

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551946 (CHEMBL4764328) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.0160 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551956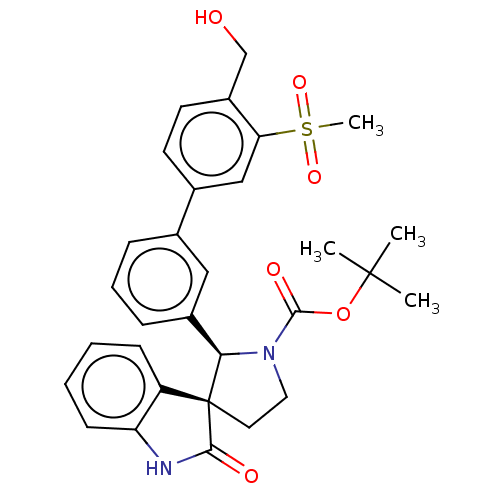 (CHEMBL4756418) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | PDB Article PubMed | n/a | n/a | n/a | n/a | 0.0350 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551953 (CHEMBL4764838) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.0520 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551945 (CHEMBL4747448) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.0670 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551964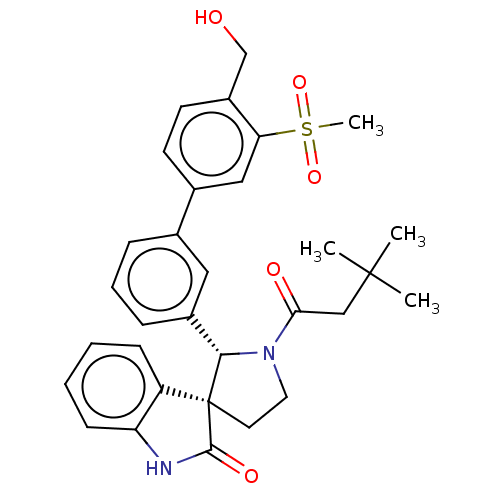 (CHEMBL4745005) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.0830 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551947 (CHEMBL4797369) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.0880 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551950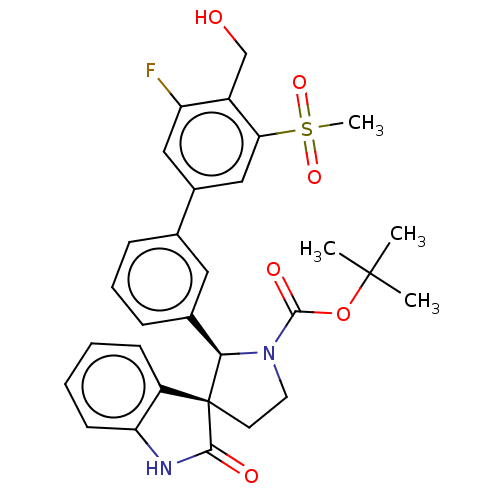 (CHEMBL4757761) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.0920 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551948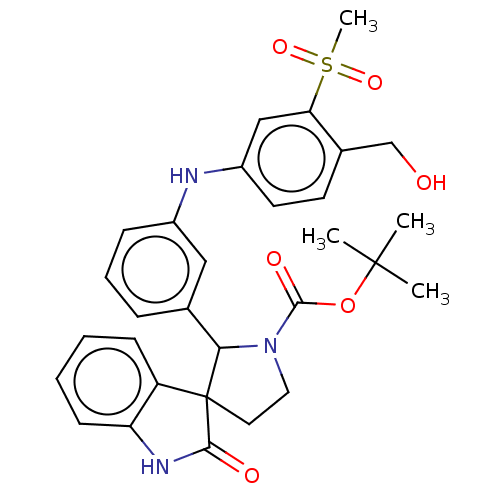 (CHEMBL4760781) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.0950 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551951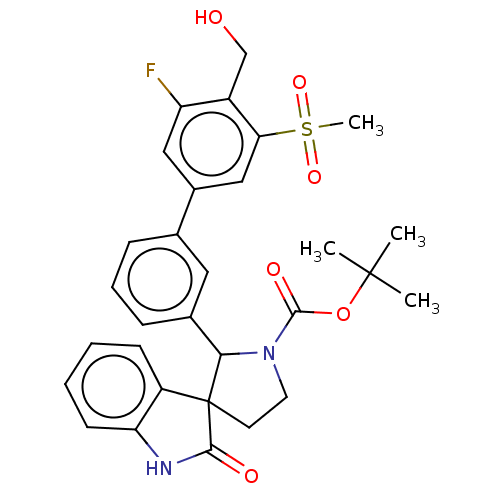 (CHEMBL4749385) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.103 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM19993 (CHEMBL62136 | N-[4-(1,1,1,3,3,3-hexafluoro-2-hydro...) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | n/a | 0.108 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551957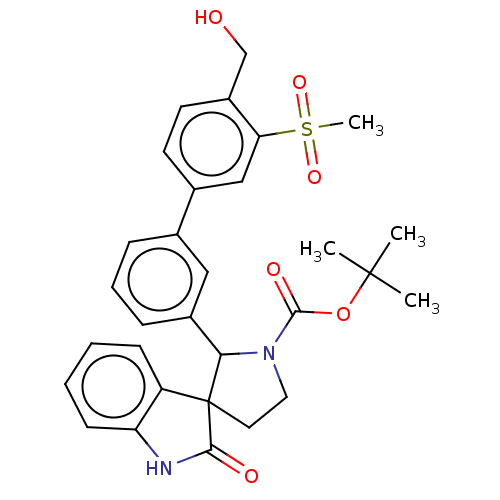 (CHEMBL4755317) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | PDB Article PubMed | n/a | n/a | n/a | n/a | 0.174 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM26066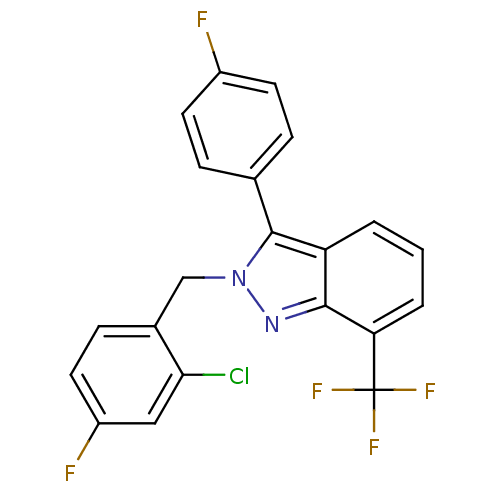 (2-[(2-chloro-4-fluorophenyl)methyl]-3-(4-fluorophe...) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.212 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM19992 (2-[3-[3-[[2-chloro-3-(trifluoromethyl)phenyl]methy...) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | n/a | 0.228 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551949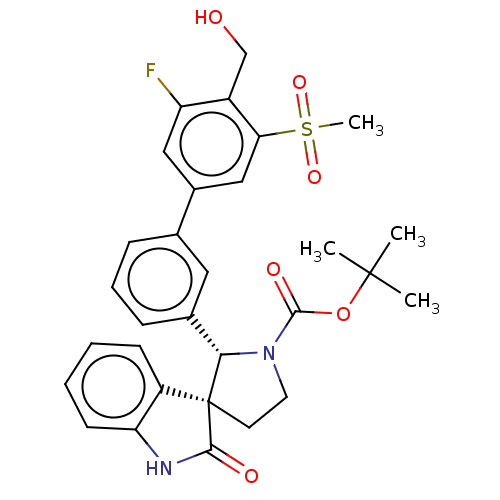 (CHEMBL4762546) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.281 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551954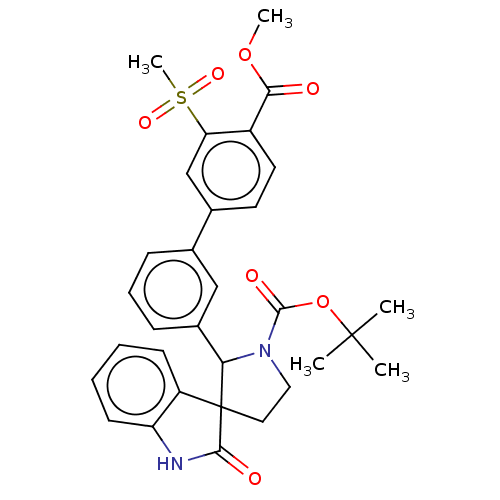 (CHEMBL4757158) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.341 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551963 (CHEMBL4764539) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.369 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551955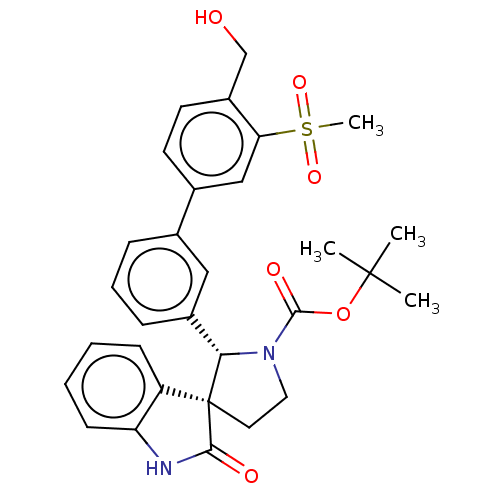 (CHEMBL4777240) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.430 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551962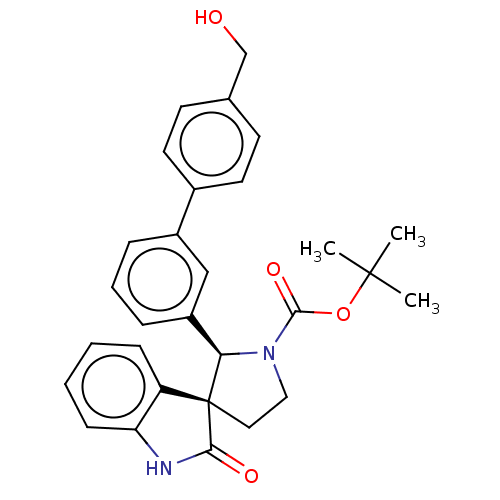 (CHEMBL4781683) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.445 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551944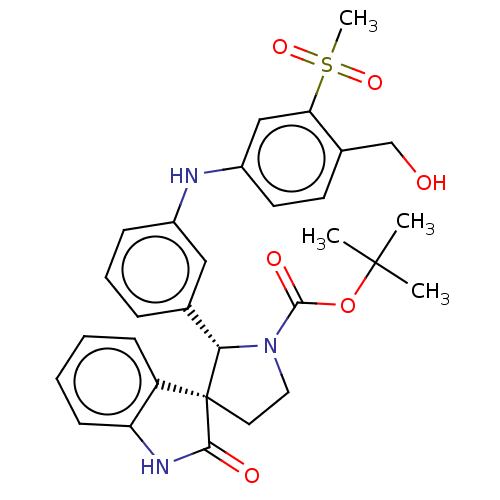 (CHEMBL4748480) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.474 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50204072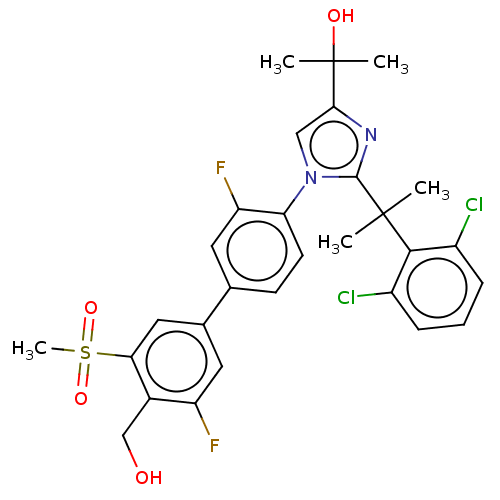 (CHEMBL3945199 | US10543183, Compound 12 | US109459...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Curated by ChEMBL | Assay Description Agonist activity at LXR-beta in human HeLa cells assessed as induction of ABCA1 by beta-galactosidase/luciferase reporter gene assay | ACS Med Chem Lett 7: 1207-1212 (2016) Article DOI: 10.1021/acsmedchemlett.6b00234 BindingDB Entry DOI: 10.7270/Q2V98B13 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551961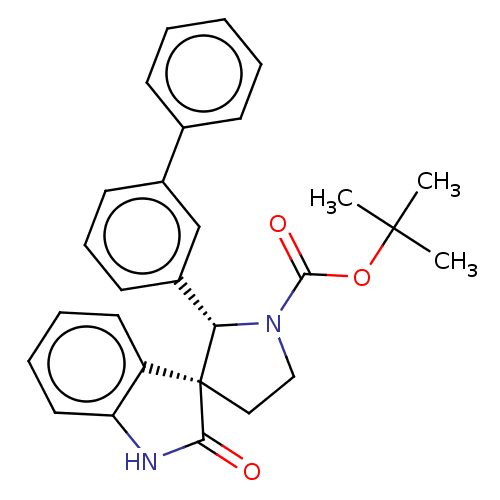 (CHEMBL4791199) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.632 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551958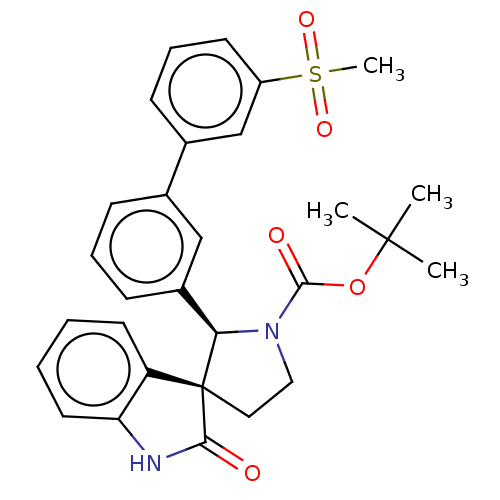 (CHEMBL4780872) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.680 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551952 (CHEMBL4762542) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.905 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50167697 ((4aR,9S)-6-Hydroxy-1,4a-dimethyl-1,2,3,4,4a,9,10,1...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Effective concentration for cofactor association with recombinant liver X receptor-beta | Bioorg Med Chem Lett 15: 2824-8 (2005) Article DOI: 10.1016/j.bmcl.2005.03.100 BindingDB Entry DOI: 10.7270/Q29G5NM2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50204073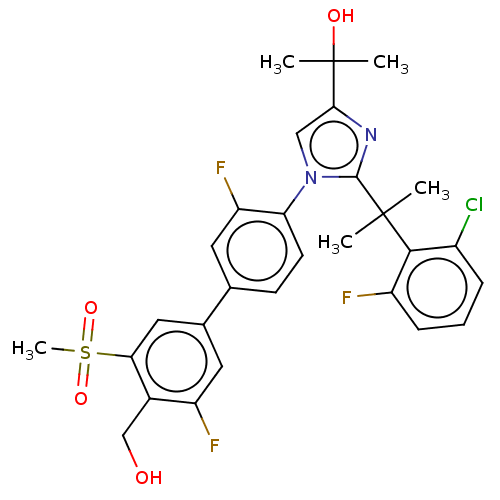 (CHEMBL3917300) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Curated by ChEMBL | Assay Description Agonist activity at LXR-beta in human HeLa cells assessed as induction of ABCA1 by beta-galactosidase/luciferase reporter gene assay | ACS Med Chem Lett 7: 1207-1212 (2016) Article DOI: 10.1021/acsmedchemlett.6b00234 BindingDB Entry DOI: 10.7270/Q2V98B13 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50167698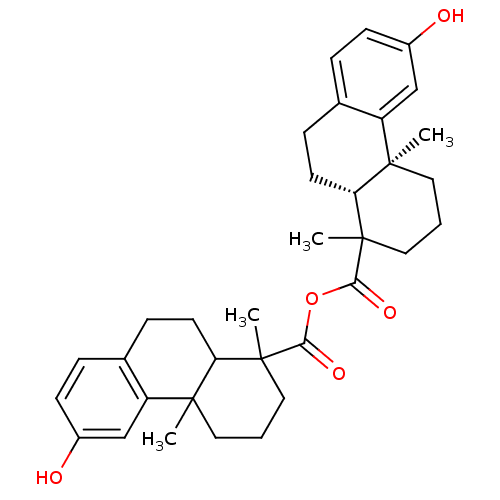 (13-hydroxy-2,6-dimethyl-(2S,7R)-tricyclo[8.4.0.02,...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Effective concentration against liver X receptor-beta in HEK293 cell transactivation assay | Bioorg Med Chem Lett 15: 2824-8 (2005) Article DOI: 10.1016/j.bmcl.2005.03.100 BindingDB Entry DOI: 10.7270/Q29G5NM2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50167697 ((4aR,9S)-6-Hydroxy-1,4a-dimethyl-1,2,3,4,4a,9,10,1...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Effective concentration in recombinant human LXRbeta ligand binding domain in homogeneous time-resolved fluorescence assay | Bioorg Med Chem Lett 15: 4574-8 (2005) Article DOI: 10.1016/j.bmcl.2005.06.100 BindingDB Entry DOI: 10.7270/Q2154GKK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50415821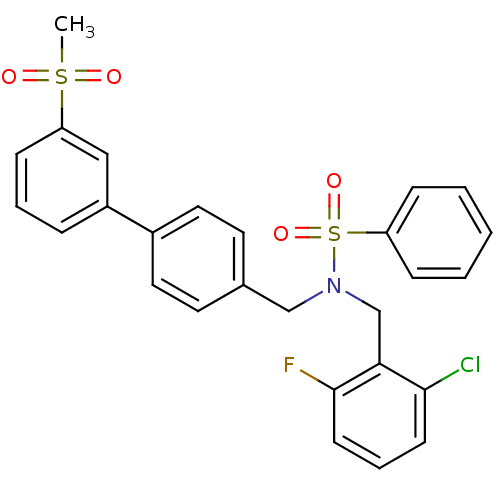 (CHEMBL1093554) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Agonist activity at LXRbeta ligand binding domain-mediated transcriptional activity in african green monkey CV1 cells co-transfected with Gal4-SRC1 b... | J Med Chem 53: 3412-6 (2010) Article DOI: 10.1021/jm901797p BindingDB Entry DOI: 10.7270/Q2NP25P6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50177010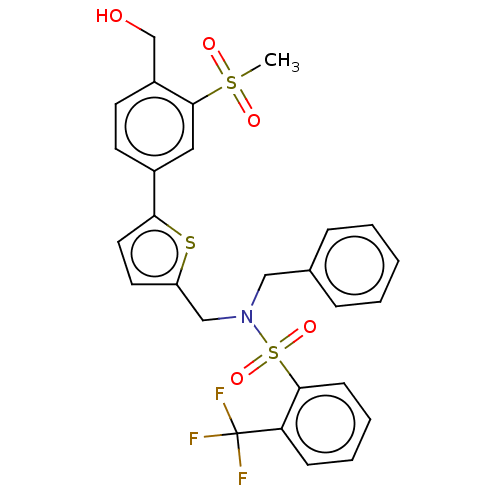 (CHEMBL3814006) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Binding to human LXRbeta | J Med Chem 59: 3264-71 (2016) Article DOI: 10.1021/acs.jmedchem.5b02029 BindingDB Entry DOI: 10.7270/Q2XP76V7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50551959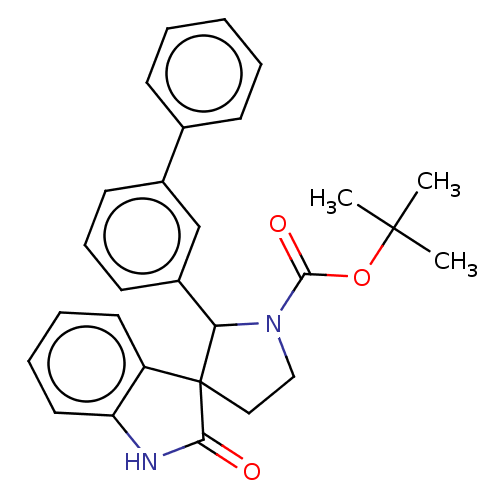 (CHEMBL4759393) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 1.60 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human RXRalpha/LXRbeta expressed in HEK293 cells measured after 20 hrs by dual luciferase reporter gene assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112240 BindingDB Entry DOI: 10.7270/Q2MG7T4D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50204082 (CHEMBL3945820) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Curated by ChEMBL | Assay Description Agonist activity at LXR-beta in human HeLa cells assessed as induction of ABCA1 by beta-galactosidase/luciferase reporter gene assay | ACS Med Chem Lett 7: 1207-1212 (2016) Article DOI: 10.1021/acsmedchemlett.6b00234 BindingDB Entry DOI: 10.7270/Q2V98B13 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50204078 (CHEMBL3926292) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Curated by ChEMBL | Assay Description Agonist activity at LXR-beta in human HeLa cells assessed as induction of ABCA1 by beta-galactosidase/luciferase reporter gene assay | ACS Med Chem Lett 7: 1207-1212 (2016) Article DOI: 10.1021/acsmedchemlett.6b00234 BindingDB Entry DOI: 10.7270/Q2V98B13 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50167694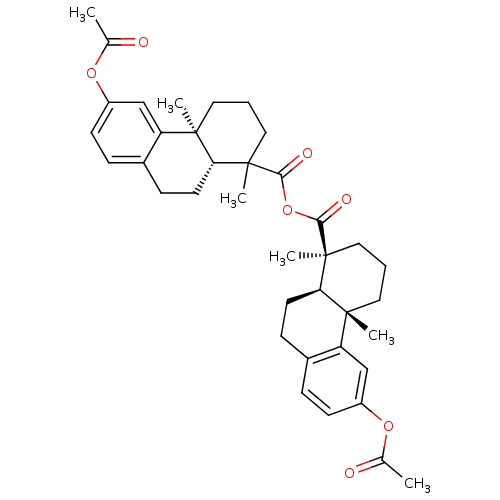 (2,6-dimethyl-13-methylcarbonyloxy-(2S)-tricyclo[8....) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Effective concentration in recombinant human LXRbeta ligand binding domain in homogeneous time-resolved fluorescence assay | Bioorg Med Chem Lett 15: 4574-8 (2005) Article DOI: 10.1016/j.bmcl.2005.06.100 BindingDB Entry DOI: 10.7270/Q2154GKK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50167694 (2,6-dimethyl-13-methylcarbonyloxy-(2S)-tricyclo[8....) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Effective concentration for cofactor association with recombinant liver X receptor-beta | Bioorg Med Chem Lett 15: 2824-8 (2005) Article DOI: 10.1016/j.bmcl.2005.03.100 BindingDB Entry DOI: 10.7270/Q29G5NM2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50034791 (CHEMBL3360960) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Curated by ChEMBL | Assay Description Transactivation of LXRbeta (unknown origin) expressed in CV1 cells by luciferase reporter gene assay | Bioorg Med Chem Lett 25: 372-7 (2014) Article DOI: 10.1016/j.bmcl.2014.11.029 BindingDB Entry DOI: 10.7270/Q2154JN9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50204074 (CHEMBL3910597) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Curated by ChEMBL | Assay Description Agonist activity at LXR-beta in human HeLa cells assessed as induction of ABCA1 by beta-galactosidase/luciferase reporter gene assay | ACS Med Chem Lett 7: 1207-1212 (2016) Article DOI: 10.1021/acsmedchemlett.6b00234 BindingDB Entry DOI: 10.7270/Q2V98B13 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50204079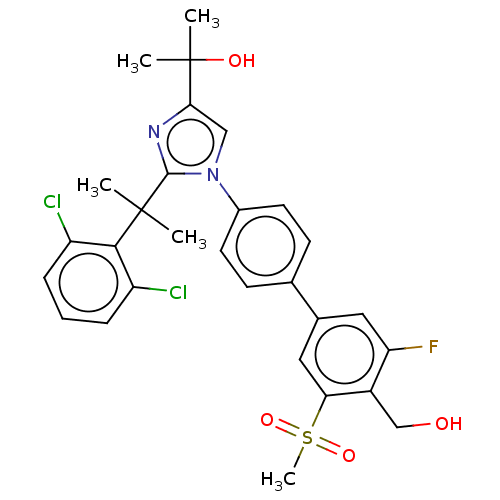 (CHEMBL3935187) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 3 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Curated by ChEMBL | Assay Description Agonist activity at LXR-beta in human HeLa cells assessed as induction of ABCA1 by beta-galactosidase/luciferase reporter gene assay | ACS Med Chem Lett 7: 1207-1212 (2016) Article DOI: 10.1021/acsmedchemlett.6b00234 BindingDB Entry DOI: 10.7270/Q2V98B13 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50167697 ((4aR,9S)-6-Hydroxy-1,4a-dimethyl-1,2,3,4,4a,9,10,1...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | <3 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Effective concentration in transactivation assay using a chimeric LXR construct in HEK-293 cells for LXRbeta receptor | Bioorg Med Chem Lett 15: 4574-8 (2005) Article DOI: 10.1016/j.bmcl.2005.06.100 BindingDB Entry DOI: 10.7270/Q2154GKK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50241903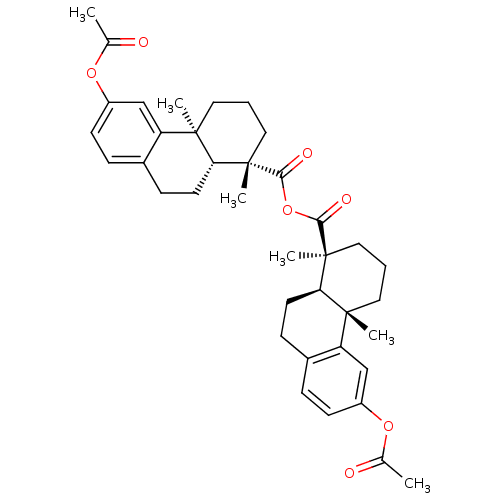 (CHEMBL506838 | acetyl Podocarpic acid anhydride) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | <3 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Agonist activity at human recombinant LXRbeta expressed in Escherichia coli BL21 cells assessed as association of recombinant SRC1 to LXRbeta ligand ... | J Nat Prod 68: 1247-52 (2005) Article DOI: 10.1021/np050182g BindingDB Entry DOI: 10.7270/Q2GH9HQ5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50167697 ((4aR,9S)-6-Hydroxy-1,4a-dimethyl-1,2,3,4,4a,9,10,1...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | <3 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Effective concentration against liver X receptor-beta in HEK293 cell transactivation assay | Bioorg Med Chem Lett 15: 2824-8 (2005) Article DOI: 10.1016/j.bmcl.2005.03.100 BindingDB Entry DOI: 10.7270/Q29G5NM2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50167698 (13-hydroxy-2,6-dimethyl-(2S,7R)-tricyclo[8.4.0.02,...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | <3 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Effective concentration for cofactor association with recombinant liver X receptor-beta | Bioorg Med Chem Lett 15: 2824-8 (2005) Article DOI: 10.1016/j.bmcl.2005.03.100 BindingDB Entry DOI: 10.7270/Q29G5NM2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50415819 (CHEMBL1091976) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 3.20 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Agonist activity at LXRbeta ligand binding domain-mediated transcriptional activity in african green monkey CV1 cells co-transfected with Gal4-SRC1 b... | J Med Chem 53: 3412-6 (2010) Article DOI: 10.1021/jm901797p BindingDB Entry DOI: 10.7270/Q2NP25P6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50415825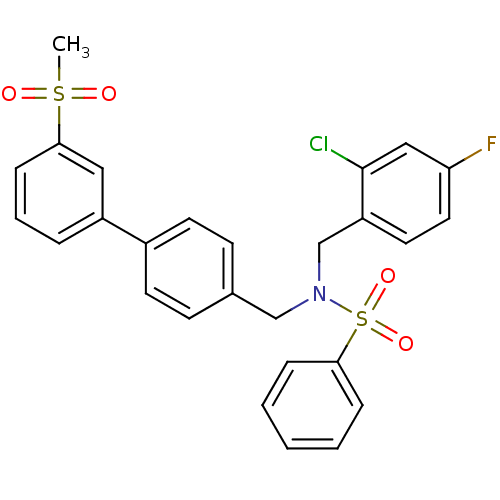 (CHEMBL1091034) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 3.20 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Agonist activity at LXRbeta ligand binding domain-mediated transcriptional activity in african green monkey CV1 cells co-transfected with Gal4-SRC1 b... | J Med Chem 53: 3412-6 (2010) Article DOI: 10.1021/jm901797p BindingDB Entry DOI: 10.7270/Q2NP25P6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50177011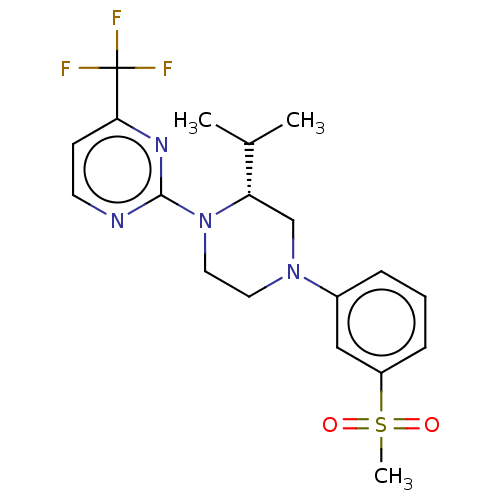 (CHEMBL3815014 | US10144715, Compound 7-13) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | n/a | n/a | 4 | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc. US Patent | Assay Description This assay is based on the ability of the LXR-LBDs (LXRα and LXRβ) to recruit and interact with a co-activator peptide. This assay was run ... | US Patent US10144715 (2018) BindingDB Entry DOI: 10.7270/Q2PV6NFD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM304359 ((R)-2-isopropyl-4-(3-(methylsulfonyl)phenyl)-1-(5-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | n/a | n/a | 4 | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc. US Patent | Assay Description This assay is based on the ability of the LXR-LBDs (LXRα and LXRβ) to recruit and interact with a co-activator peptide. This assay was run ... | US Patent US10144715 (2018) BindingDB Entry DOI: 10.7270/Q2PV6NFD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM27174 (1H-indol-1-yl tertiary amine, 18 | 2-{4-[3-({[2-ch...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 4 | n/a | n/a | 7.5 | 22 |
GSK | Assay Description The LXR LiSA measures the ligand-dependent recruitment of a 25 amino acid fragment of the steroid receptor coactivator 1 (SRC1) to the ligand-binding... | Bioorg Med Chem Lett 19: 1097-100 (2009) Article DOI: 10.1016/j.bmcl.2009.01.004 BindingDB Entry DOI: 10.7270/Q28W3BMD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-alpha/LXR-beta (Homo sapiens (Human)) | BDBM50177015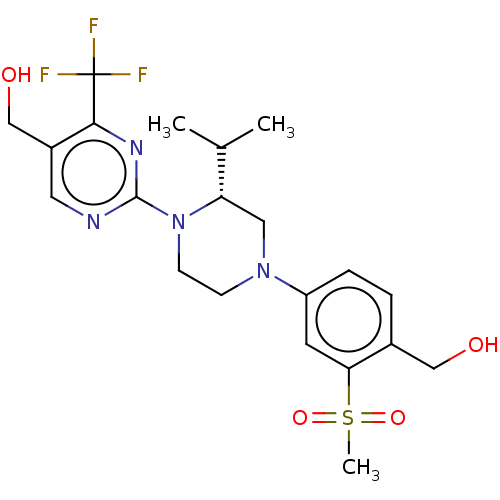 (CHEMBL3814206 | US10144715, Compound 19-1) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | n/a | n/a | 4 | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Agonist activity at LXR in human THP1 cells assessed as upregulation of ABCA1 gene expression after 24 hrs by RT-PCR analysis | J Med Chem 59: 3264-71 (2016) Article DOI: 10.1021/acs.jmedchem.5b02029 BindingDB Entry DOI: 10.7270/Q2XP76V7 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM19993 (CHEMBL62136 | N-[4-(1,1,1,3,3,3-hexafluoro-2-hydro...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | n/a | 4.70 | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research and Development Curated by ChEMBL | Assay Description Agonist activity at LXRbeta by TR-FRET assay | J Med Chem 54: 788-808 (2012) Article DOI: 10.1021/jm101063h BindingDB Entry DOI: 10.7270/Q208668Q | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM50204075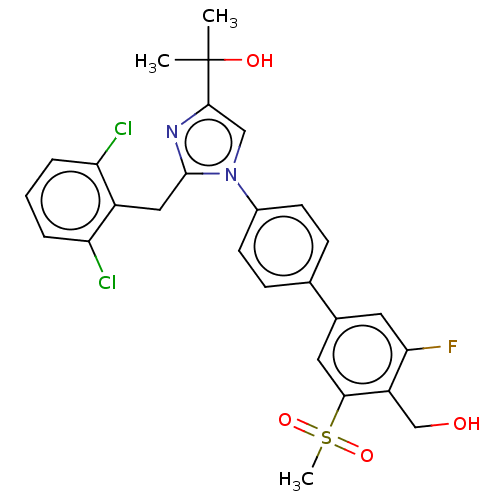 (CHEMBL3980683) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 5 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Curated by ChEMBL | Assay Description Agonist activity at LXR-beta in human HeLa cells assessed as induction of ABCA1 by beta-galactosidase/luciferase reporter gene assay | ACS Med Chem Lett 7: 1207-1212 (2016) Article DOI: 10.1021/acsmedchemlett.6b00234 BindingDB Entry DOI: 10.7270/Q2V98B13 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxysterols receptor LXR-beta (Homo sapiens (Human)) | BDBM19992 (2-[3-[3-[[2-chloro-3-(trifluoromethyl)phenyl]methy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | n/a | 5 | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Binding to human LXRbeta | J Med Chem 59: 3264-71 (2016) Article DOI: 10.1021/acs.jmedchem.5b02029 BindingDB Entry DOI: 10.7270/Q2XP76V7 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Displayed 1 to 50 (of 1590 total ) | Next | Last >> |