 Found 17 hits Enz. Inhib. hit(s) with all data for entry = 50049184
Found 17 hits Enz. Inhib. hit(s) with all data for entry = 50049184 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50235922

(CHEMBL4093614)Show SMILES Cl.Cc1[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c(C)c1NC(=O)[C@H]1CCCN1 |r| Show InChI InChI=1S/C20H21FN4O2.ClH/c1-10-17(9-14-13-8-12(21)5-6-15(13)24-19(14)26)23-11(2)18(10)25-20(27)16-4-3-7-22-16;/h5-6,8-9,16,22-23H,3-4,7H2,1-2H3,(H,24,26)(H,25,27);1H/b14-9-;/t16-;/m1./s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of human FLT3 |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM4814
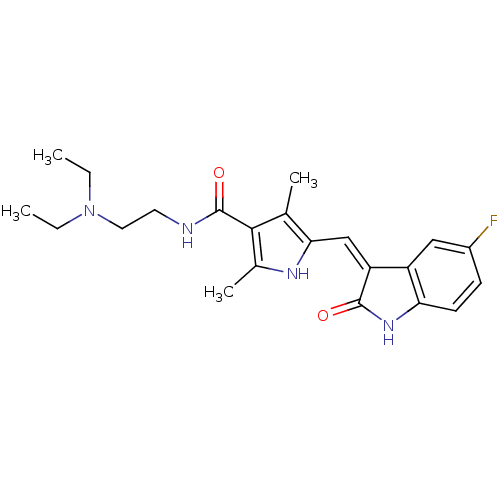
(CHEMBL535 | N-[2-(diethylamino)ethyl]-5-[(Z)-(5-fl...)Show SMILES CCN(CC)CCNC(=O)c1c(C)[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c1C Show InChI InChI=1S/C22H27FN4O2/c1-5-27(6-2)10-9-24-22(29)20-13(3)19(25-14(20)4)12-17-16-11-15(23)7-8-18(16)26-21(17)28/h7-8,11-12,25H,5-6,9-10H2,1-4H3,(H,24,29)(H,26,28)/b17-12- | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of human FLT3 |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha
(Homo sapiens (Human)) | BDBM4814

(CHEMBL535 | N-[2-(diethylamino)ethyl]-5-[(Z)-(5-fl...)Show SMILES CCN(CC)CCNC(=O)c1c(C)[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c1C Show InChI InChI=1S/C22H27FN4O2/c1-5-27(6-2)10-9-24-22(29)20-13(3)19(25-14(20)4)12-17-16-11-15(23)7-8-18(16)26-21(17)28/h7-8,11-12,25H,5-6,9-10H2,1-4H3,(H,24,29)(H,26,28)/b17-12- | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of human PDGFRalpha |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50388686
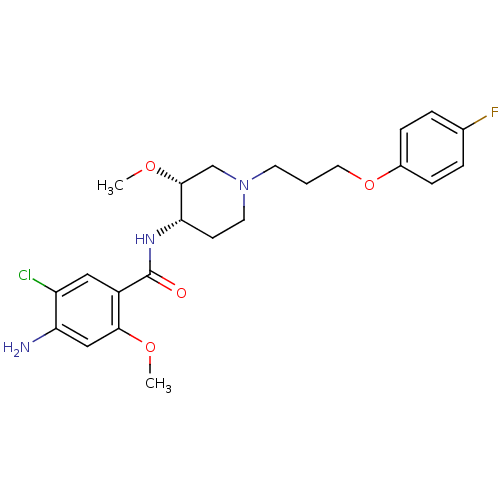
(CHEMBL74656)Show SMILES CO[C@@H]1CN(CCCOc2ccc(F)cc2)CC[C@@H]1NC(=O)c1cc(Cl)c(N)cc1OC |r| Show InChI InChI=1S/C23H29ClFN3O4/c1-30-21-13-19(26)18(24)12-17(21)23(29)27-20-8-10-28(14-22(20)31-2)9-3-11-32-16-6-4-15(25)5-7-16/h4-7,12-13,20,22H,3,8-11,14,26H2,1-2H3,(H,27,29)/t20-,22+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of human ERG expressed in CHO cells by Qpatch assay |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha
(Homo sapiens (Human)) | BDBM50235922

(CHEMBL4093614)Show SMILES Cl.Cc1[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c(C)c1NC(=O)[C@H]1CCCN1 |r| Show InChI InChI=1S/C20H21FN4O2.ClH/c1-10-17(9-14-13-8-12(21)5-6-15(13)24-19(14)26)23-11(2)18(10)25-20(27)16-4-3-7-22-16;/h5-6,8-9,16,22-23H,3-4,7H2,1-2H3,(H,24,26)(H,25,27);1H/b14-9-;/t16-;/m1./s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 175 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of human PDGFRalpha |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Mast/stem cell growth factor receptor Kit
(Homo sapiens (Human)) | BDBM50235922

(CHEMBL4093614)Show SMILES Cl.Cc1[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c(C)c1NC(=O)[C@H]1CCCN1 |r| Show InChI InChI=1S/C20H21FN4O2.ClH/c1-10-17(9-14-13-8-12(21)5-6-15(13)24-19(14)26)23-11(2)18(10)25-20(27)16-4-3-7-22-16;/h5-6,8-9,16,22-23H,3-4,7H2,1-2H3,(H,24,26)(H,25,27);1H/b14-9-;/t16-;/m1./s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 227 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of human KIT |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Mast/stem cell growth factor receptor Kit
(Homo sapiens (Human)) | BDBM4814

(CHEMBL535 | N-[2-(diethylamino)ethyl]-5-[(Z)-(5-fl...)Show SMILES CCN(CC)CCNC(=O)c1c(C)[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c1C Show InChI InChI=1S/C22H27FN4O2/c1-5-27(6-2)10-9-24-22(29)20-13(3)19(25-14(20)4)12-17-16-11-15(23)7-8-18(16)26-21(17)28/h7-8,11-12,25H,5-6,9-10H2,1-4H3,(H,24,29)(H,26,28)/b17-12- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 350 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of human KIT |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM4814

(CHEMBL535 | N-[2-(diethylamino)ethyl]-5-[(Z)-(5-fl...)Show SMILES CCN(CC)CCNC(=O)c1c(C)[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c1C Show InChI InChI=1S/C22H27FN4O2/c1-5-27(6-2)10-9-24-22(29)20-13(3)19(25-14(20)4)12-17-16-11-15(23)7-8-18(16)26-21(17)28/h7-8,11-12,25H,5-6,9-10H2,1-4H3,(H,24,29)(H,26,28)/b17-12- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of human ERG expressed in CHO cells by Qpatch assay |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50235922

(CHEMBL4093614)Show SMILES Cl.Cc1[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c(C)c1NC(=O)[C@H]1CCCN1 |r| Show InChI InChI=1S/C20H21FN4O2.ClH/c1-10-17(9-14-13-8-12(21)5-6-15(13)24-19(14)26)23-11(2)18(10)25-20(27)16-4-3-7-22-16;/h5-6,8-9,16,22-23H,3-4,7H2,1-2H3,(H,24,26)(H,25,27);1H/b14-9-;/t16-;/m1./s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.73E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of human ERG expressed in CHO cells by Qpatch assay |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50235922

(CHEMBL4093614)Show SMILES Cl.Cc1[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c(C)c1NC(=O)[C@H]1CCCN1 |r| Show InChI InChI=1S/C20H21FN4O2.ClH/c1-10-17(9-14-13-8-12(21)5-6-15(13)24-19(14)26)23-11(2)18(10)25-20(27)16-4-3-7-22-16;/h5-6,8-9,16,22-23H,3-4,7H2,1-2H3,(H,24,26)(H,25,27);1H/b14-9-;/t16-;/m1./s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.77E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP1A2 (unknown origin) using phenacetin as substrate preincubated for 5 mins followed by NADPH addition by LC-MS/MS analysi... |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50235922

(CHEMBL4093614)Show SMILES Cl.Cc1[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c(C)c1NC(=O)[C@H]1CCCN1 |r| Show InChI InChI=1S/C20H21FN4O2.ClH/c1-10-17(9-14-13-8-12(21)5-6-15(13)24-19(14)26)23-11(2)18(10)25-20(27)16-4-3-7-22-16;/h5-6,8-9,16,22-23H,3-4,7H2,1-2H3,(H,24,26)(H,25,27);1H/b14-9-;/t16-;/m1./s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP3A4 (unknown origin) using testosterone as substrate preincubated for 5 mins followed by NADPH addition by LC-MS/MS analy... |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50235922

(CHEMBL4093614)Show SMILES Cl.Cc1[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c(C)c1NC(=O)[C@H]1CCCN1 |r| Show InChI InChI=1S/C20H21FN4O2.ClH/c1-10-17(9-14-13-8-12(21)5-6-15(13)24-19(14)26)23-11(2)18(10)25-20(27)16-4-3-7-22-16;/h5-6,8-9,16,22-23H,3-4,7H2,1-2H3,(H,24,26)(H,25,27);1H/b14-9-;/t16-;/m1./s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP3A4 (unknown origin) using midazolam as substrate preincubated for 5 mins followed by NADPH addition by LC-MS/MS analysis |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50235922

(CHEMBL4093614)Show SMILES Cl.Cc1[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c(C)c1NC(=O)[C@H]1CCCN1 |r| Show InChI InChI=1S/C20H21FN4O2.ClH/c1-10-17(9-14-13-8-12(21)5-6-15(13)24-19(14)26)23-11(2)18(10)25-20(27)16-4-3-7-22-16;/h5-6,8-9,16,22-23H,3-4,7H2,1-2H3,(H,24,26)(H,25,27);1H/b14-9-;/t16-;/m1./s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP2C19 (unknown origin) using S-mephenytoin as substrate preincubated for 5 mins followed by NADPH addition by LC-MS/MS ana... |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50235922

(CHEMBL4093614)Show SMILES Cl.Cc1[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c(C)c1NC(=O)[C@H]1CCCN1 |r| Show InChI InChI=1S/C20H21FN4O2.ClH/c1-10-17(9-14-13-8-12(21)5-6-15(13)24-19(14)26)23-11(2)18(10)25-20(27)16-4-3-7-22-16;/h5-6,8-9,16,22-23H,3-4,7H2,1-2H3,(H,24,26)(H,25,27);1H/b14-9-;/t16-;/m1./s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP2C9 (unknown origin) using diclofenac as substrate preincubated for 5 mins followed by NADPH addition by LC-MS/MS analysi... |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50235922

(CHEMBL4093614)Show SMILES Cl.Cc1[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c(C)c1NC(=O)[C@H]1CCCN1 |r| Show InChI InChI=1S/C20H21FN4O2.ClH/c1-10-17(9-14-13-8-12(21)5-6-15(13)24-19(14)26)23-11(2)18(10)25-20(27)16-4-3-7-22-16;/h5-6,8-9,16,22-23H,3-4,7H2,1-2H3,(H,24,26)(H,25,27);1H/b14-9-;/t16-;/m1./s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP2C8 (unknown origin) using amodiaquine as substrate preincubated for 5 mins followed by NADPH addition by LC-MS/MS analys... |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2B6
(Homo sapiens (Human)) | BDBM50235922

(CHEMBL4093614)Show SMILES Cl.Cc1[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c(C)c1NC(=O)[C@H]1CCCN1 |r| Show InChI InChI=1S/C20H21FN4O2.ClH/c1-10-17(9-14-13-8-12(21)5-6-15(13)24-19(14)26)23-11(2)18(10)25-20(27)16-4-3-7-22-16;/h5-6,8-9,16,22-23H,3-4,7H2,1-2H3,(H,24,26)(H,25,27);1H/b14-9-;/t16-;/m1./s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP2B6 (unknown origin) using bupropion as substrate preincubated for 5 mins followed by NADPH addition by LC-MS/MS analysis |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50235922

(CHEMBL4093614)Show SMILES Cl.Cc1[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c(C)c1NC(=O)[C@H]1CCCN1 |r| Show InChI InChI=1S/C20H21FN4O2.ClH/c1-10-17(9-14-13-8-12(21)5-6-15(13)24-19(14)26)23-11(2)18(10)25-20(27)16-4-3-7-22-16;/h5-6,8-9,16,22-23H,3-4,7H2,1-2H3,(H,24,26)(H,25,27);1H/b14-9-;/t16-;/m1./s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP2D6 (unknown origin) using dextromethorphan as substrate preincubated for 5 mins followed by NADPH addition by LC-MS/MS a... |
Eur J Med Chem 127: 72-86 (2017)
Article DOI: 10.1016/j.ejmech.2016.12.038
BindingDB Entry DOI: 10.7270/Q2N58PMM |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data