | n/a | n/a | n/a | n/a |
School of Pharmaceutical Sciences & The Fifth Affiliated Hospital
Curated by ChEMBL
| Assay Description
Agonist activity at human beta2 adrenoceptor |
J Med Chem 62: 5944-5978 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01520
BindingDB Entry DOI: 10.7270/Q2GT5RNR |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

