

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50240612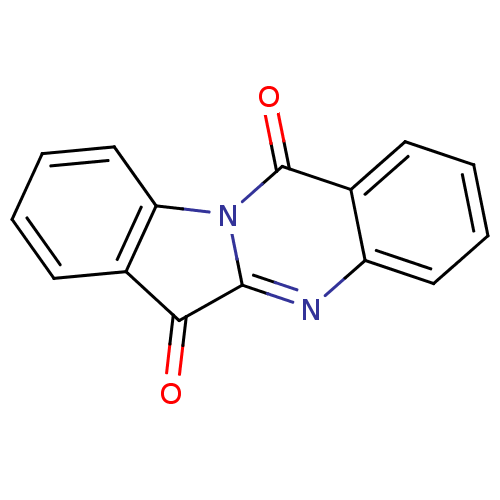 (CHEMBL306946 | GNF-PF-2691 | Indolo[2,1-b]quinazol...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 4.81E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University Curated by ChEMBL | Assay Description Mixed competitive inhibition of human recombinant IDO1 using L-tryptophan as substrate | J Med Chem 56: 8321-31 (2013) Article DOI: 10.1021/jm401195n BindingDB Entry DOI: 10.7270/Q2X34ZWS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50240612 (CHEMBL306946 | GNF-PF-2691 | Indolo[2,1-b]quinazol...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 54 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University Curated by ChEMBL | Assay Description Inhibition of human IDO1 expressed in HEK293 cells assessed as kynurenine release after 5 hrs by spectrophotometry | J Med Chem 56: 8321-31 (2013) Article DOI: 10.1021/jm401195n BindingDB Entry DOI: 10.7270/Q2X34ZWS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50240612 (CHEMBL306946 | GNF-PF-2691 | Indolo[2,1-b]quinazol...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem Patents Similars | US Patent | n/a | n/a | 5.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Peking University US Patent | Assay Description Experimental method: IDO-1 can oxidatively cleave the indole ring of tryptophan to form N-formylkynurenine. Referring to the method in the literature... | US Patent US10669273 (2020) BindingDB Entry DOI: 10.7270/Q2GQ71SK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50240612 (CHEMBL306946 | GNF-PF-2691 | Indolo[2,1-b]quinazol...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 7.15E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University Curated by ChEMBL | Assay Description Mixed competitive inhibition of human recombinant IDO1 using L-tryptophan as substrate | J Med Chem 56: 8321-31 (2013) Article DOI: 10.1021/jm401195n BindingDB Entry DOI: 10.7270/Q2X34ZWS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50240612 (CHEMBL306946 | GNF-PF-2691 | Indolo[2,1-b]quinazol...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.99E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Indo-Soviet Friendship College of Pharmacy (ISFCP) Curated by ChEMBL | Assay Description Inhibition of human cytomegalovirus DNA polymerase (95 uL) activity in a solution containing 6.4 mM HEPES (pH 7.5), incubation for 12 minutes at 26 d... | Bioorg Med Chem 25: 4533-4552 (2017) Article DOI: 10.1016/j.bmc.2017.07.003 BindingDB Entry DOI: 10.7270/Q2125W42 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||