 Found 7 hits Enz. Inhib. hit(s) with Target = 'Prostaglandin G/H synthase 2' and Ligand = 'BDBM85245'
Found 7 hits Enz. Inhib. hit(s) with Target = 'Prostaglandin G/H synthase 2' and Ligand = 'BDBM85245' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM85245
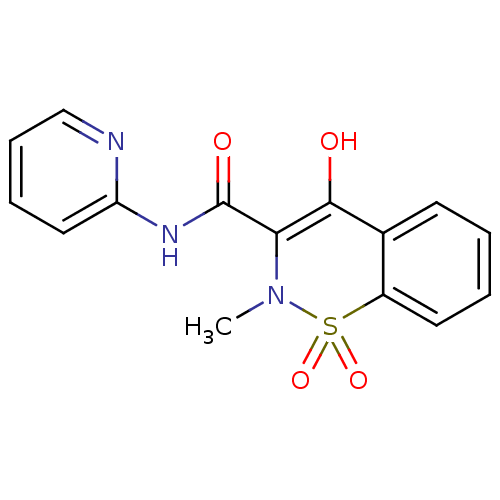
(CAS_36322-90-4 | NSC_4856 | Piroxicam)Show SMILES CN1C(C(=O)Nc2ccccn2)=C(O)c2ccccc2S1(=O)=O |t:12| Show InChI InChI=1S/C15H13N3O4S/c1-18-13(15(20)17-12-8-4-5-9-16-12)14(19)10-6-2-3-7-11(10)23(18,21)22/h2-9,19H,1H3,(H,16,17,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
St. Bartholomew's and the Royal London School of Medicine and Dentistry
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 96: 7563-8 (1999)
Article DOI: 10.1073/pnas.96.13.7563
BindingDB Entry DOI: 10.7270/Q21G0JT4 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM85245

(CAS_36322-90-4 | NSC_4856 | Piroxicam)Show SMILES CN1C(C(=O)Nc2ccccn2)=C(O)c2ccccc2S1(=O)=O |t:12| Show InChI InChI=1S/C15H13N3O4S/c1-18-13(15(20)17-12-8-4-5-9-16-12)14(19)10-6-2-3-7-11(10)23(18,21)22/h2-9,19H,1H3,(H,16,17,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 2.11E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dallas Department of Veterans Affairs Medical Center
Curated by PDSP Ki Database
| |
Am J Med 104: 413-21 (1998)
Article DOI: 10.1016/s0002-9343(98)00091-6
BindingDB Entry DOI: 10.7270/Q2F18X97 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM85245

(CAS_36322-90-4 | NSC_4856 | Piroxicam)Show SMILES CN1C(C(=O)Nc2ccccn2)=C(O)c2ccccc2S1(=O)=O |t:12| Show InChI InChI=1S/C15H13N3O4S/c1-18-13(15(20)17-12-8-4-5-9-16-12)14(19)10-6-2-3-7-11(10)23(18,21)22/h2-9,19H,1H3,(H,16,17,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 7.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
St. Bartholomew's and the Royal London School of Medicine and Dentistry
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 96: 7563-8 (1999)
Article DOI: 10.1073/pnas.96.13.7563
BindingDB Entry DOI: 10.7270/Q21G0JT4 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM85245

(CAS_36322-90-4 | NSC_4856 | Piroxicam)Show SMILES CN1C(C(=O)Nc2ccccn2)=C(O)c2ccccc2S1(=O)=O |t:12| Show InChI InChI=1S/C15H13N3O4S/c1-18-13(15(20)17-12-8-4-5-9-16-12)14(19)10-6-2-3-7-11(10)23(18,21)22/h2-9,19H,1H3,(H,16,17,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
PubMed
| 9.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 296: 558-66 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4VPJ |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM85245

(CAS_36322-90-4 | NSC_4856 | Piroxicam)Show SMILES CN1C(C(=O)Nc2ccccn2)=C(O)c2ccccc2S1(=O)=O |t:12| Show InChI InChI=1S/C15H13N3O4S/c1-18-13(15(20)17-12-8-4-5-9-16-12)14(19)10-6-2-3-7-11(10)23(18,21)22/h2-9,19H,1H3,(H,16,17,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 1.02E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Wroclaw Medical University
Curated by ChEMBL
| Assay Description
Inhibition of COX-2 (unknown origin) using arachidonic acid as substrate preincubated for 2 mins followed by substrate addition in presence of TMPD b... |
Bioorg Med Chem 25: 316-326 (2017)
Article DOI: 10.1016/j.bmc.2016.10.036
BindingDB Entry DOI: 10.7270/Q21V5GZS |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM85245

(CAS_36322-90-4 | NSC_4856 | Piroxicam)Show SMILES CN1C(C(=O)Nc2ccccn2)=C(O)c2ccccc2S1(=O)=O |t:12| Show InChI InChI=1S/C15H13N3O4S/c1-18-13(15(20)17-12-8-4-5-9-16-12)14(19)10-6-2-3-7-11(10)23(18,21)22/h2-9,19H,1H3,(H,16,17,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 1.03E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Wroclaw Medical University
Curated by ChEMBL
| Assay Description
Inhibition of COX-2 (unknown origin) using arachidonic acid as substrate preincubated for 2 mins followed by substrate addition in presence of TMPD b... |
Bioorg Med Chem 25: 316-326 (2017)
Article DOI: 10.1016/j.bmc.2016.10.036
BindingDB Entry DOI: 10.7270/Q21V5GZS |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM85245

(CAS_36322-90-4 | NSC_4856 | Piroxicam)Show SMILES CN1C(C(=O)Nc2ccccn2)=C(O)c2ccccc2S1(=O)=O |t:12| Show InChI InChI=1S/C15H13N3O4S/c1-18-13(15(20)17-12-8-4-5-9-16-12)14(19)10-6-2-3-7-11(10)23(18,21)22/h2-9,19H,1H3,(H,16,17,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
PubMed
| n/a | n/a | 2.18E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Pharmaceutical Company
Curated by ChEMBL
| Assay Description
Tested for inhibitory activity against Prostaglandin G/H synthase 2 from human |
J Med Chem 38: 3895-901 (1995)
BindingDB Entry DOI: 10.7270/Q28C9WX5 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data