

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Type-2 angiotensin II receptor (RAT) | BDBM82433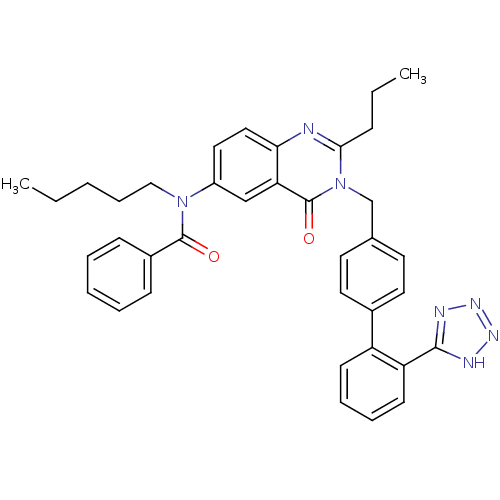 (CHEMBL302102 | L-159,689 | L-159689 | N-Pentyl-N-[...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.840 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by PDSP Ki Database | J Pharmacol Exp Ther 272: 612-8 (1995) BindingDB Entry DOI: 10.7270/Q2FB51GP | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Type-2 angiotensin II receptor (RAT) | BDBM82433 (CHEMBL302102 | L-159,689 | L-159689 | N-Pentyl-N-[...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 351 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by PDSP Ki Database | J Pharmacol Exp Ther 272: 612-8 (1995) BindingDB Entry DOI: 10.7270/Q2FB51GP | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Type-2 angiotensin II receptor (RAT) | BDBM82433 (CHEMBL302102 | L-159,689 | L-159689 | N-Pentyl-N-[...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity against Angiotensin II receptor type 2 from rat mid brain using [125I]-Sar1-Ile8-Ang II without BSA | J Med Chem 36: 3207-10 (1993) BindingDB Entry DOI: 10.7270/Q2QR4W6P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Type-2 angiotensin II receptor (RAT) | BDBM82433 (CHEMBL302102 | L-159,689 | L-159689 | N-Pentyl-N-[...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description In vitro inhibitory activity against Angiotensin II receptor, type 2 in rat midbrain membrane preparations. | J Med Chem 38: 3741-58 (1995) BindingDB Entry DOI: 10.7270/Q2Q81C35 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Type-2 angiotensin II receptor (RAT) | BDBM82433 (CHEMBL302102 | L-159,689 | L-159689 | N-Pentyl-N-[...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description In vivo inhibitory concentration of the compound against Angiotensin II, type 2 of rat midbrain membrane | Bioorg Med Chem Lett 4: 2337-2342 (1994) Article DOI: 10.1016/0960-894X(94)85036-4 BindingDB Entry DOI: 10.7270/Q2X066Z9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Type-2 angiotensin II receptor (Homo sapiens (Human)) | BDBM82433 (CHEMBL302102 | L-159,689 | L-159689 | N-Pentyl-N-[...) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Organon Laboratories Curated by ChEMBL | Assay Description Inhibitory concentration against angiotensin II receptor, type 2 | J Med Chem 48: 6523-43 (2005) Article DOI: 10.1021/jm058225d BindingDB Entry DOI: 10.7270/Q2SF2WZ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Type-2 angiotensin II receptor (RAT) | BDBM82433 (CHEMBL302102 | L-159,689 | L-159689 | N-Pentyl-N-[...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description In vitro binding affinity determined by its ability to displace the specific binding ligand [125I]-Sar1, Ile8-AII from AT2 receptor in rat midbrain m... | Bioorg Med Chem Lett 4: 17-22 (1994) Article DOI: 10.1016/S0960-894X(01)81115-X BindingDB Entry DOI: 10.7270/Q2G73DPF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Type-2 angiotensin II receptor (RAT) | BDBM82433 (CHEMBL302102 | L-159,689 | L-159689 | N-Pentyl-N-[...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description In vitro ability of the compound to inhibit the binding of radioligand 125I[Sar1,IIe8]AII to AT2 receptor from rat midbrain | Bioorg Med Chem Lett 4: 2787-2792 (1994) Article DOI: 10.1016/S0960-894X(01)80595-3 BindingDB Entry DOI: 10.7270/Q2WS8T68 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Type-2 angiotensin II receptor (RAT) | BDBM82433 (CHEMBL302102 | L-159,689 | L-159689 | N-Pentyl-N-[...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of [125I]-Sar1-Ile8-Ang II binding to rat midbrain Angiotensin II receptor type 2 | Bioorg Med Chem Lett 6: 923-928 (1996) Article DOI: 10.1016/0960-894X(96)00116-3 BindingDB Entry DOI: 10.7270/Q2MC900D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Type-2 angiotensin II receptor (RAT) | BDBM82433 (CHEMBL302102 | L-159,689 | L-159689 | N-Pentyl-N-[...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Pharmaceutical Company Curated by ChEMBL | Assay Description Inhibitory activity at Angiotensin II type 2 receptor. | J Med Chem 39: 625-56 (1996) Article DOI: 10.1021/jm9504722 BindingDB Entry DOI: 10.7270/Q29P3299 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Type-2 angiotensin II receptor (Homo sapiens (Human)) | BDBM82433 (CHEMBL302102 | L-159,689 | L-159689 | N-Pentyl-N-[...) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description In vivo inhibitory concentration of the compound against AT2 receptor of human adrenal membrane | Bioorg Med Chem Lett 4: 2337-2342 (1994) Article DOI: 10.1016/0960-894X(94)85036-4 BindingDB Entry DOI: 10.7270/Q2X066Z9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||