

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50203248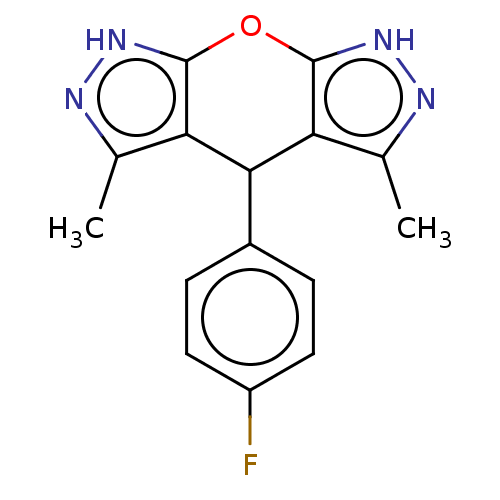 (CHEMBL3930308) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology Curated by ChEMBL | Assay Description Inhibition of N-terminus 6xHis-tagged human IDO1 expressed in Escherichia coli M15 using L-tryptophan as substrate preincubated for 1 hr measured aft... | ACS Med Chem Lett 7: 1167-1172 (2016) Article DOI: 10.1021/acsmedchemlett.6b00359 BindingDB Entry DOI: 10.7270/Q2TF009K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50300305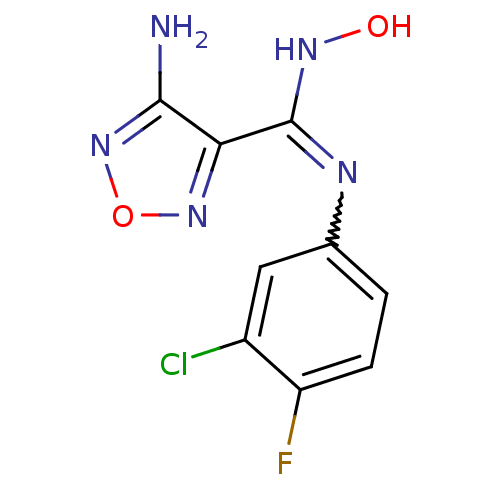 (4-Amino-N-(3-chloro-4-fluorophenyl)-N'-hydroxy-1,2...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology Curated by ChEMBL | Assay Description Inhibition of N-terminus 6xHis-tagged human IDO1 expressed in Escherichia coli M15 using L-tryptophan as substrate preincubated for 1 hr measured aft... | ACS Med Chem Lett 7: 1167-1172 (2016) Article DOI: 10.1021/acsmedchemlett.6b00359 BindingDB Entry DOI: 10.7270/Q2TF009K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50203260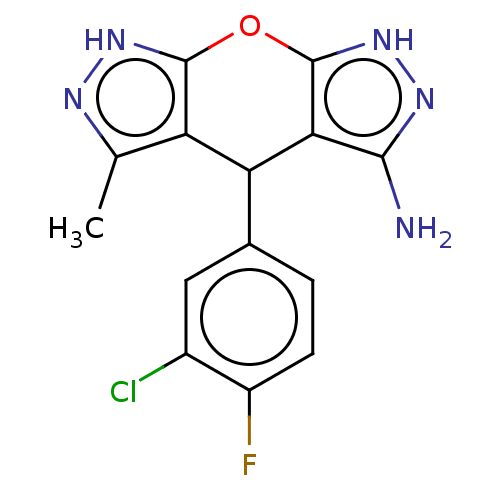 (CHEMBL3974564) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology Curated by ChEMBL | Assay Description Inhibition of N-terminus 6xHis-tagged human IDO1 expressed in Escherichia coli M15 using L-tryptophan as substrate preincubated for 1 hr measured aft... | ACS Med Chem Lett 7: 1167-1172 (2016) Article DOI: 10.1021/acsmedchemlett.6b00359 BindingDB Entry DOI: 10.7270/Q2TF009K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50203249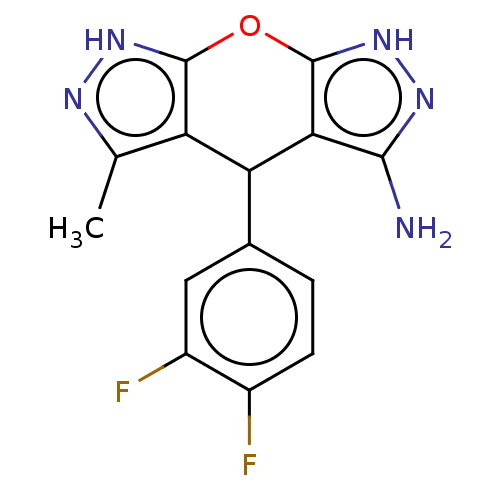 (CHEMBL3945146) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology Curated by ChEMBL | Assay Description Inhibition of N-terminus 6xHis-tagged human IDO1 expressed in Escherichia coli M15 using L-tryptophan as substrate preincubated for 1 hr measured aft... | ACS Med Chem Lett 7: 1167-1172 (2016) Article DOI: 10.1021/acsmedchemlett.6b00359 BindingDB Entry DOI: 10.7270/Q2TF009K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50203268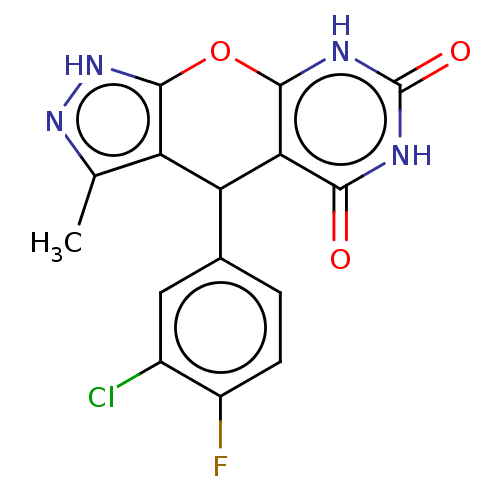 (CHEMBL3909480) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology Curated by ChEMBL | Assay Description Inhibition of N-terminus 6xHis-tagged human IDO1 expressed in Escherichia coli M15 using L-tryptophan as substrate preincubated for 1 hr measured aft... | ACS Med Chem Lett 7: 1167-1172 (2016) Article DOI: 10.1021/acsmedchemlett.6b00359 BindingDB Entry DOI: 10.7270/Q2TF009K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50203255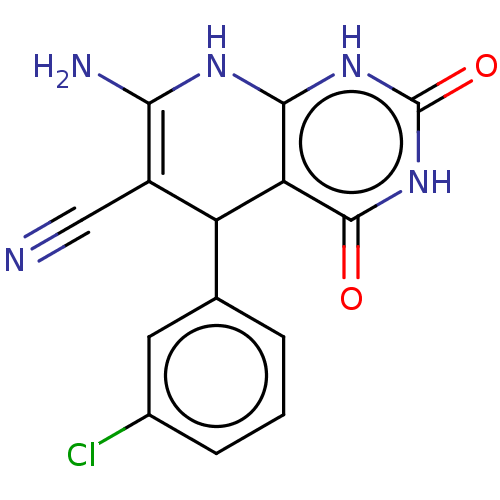 (CHEMBL3913975) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology Curated by ChEMBL | Assay Description Inhibition of N-terminus 6xHis-tagged human IDO1 expressed in Escherichia coli M15 using L-tryptophan as substrate preincubated for 1 hr measured aft... | ACS Med Chem Lett 7: 1167-1172 (2016) Article DOI: 10.1021/acsmedchemlett.6b00359 BindingDB Entry DOI: 10.7270/Q2TF009K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50241727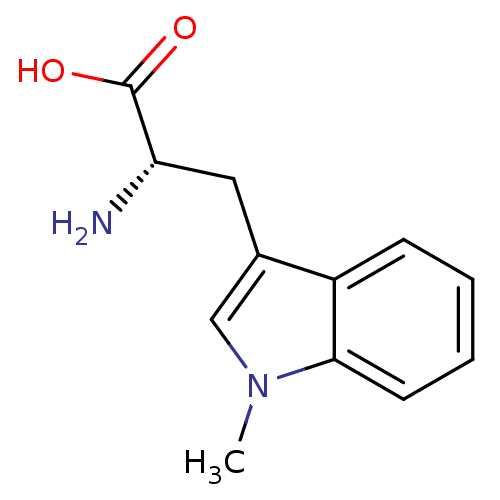 ((S)-2-amino-3-(1-methyl-1H-indol-3-yl)propanoic ac...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology Curated by ChEMBL | Assay Description Inhibition of N-terminus 6xHis-tagged human IDO1 expressed in Escherichia coli M15 using L-tryptophan as substrate preincubated for 1 hr measured aft... | ACS Med Chem Lett 7: 1167-1172 (2016) Article DOI: 10.1021/acsmedchemlett.6b00359 BindingDB Entry DOI: 10.7270/Q2TF009K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50203269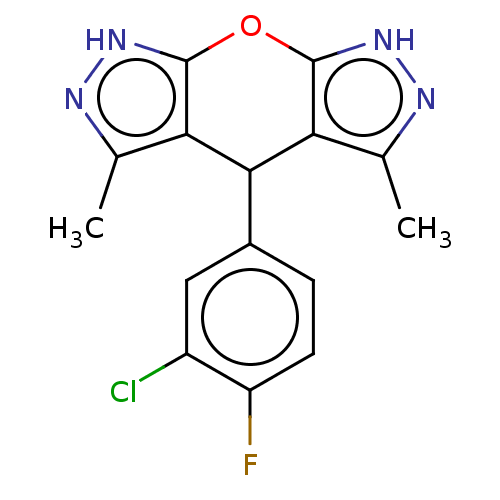 (CHEMBL3913434) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology Curated by ChEMBL | Assay Description Inhibition of N-terminus 6xHis-tagged human IDO1 expressed in Escherichia coli M15 using L-tryptophan as substrate preincubated for 1 hr measured aft... | ACS Med Chem Lett 7: 1167-1172 (2016) Article DOI: 10.1021/acsmedchemlett.6b00359 BindingDB Entry DOI: 10.7270/Q2TF009K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50203280 (CHEMBL3966057) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology Curated by ChEMBL | Assay Description Inhibition of N-terminus 6xHis-tagged human IDO1 expressed in Escherichia coli M15 using L-tryptophan as substrate preincubated for 1 hr measured aft... | ACS Med Chem Lett 7: 1167-1172 (2016) Article DOI: 10.1021/acsmedchemlett.6b00359 BindingDB Entry DOI: 10.7270/Q2TF009K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50203256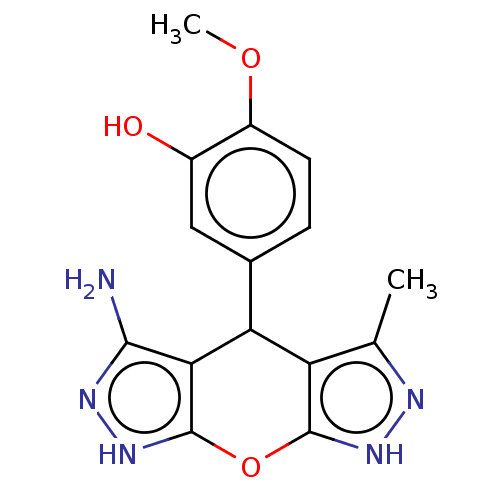 (CHEMBL3936225) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology Curated by ChEMBL | Assay Description Inhibition of N-terminus 6xHis-tagged human IDO1 expressed in Escherichia coli M15 using L-tryptophan as substrate preincubated for 1 hr measured aft... | ACS Med Chem Lett 7: 1167-1172 (2016) Article DOI: 10.1021/acsmedchemlett.6b00359 BindingDB Entry DOI: 10.7270/Q2TF009K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50203279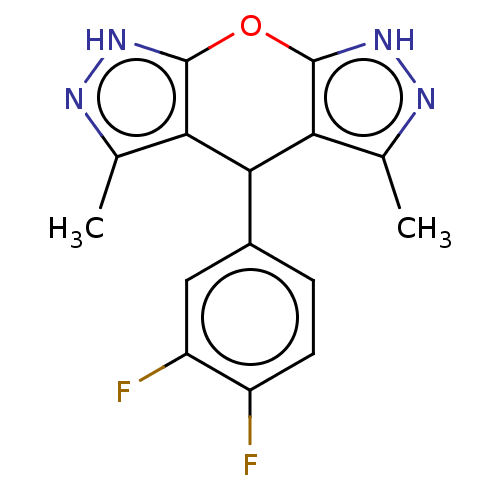 (CHEMBL3940383) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology Curated by ChEMBL | Assay Description Inhibition of N-terminus 6xHis-tagged human IDO1 expressed in Escherichia coli M15 using L-tryptophan as substrate preincubated for 1 hr measured aft... | ACS Med Chem Lett 7: 1167-1172 (2016) Article DOI: 10.1021/acsmedchemlett.6b00359 BindingDB Entry DOI: 10.7270/Q2TF009K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50203275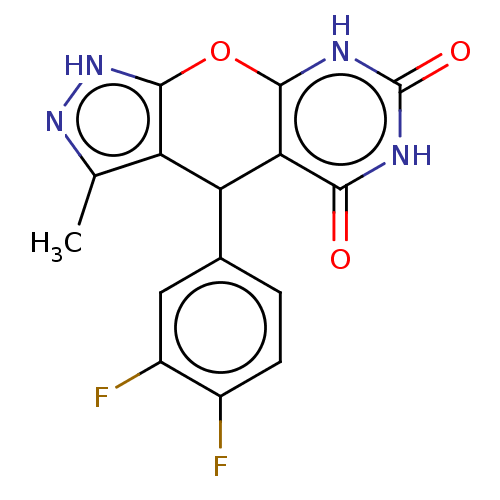 (CHEMBL3971417) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology Curated by ChEMBL | Assay Description Inhibition of N-terminus 6xHis-tagged human IDO1 expressed in Escherichia coli M15 using L-tryptophan as substrate preincubated for 1 hr measured aft... | ACS Med Chem Lett 7: 1167-1172 (2016) Article DOI: 10.1021/acsmedchemlett.6b00359 BindingDB Entry DOI: 10.7270/Q2TF009K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50203263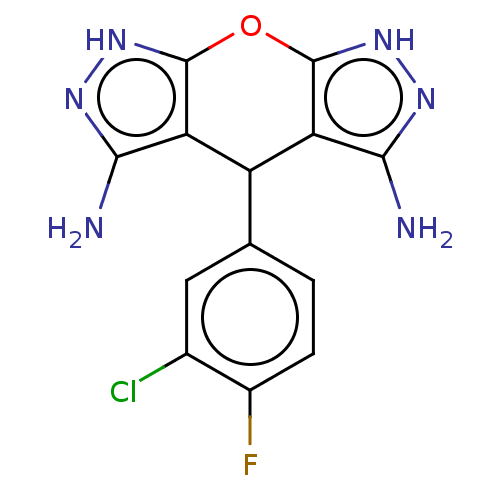 (CHEMBL3927265) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology Curated by ChEMBL | Assay Description Inhibition of N-terminus 6xHis-tagged human IDO1 expressed in Escherichia coli M15 using L-tryptophan as substrate preincubated for 1 hr measured aft... | ACS Med Chem Lett 7: 1167-1172 (2016) Article DOI: 10.1021/acsmedchemlett.6b00359 BindingDB Entry DOI: 10.7270/Q2TF009K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM50203281 (CHEMBL3971895) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian Institute of Technology Curated by ChEMBL | Assay Description Inhibition of N-terminus 6xHis-tagged human IDO1 expressed in Escherichia coli M15 using L-tryptophan as substrate preincubated for 1 hr measured aft... | ACS Med Chem Lett 7: 1167-1172 (2016) Article DOI: 10.1021/acsmedchemlett.6b00359 BindingDB Entry DOI: 10.7270/Q2TF009K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||