 Found 6 hits Enz. Inhib. hit(s) with Target = 'Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform' and Ligand = 'BDBM50395821'
Found 6 hits Enz. Inhib. hit(s) with Target = 'Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform' and Ligand = 'BDBM50395821' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50395821
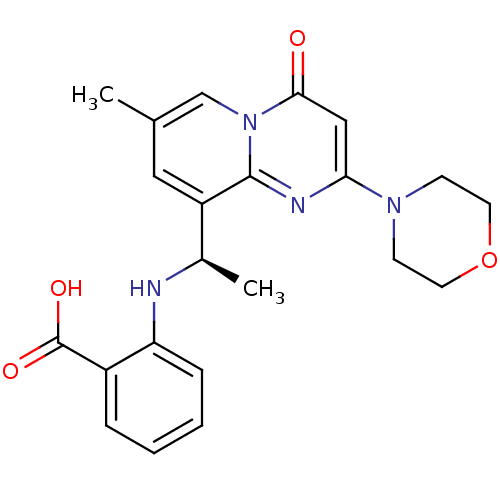
(CHEMBL2165191)Show SMILES C[C@@H](Nc1ccccc1C(O)=O)c1cc(C)cn2c1nc(cc2=O)N1CCOCC1 |r| Show InChI InChI=1S/C22H24N4O4/c1-14-11-17(15(2)23-18-6-4-3-5-16(18)22(28)29)21-24-19(12-20(27)26(21)13-14)25-7-9-30-10-8-25/h3-6,11-13,15,23H,7-10H2,1-2H3,(H,28,29)/t15-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant His6-tagged PI3K p110beta expressed in baculovirus infected cells using DiC8-PI(4,5)P2 as substrate after 20 mins by ... |
Bioorg Med Chem Lett 22: 6671-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.102
BindingDB Entry DOI: 10.7270/Q2445NKM |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50395821

(CHEMBL2165191)Show SMILES C[C@@H](Nc1ccccc1C(O)=O)c1cc(C)cn2c1nc(cc2=O)N1CCOCC1 |r| Show InChI InChI=1S/C22H24N4O4/c1-14-11-17(15(2)23-18-6-4-3-5-16(18)22(28)29)21-24-19(12-20(27)26(21)13-14)25-7-9-30-10-8-25/h3-6,11-13,15,23H,7-10H2,1-2H3,(H,28,29)/t15-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Curtin University
Curated by ChEMBL
| Assay Description
ATP competitive inhibition of human recombinant PI3Kbeta assessed as PIP3 production by Alpha-screen assay |
J Med Chem 60: 47-65 (2017)
Article DOI: 10.1021/acs.jmedchem.6b00963
BindingDB Entry DOI: 10.7270/Q2PG1TPX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50395821

(CHEMBL2165191)Show SMILES C[C@@H](Nc1ccccc1C(O)=O)c1cc(C)cn2c1nc(cc2=O)N1CCOCC1 |r| Show InChI InChI=1S/C22H24N4O4/c1-14-11-17(15(2)23-18-6-4-3-5-16(18)22(28)29)21-24-19(12-20(27)26(21)13-14)25-7-9-30-10-8-25/h3-6,11-13,15,23H,7-10H2,1-2H3,(H,28,29)/t15-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PI3Kbeta assessed as PIP3 production by AlphaScreen assay |
Bioorg Med Chem Lett 24: 3936-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.07.007
BindingDB Entry DOI: 10.7270/Q2BP04F3 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50395821

(CHEMBL2165191)Show SMILES C[C@@H](Nc1ccccc1C(O)=O)c1cc(C)cn2c1nc(cc2=O)N1CCOCC1 |r| Show InChI InChI=1S/C22H24N4O4/c1-14-11-17(15(2)23-18-6-4-3-5-16(18)22(28)29)21-24-19(12-20(27)26(21)13-14)25-7-9-30-10-8-25/h3-6,11-13,15,23H,7-10H2,1-2H3,(H,28,29)/t15-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human PI3Kbeta assessed as reduction in PIP3 formation by AlphaScreen assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00986
BindingDB Entry DOI: 10.7270/Q27M0CRD |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50395821

(CHEMBL2165191)Show SMILES C[C@@H](Nc1ccccc1C(O)=O)c1cc(C)cn2c1nc(cc2=O)N1CCOCC1 |r| Show InChI InChI=1S/C22H24N4O4/c1-14-11-17(15(2)23-18-6-4-3-5-16(18)22(28)29)21-24-19(12-20(27)26(21)13-14)25-7-9-30-10-8-25/h3-6,11-13,15,23H,7-10H2,1-2H3,(H,28,29)/t15-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arkansas for Medical Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human PIK3CB by alphascreen assay |
J Med Chem 63: 441-469 (2020)
Article DOI: 10.1021/acs.jmedchem.9b00640
BindingDB Entry DOI: 10.7270/Q2HD800F |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50395821

(CHEMBL2165191)Show SMILES C[C@@H](Nc1ccccc1C(O)=O)c1cc(C)cn2c1nc(cc2=O)N1CCOCC1 |r| Show InChI InChI=1S/C22H24N4O4/c1-14-11-17(15(2)23-18-6-4-3-5-16(18)22(28)29)21-24-19(12-20(27)26(21)13-14)25-7-9-30-10-8-25/h3-6,11-13,15,23H,7-10H2,1-2H3,(H,28,29)/t15-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kbeta in human MAD-MB-468 cells assessed as inhibition of Ser473 Akt phosphorylation by cellular potency assay |
Bioorg Med Chem Lett 24: 3928-35 (2014)
Article DOI: 10.1016/j.bmcl.2014.06.040
BindingDB Entry DOI: 10.7270/Q2BZ67PH |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data