

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cytochrome P450 2C9 (Homo sapiens (Human)) | BDBM50333117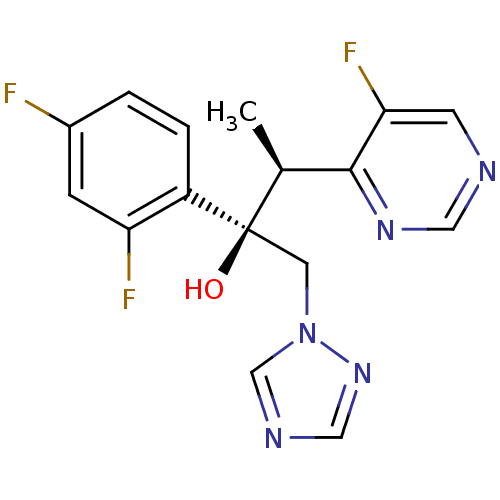 ((2R,3S)-2,3-bis(2,4-difluorophenyl)-1-(1H-1,2,4-tr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents | DrugBank Article PubMed | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University Curated by ChEMBL | Assay Description Inhibition of CYP2C9 in human liver microsomes assessed as tolbutamide 4-methylhydroxylation after 60 mins by Dixon plot analysis | Antimicrob Agents Chemother 53: 541-51 (2009) Article DOI: 10.1128/AAC.01123-08 BindingDB Entry DOI: 10.7270/Q2H13285 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C9 (Homo sapiens (Human)) | BDBM50333117 ((2R,3S)-2,3-bis(2,4-difluorophenyl)-1-(1H-1,2,4-tr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents | DrugBank Article PubMed | n/a | n/a | 3.62E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University Curated by ChEMBL | Assay Description Inhibition of CYP2C9 in human liver microsomes assessed as tolbutamide 4'-hydroxylation after 60 mins | Antimicrob Agents Chemother 53: 541-51 (2009) Article DOI: 10.1128/AAC.01123-08 BindingDB Entry DOI: 10.7270/Q2H13285 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C9 (Homo sapiens (Human)) | BDBM50333117 ((2R,3S)-2,3-bis(2,4-difluorophenyl)-1-(1H-1,2,4-tr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents | DrugBank Article PubMed | n/a | n/a | 8.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University Curated by ChEMBL | Assay Description Inhibition of CYP2C9 using tolbutamide as probe | Antimicrob Agents Chemother 53: 541-51 (2009) Article DOI: 10.1128/AAC.01123-08 BindingDB Entry DOI: 10.7270/Q2H13285 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C9 (Homo sapiens (Human)) | BDBM50333117 ((2R,3S)-2,3-bis(2,4-difluorophenyl)-1-(1H-1,2,4-tr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents | DrugBank US Patent | n/a | n/a | 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Pooled human liver microsome suspension (20 mg/mL) was diluted with phosphate buffer to obtain a 5 mg/mL suspension. A solution of NADPH was prepared... | Citation and Details BindingDB Entry DOI: 10.7270/Q2F76GSF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C9 (Homo sapiens (Human)) | BDBM50333117 ((2R,3S)-2,3-bis(2,4-difluorophenyl)-1-(1H-1,2,4-tr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents | DrugBank US Patent | n/a | n/a | 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Viamet Pharmaceuticals, Inc. US Patent | Assay Description Solutions of each test compound were separately prepared at concentrations of 20000, 6000, 2000, 600, 200, and 60 μM by serial dilution with DMS... | US Patent US8883797 (2014) BindingDB Entry DOI: 10.7270/Q2FT8JRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C9 (Homo sapiens (Human)) | BDBM50333117 ((2R,3S)-2,3-bis(2,4-difluorophenyl)-1-(1H-1,2,4-tr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents | DrugBank US Patent | n/a | n/a | 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Viamet Pharmaceuticals, Inc. US Patent | Assay Description Solutions of each test compound were separately prepared at concentrations of 20000, 6000, 2000, 600, 200, and 60 uM by serial dilution with DMSO:M... | US Patent US9221791 (2015) BindingDB Entry DOI: 10.7270/Q24M93B6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||