 Found 127 hits Enz. Inhib. hit(s) with all data for entry = 50035363
Found 127 hits Enz. Inhib. hit(s) with all data for entry = 50035363 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10870
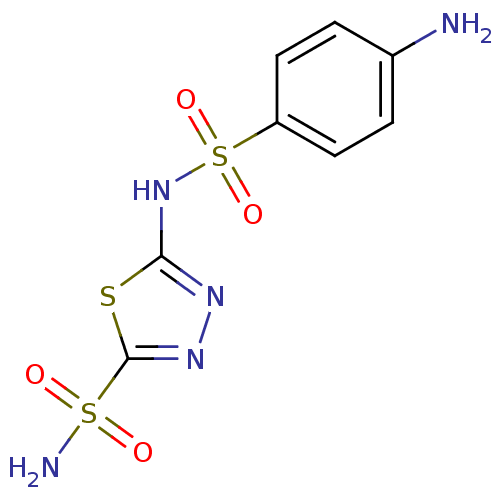
(2-N-(4-aminobenzene)-1,3,4-thiadiazole-2,5-disulfo...)Show InChI InChI=1S/C8H9N5O4S3/c9-5-1-3-6(4-2-5)20(16,17)13-7-11-12-8(18-7)19(10,14)15/h1-4H,9H2,(H,11,13)(H2,10,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM10885
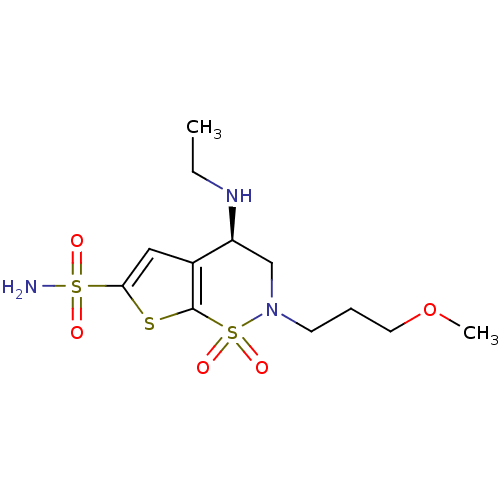
((4R)-4-(ethylamino)-2-(3-methoxypropyl)-1,1-dioxo-...)Show SMILES CCN[C@H]1CN(CCCOC)S(=O)(=O)c2sc(cc12)S(N)(=O)=O |r| Show InChI InChI=1S/C12H21N3O5S3/c1-3-14-10-8-15(5-4-6-20-2)23(18,19)12-9(10)7-11(21-12)22(13,16)17/h7,10,14H,3-6,8H2,1-2H3,(H2,13,16,17)/t10-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme I by esterase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 4
(Bos taurus (bovine)) | BDBM10870

(2-N-(4-aminobenzene)-1,3,4-thiadiazole-2,5-disulfo...)Show InChI InChI=1S/C8H9N5O4S3/c9-5-1-3-6(4-2-5)20(16,17)13-7-11-12-8(18-7)19(10,14)15/h1-4H,9H2,(H,11,13)(H2,10,14,15) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against bovine carbonic anhydrase isozyme IV isolated from bovine lung microsomes, by esterase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM10870

(2-N-(4-aminobenzene)-1,3,4-thiadiazole-2,5-disulfo...)Show InChI InChI=1S/C8H9N5O4S3/c9-5-1-3-6(4-2-5)20(16,17)13-7-11-12-8(18-7)19(10,14)15/h1-4H,9H2,(H,11,13)(H2,10,14,15) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme I by esterase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10882
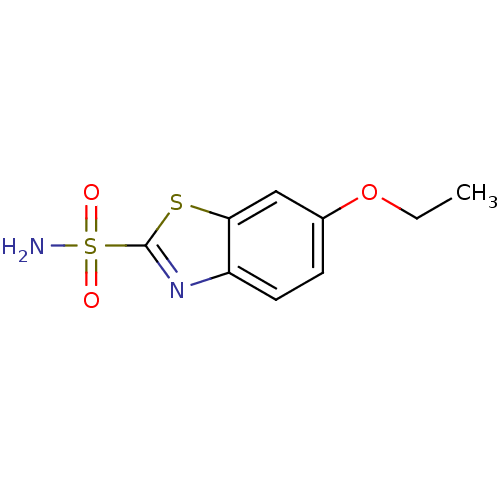
(6-ethoxy-1,3-benzothiazole-2-sulfonamide | CHEMBL1...)Show InChI InChI=1S/C9H10N2O3S2/c1-2-14-6-3-4-7-8(5-6)15-9(11-7)16(10,12)13/h3-5H,2H2,1H3,(H2,10,12,13) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10874
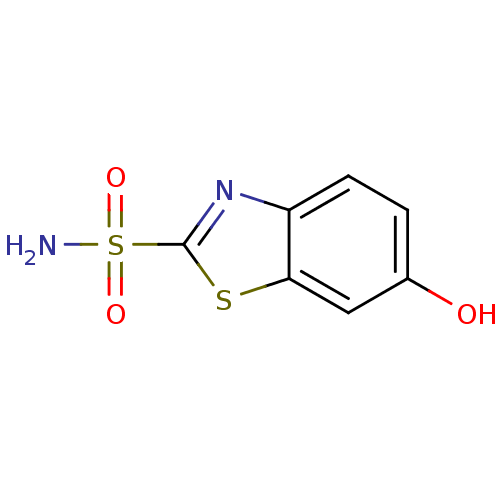
(6-hydroxy-1,3-benzothiazole-2-sulfonamide | CHEMBL...)Show InChI InChI=1S/C7H6N2O3S2/c8-14(11,12)7-9-5-2-1-4(10)3-6(5)13-7/h1-3,10H,(H2,8,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10884

((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...)Show SMILES CCN[C@H]1C[C@H](C)S(=O)(=O)c2sc(cc12)S(N)(=O)=O |r| Show InChI InChI=1S/C10H16N2O4S3/c1-3-12-8-4-6(2)18(13,14)10-7(8)5-9(17-10)19(11,15)16/h5-6,8,12H,3-4H2,1-2H3,(H2,11,15,16)/t6-,8-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10880

(AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...)Show InChI InChI=1S/C4H6N4O3S2/c1-2(9)6-3-7-8-4(12-3)13(5,10)11/h1H3,(H2,5,10,11)(H,6,7,9) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10873
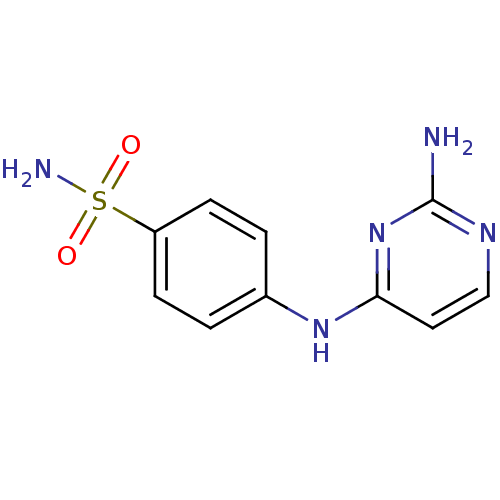
(4-[(2-aminopyrimidin-4-yl)amino]benzene-1-sulfonam...)Show InChI InChI=1S/C10H11N5O2S/c11-10-13-6-5-9(15-10)14-7-1-3-8(4-2-7)18(12,16)17/h1-6H,(H2,12,16,17)(H3,11,13,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Carbonic anhydrase 4
(Bos taurus (bovine)) | BDBM10882

(6-ethoxy-1,3-benzothiazole-2-sulfonamide | CHEMBL1...)Show InChI InChI=1S/C9H10N2O3S2/c1-2-14-6-3-4-7-8(5-6)15-9(11-7)16(10,12)13/h3-5H,2H2,1H3,(H2,10,12,13) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against bovine carbonic anhydrase isozyme IV isolated from bovine lung microsomes, by esterase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10881
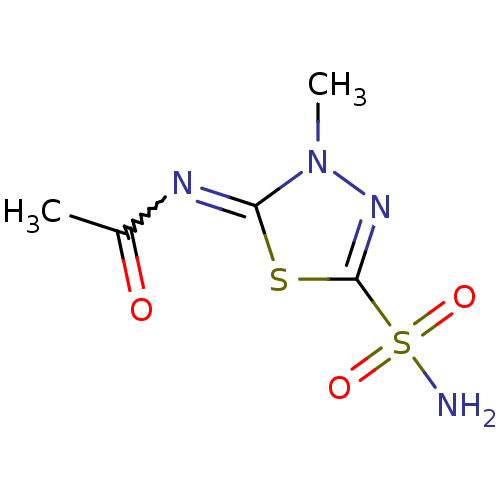
(CHEMBL288100 | MZA3 | Methazolamide | Methazolamid...)Show InChI InChI=1S/C5H8N4O3S2/c1-3(10)7-4-9(2)8-5(13-4)14(6,11)12/h1-2H3,(H2,6,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10874

(6-hydroxy-1,3-benzothiazole-2-sulfonamide | CHEMBL...)Show InChI InChI=1S/C7H6N2O3S2/c8-14(11,12)7-9-5-2-1-4(10)3-6(5)13-7/h1-3,10H,(H2,8,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10873

(4-[(2-aminopyrimidin-4-yl)amino]benzene-1-sulfonam...)Show InChI InChI=1S/C10H11N5O2S/c11-10-13-6-5-9(15-10)14-7-1-3-8(4-2-7)18(12,16)17/h1-6H,(H2,12,16,17)(H3,11,13,14,15) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 4
(Bos taurus (bovine)) | BDBM10874

(6-hydroxy-1,3-benzothiazole-2-sulfonamide | CHEMBL...)Show InChI InChI=1S/C7H6N2O3S2/c8-14(11,12)7-9-5-2-1-4(10)3-6(5)13-7/h1-3,10H,(H2,8,11,12) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against bovine carbonic anhydrase isozyme IV isolated from bovine lung microsomes, by esterase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10869
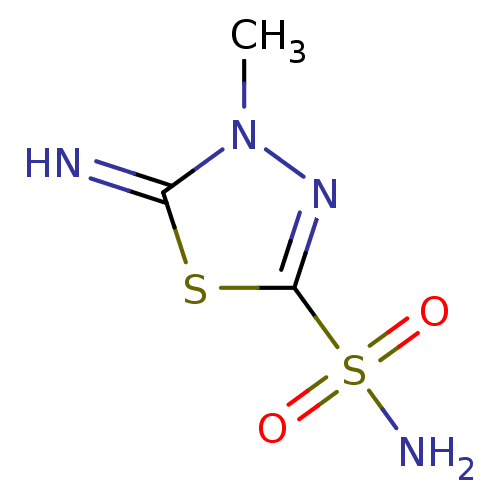
(5-imino-4-methyl-4,5-dihydro-1,3,4-thiadiazole-2-s...)Show InChI InChI=1S/C3H6N4O2S2/c1-7-2(4)10-3(6-7)11(5,8)9/h4H,1H3,(H2,5,8,9) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50125191
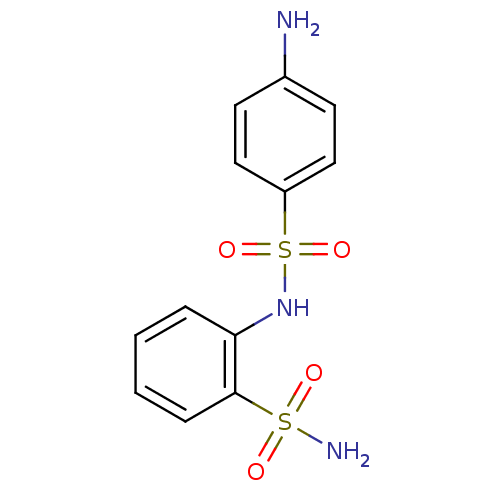
(2-(4-aminophenylsulfonamido)-1-benzenesulfonamide ...)Show InChI InChI=1S/C12H13N3O4S2/c13-9-5-7-10(8-6-9)21(18,19)15-11-3-1-2-4-12(11)20(14,16)17/h1-8,15H,13H2,(H2,14,16,17) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10876
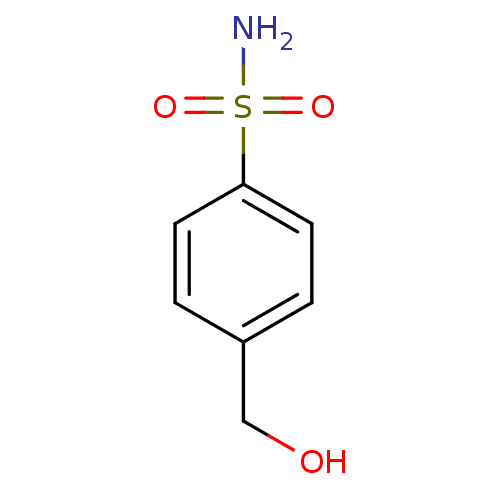
(4-(hydroxymethyl)benzene-1-sulfonamide | CHEMBL691...)Show InChI InChI=1S/C7H9NO3S/c8-12(10,11)7-3-1-6(5-9)2-4-7/h1-4,9H,5H2,(H2,8,10,11) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10877
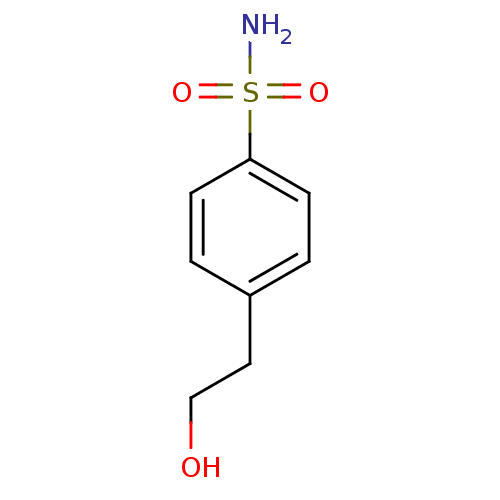
(4-(2-hydroxyethyl)benzene-1-sulfonamide | CHEMBL67...)Show InChI InChI=1S/C8H11NO3S/c9-13(11,12)8-3-1-7(2-4-8)5-6-10/h1-4,10H,5-6H2,(H2,9,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10872
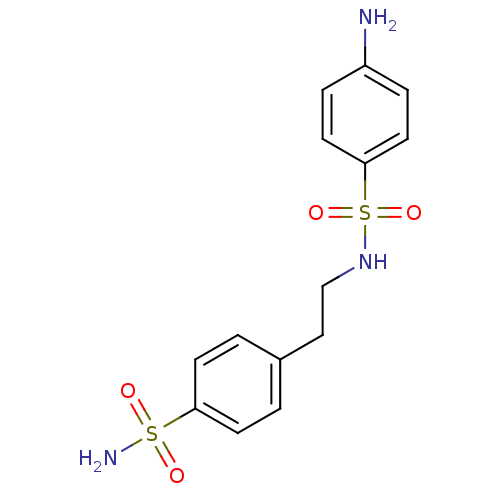
(4-amino-N-[2-(4-sulfamoylphenyl)ethyl]benzene-1-su...)Show SMILES Nc1ccc(cc1)S(=O)(=O)NCCc1ccc(cc1)S(N)(=O)=O Show InChI InChI=1S/C14H17N3O4S2/c15-12-3-7-14(8-4-12)23(20,21)17-10-9-11-1-5-13(6-2-11)22(16,18)19/h1-8,17H,9-10,15H2,(H2,16,18,19) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10866
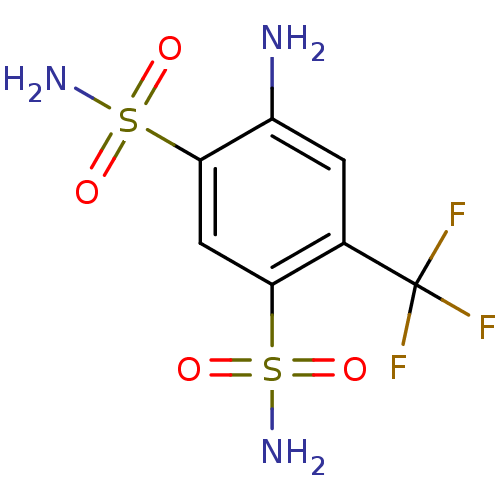
(4-amino-6-(trifluoromethyl)benzene-1,3-disulfonami...)Show InChI InChI=1S/C7H8F3N3O4S2/c8-7(9,10)3-1-4(11)6(19(13,16)17)2-5(3)18(12,14)15/h1-2H,11H2,(H2,12,14,15)(H2,13,16,17) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
| PubMed
| 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10880

(AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...)Show InChI InChI=1S/C4H6N4O3S2/c1-2(9)6-3-7-8-4(12-3)13(5,10)11/h1H3,(H2,5,10,11)(H,6,7,9) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM10882

(6-ethoxy-1,3-benzothiazole-2-sulfonamide | CHEMBL1...)Show InChI InChI=1S/C9H10N2O3S2/c1-2-14-6-3-4-7-8(5-6)15-9(11-7)16(10,12)13/h3-5H,2H2,1H3,(H2,10,12,13) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme I by esterase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM4703
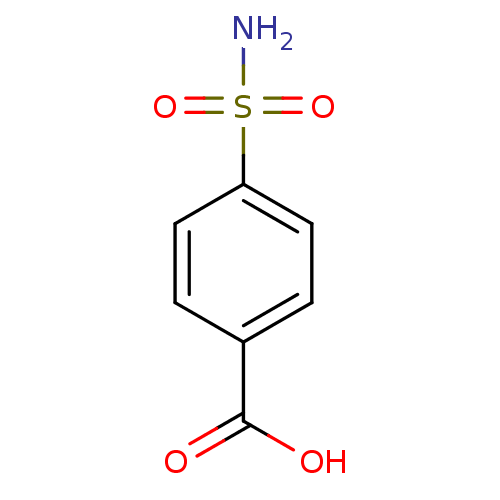
(4-carboxybenzenesulfonamide 1 | 4-sulfamoylbenzoic...)Show InChI InChI=1S/C7H7NO4S/c8-13(11,12)6-3-1-5(2-4-6)7(9)10/h1-4H,(H,9,10)(H2,8,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10881

(CHEMBL288100 | MZA3 | Methazolamide | Methazolamid...)Show InChI InChI=1S/C5H8N4O3S2/c1-3(10)7-4-9(2)8-5(13-4)14(6,11)12/h1-2H3,(H2,6,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10869

(5-imino-4-methyl-4,5-dihydro-1,3,4-thiadiazole-2-s...)Show InChI InChI=1S/C3H6N4O2S2/c1-7-2(4)10-3(6-7)11(5,8)9/h4H,1H3,(H2,5,8,9) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50125192
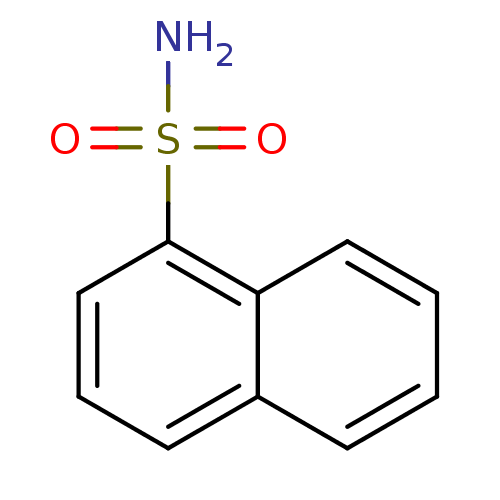
(CHEMBL6648 | Naphthalene-1-sulfonic acid amide | n...)Show InChI InChI=1S/C10H9NO2S/c11-14(12,13)10-7-3-5-8-4-1-2-6-9(8)10/h1-7H,(H2,11,12,13) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10872

(4-amino-N-[2-(4-sulfamoylphenyl)ethyl]benzene-1-su...)Show SMILES Nc1ccc(cc1)S(=O)(=O)NCCc1ccc(cc1)S(N)(=O)=O Show InChI InChI=1S/C14H17N3O4S2/c15-12-3-7-14(8-4-12)23(20,21)17-10-9-11-1-5-13(6-2-11)22(16,18)19/h1-8,17H,9-10,15H2,(H2,16,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10871
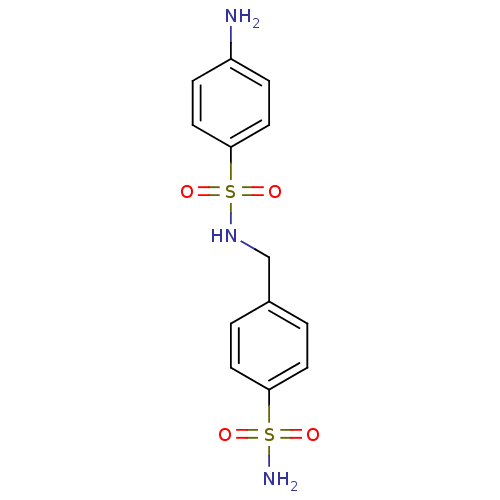
(4-amino-N-[(4-sulfamoylphenyl)methyl]benzene-1-sul...)Show InChI InChI=1S/C13H15N3O4S2/c14-11-3-7-13(8-4-11)22(19,20)16-9-10-1-5-12(6-2-10)21(15,17)18/h1-8,16H,9,14H2,(H2,15,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 31 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50125190
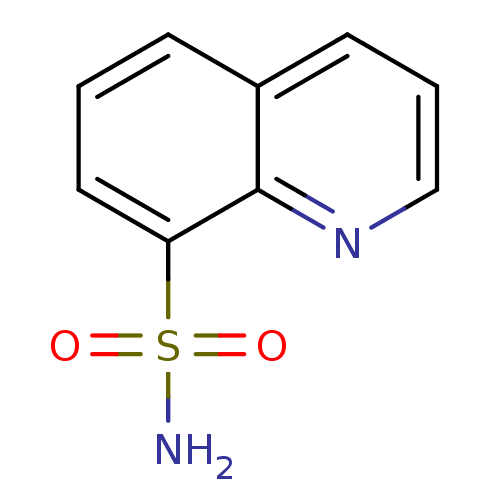
(CHEMBL268809 | Quinoline-8-sulfonic acid amide | q...)Show InChI InChI=1S/C9H8N2O2S/c10-14(12,13)8-5-1-3-7-4-2-6-11-9(7)8/h1-6H,(H2,10,12,13) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
| PubMed
| 32 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10855
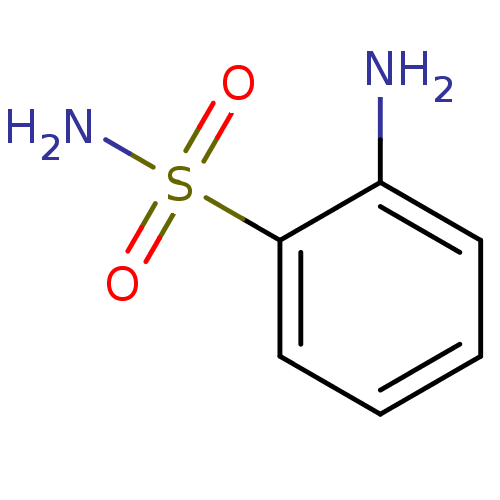
(2-aminobenzene-1-sulfonamide | CHEMBL6705 | US1017...)Show InChI InChI=1S/C6H8N2O2S/c7-5-3-1-2-4-6(5)11(8,9)10/h1-4H,7H2,(H2,8,9,10) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| PubMed
| 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10871

(4-amino-N-[(4-sulfamoylphenyl)methyl]benzene-1-sul...)Show InChI InChI=1S/C13H15N3O4S2/c14-11-3-7-13(8-4-11)22(19,20)16-9-10-1-5-12(6-2-10)21(15,17)18/h1-8,16H,9,14H2,(H2,15,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10861
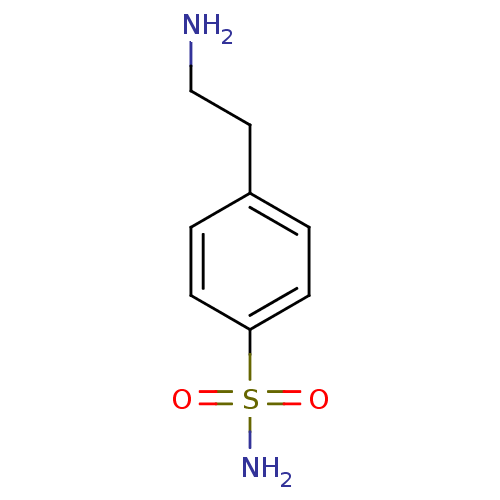
(4-(2-aminoethyl)benzene-1-sulfonamide | CHEMBL7087...)Show InChI InChI=1S/C8H12N2O2S/c9-6-5-7-1-3-8(4-2-7)13(10,11)12/h1-4H,5-6,9H2,(H2,10,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10882

(6-ethoxy-1,3-benzothiazole-2-sulfonamide | CHEMBL1...)Show InChI InChI=1S/C9H10N2O3S2/c1-2-14-6-3-4-7-8(5-6)15-9(11-7)16(10,12)13/h3-5H,2H2,1H3,(H2,10,12,13) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 34 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50080733
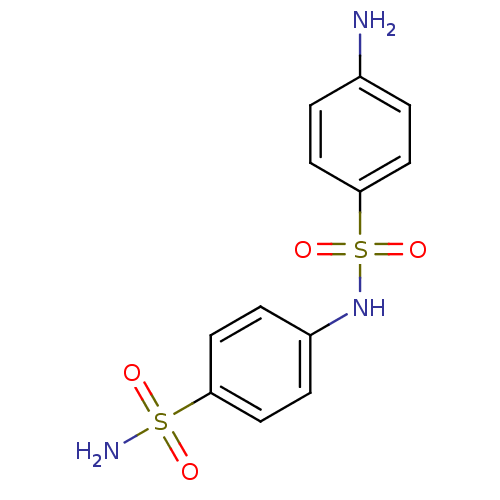
(4-(4-aminophenylsulfonamido)-1-benzenesulfonamide ...)Show InChI InChI=1S/C12H13N3O4S2/c13-9-1-5-12(6-2-9)21(18,19)15-10-3-7-11(8-4-10)20(14,16)17/h1-8,15H,13H2,(H2,14,16,17) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 34 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 4
(Bos taurus (bovine)) | BDBM10881

(CHEMBL288100 | MZA3 | Methazolamide | Methazolamid...)Show InChI InChI=1S/C5H8N4O3S2/c1-3(10)7-4-9(2)8-5(13-4)14(6,11)12/h1-2H3,(H2,6,11,12) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 36 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against bovine carbonic anhydrase isozyme IV isolated from bovine lung microsomes, by esterase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10885

((4R)-4-(ethylamino)-2-(3-methoxypropyl)-1,1-dioxo-...)Show SMILES CCN[C@H]1CN(CCCOC)S(=O)(=O)c2sc(cc12)S(N)(=O)=O |r| Show InChI InChI=1S/C12H21N3O5S3/c1-3-14-10-8-15(5-4-6-20-2)23(18,19)12-9(10)7-11(21-12)22(13,16)17/h7,10,14H,3-6,8H2,1-2H3,(H2,13,16,17)/t10-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10870

(2-N-(4-aminobenzene)-1,3,4-thiadiazole-2,5-disulfo...)Show InChI InChI=1S/C8H9N5O4S3/c9-5-1-3-6(4-2-5)20(16,17)13-7-11-12-8(18-7)19(10,14)15/h1-4H,9H2,(H,11,13)(H2,10,14,15) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 38 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10883
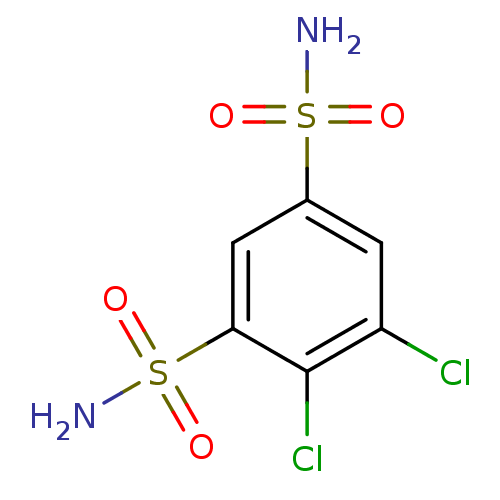
(4,5-dichlorobenzene-1,3-disulfonamide | CHEMBL17 |...)Show InChI InChI=1S/C6H6Cl2N2O4S2/c7-4-1-3(15(9,11)12)2-5(6(4)8)16(10,13)14/h1-2H,(H2,9,11,12)(H2,10,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
| DrugBank
MMDB
PDB
PubMed
| 38 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10867
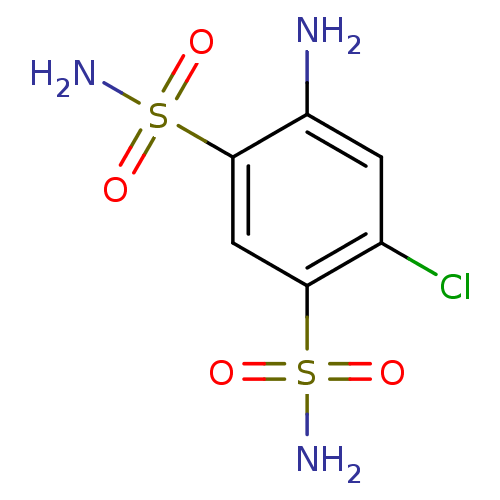
(4-amino-6-chlorobenzene-1,3-disulfonamide | CHEMBL...)Show InChI InChI=1S/C6H8ClN3O4S2/c7-3-1-4(8)6(16(10,13)14)2-5(3)15(9,11)12/h1-2H,8H2,(H2,9,11,12)(H2,10,13,14) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 39 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM4703

(4-carboxybenzenesulfonamide 1 | 4-sulfamoylbenzoic...)Show InChI InChI=1S/C7H7NO4S/c8-13(11,12)6-3-1-5(2-4-6)7(9)10/h1-4H,(H,9,10)(H2,8,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10864
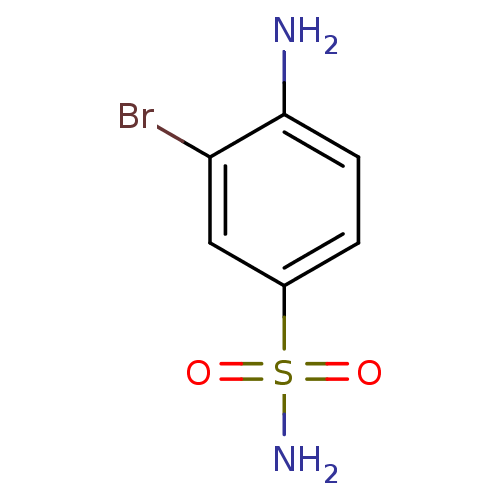
(4-Amino-3-bromobenzenesulfonamide | 4-amino-3-brom...)Show InChI InChI=1S/C6H7BrN2O2S/c7-5-3-4(12(9,10)11)1-2-6(5)8/h1-3H,8H2,(H2,9,10,11) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10868
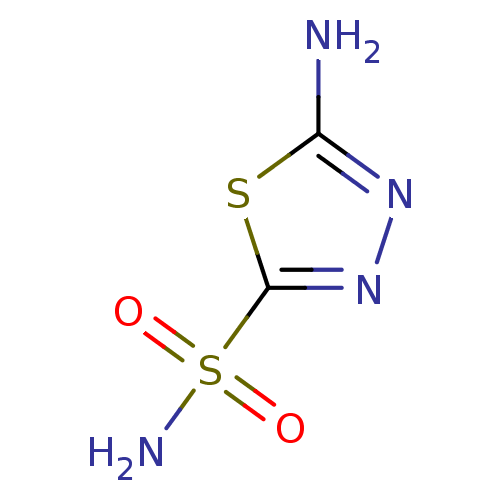
(1,3,4-Thiadiazole-2-sulfonamide, 6 | 1,3,4-thiadia...)Show InChI InChI=1S/C2H4N4O2S2/c3-1-5-6-2(9-1)10(4,7)8/h(H2,3,5)(H2,4,7,8) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
| PubMed
| 41 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 4
(Bos taurus (bovine)) | BDBM10884

((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...)Show SMILES CCN[C@H]1C[C@H](C)S(=O)(=O)c2sc(cc12)S(N)(=O)=O |r| Show InChI InChI=1S/C10H16N2O4S3/c1-3-12-8-4-6(2)18(13,14)10-7(8)5-9(17-10)19(11,15)16/h5-6,8,12H,3-4H2,1-2H3,(H2,11,15,16)/t6-,8-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 43 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against bovine carbonic anhydrase isozyme IV isolated from bovine lung microsomes, by esterase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 4
(Bos taurus (bovine)) | BDBM10885

((4R)-4-(ethylamino)-2-(3-methoxypropyl)-1,1-dioxo-...)Show SMILES CCN[C@H]1CN(CCCOC)S(=O)(=O)c2sc(cc12)S(N)(=O)=O |r| Show InChI InChI=1S/C12H21N3O5S3/c1-3-14-10-8-15(5-4-6-20-2)23(18,19)12-9(10)7-11(21-12)22(13,16)17/h7,10,14H,3-6,8H2,1-2H3,(H2,13,16,17)/t10-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 45 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against bovine carbonic anhydrase isozyme IV isolated from bovine lung microsomes, by esterase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50080733

(4-(4-aminophenylsulfonamido)-1-benzenesulfonamide ...)Show InChI InChI=1S/C12H13N3O4S2/c13-9-1-5-12(6-2-9)21(18,19)15-10-3-7-11(8-4-10)20(14,16)17/h1-8,15H,13H2,(H2,14,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 46 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM10881

(CHEMBL288100 | MZA3 | Methazolamide | Methazolamid...)Show InChI InChI=1S/C5H8N4O3S2/c1-3(10)7-4-9(2)8-5(13-4)14(6,11)12/h1-2H3,(H2,6,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme I by esterase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50125191

(2-(4-aminophenylsulfonamido)-1-benzenesulfonamide ...)Show InChI InChI=1S/C12H13N3O4S2/c13-9-5-7-10(8-6-9)21(18,19)15-11-3-1-2-4-12(11)20(14,16)17/h1-8,15H,13H2,(H2,14,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase II |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10883

(4,5-dichlorobenzene-1,3-disulfonamide | CHEMBL17 |...)Show InChI InChI=1S/C6H6Cl2N2O4S2/c7-4-1-3(15(9,11)12)2-5(6(4)8)16(10,13)14/h1-2H,(H2,9,11,12)(H2,10,13,14) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
| PubMed
| 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10884

((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...)Show SMILES CCN[C@H]1C[C@H](C)S(=O)(=O)c2sc(cc12)S(N)(=O)=O |r| Show InChI InChI=1S/C10H16N2O4S3/c1-3-12-8-4-6(2)18(13,14)10-7(8)5-9(17-10)19(11,15)16/h5-6,8,12H,3-4H2,1-2H3,(H2,11,15,16)/t6-,8-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 52 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme IX, by CO2 hydrase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM10874

(6-hydroxy-1,3-benzothiazole-2-sulfonamide | CHEMBL...)Show InChI InChI=1S/C7H6N2O3S2/c8-14(11,12)7-9-5-2-1-4(10)3-6(5)13-7/h1-3,10H,(H2,8,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze
Curated by ChEMBL
| Assay Description
Inhibitory activity against human cloned carbonic anhydrase isozyme I by esterase assay method. |
Bioorg Med Chem Lett 13: 1005-9 (2003)
BindingDB Entry DOI: 10.7270/Q2PV6KXV |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data