

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181669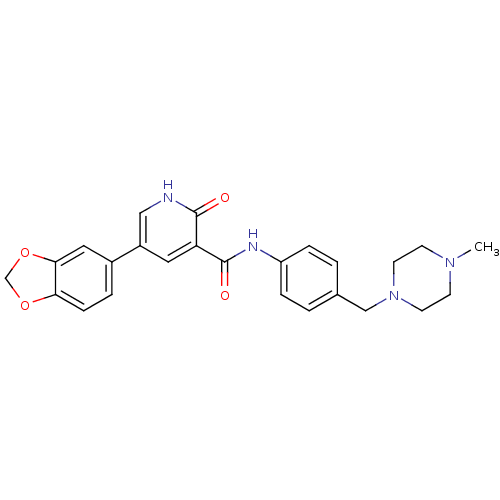 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181675 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181669 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181670 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 8.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181675 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 9.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181672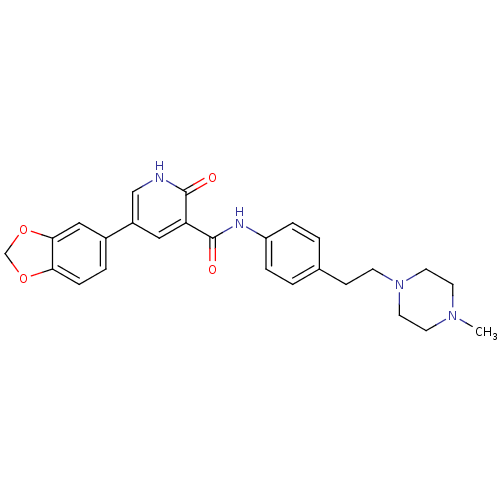 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181670 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181674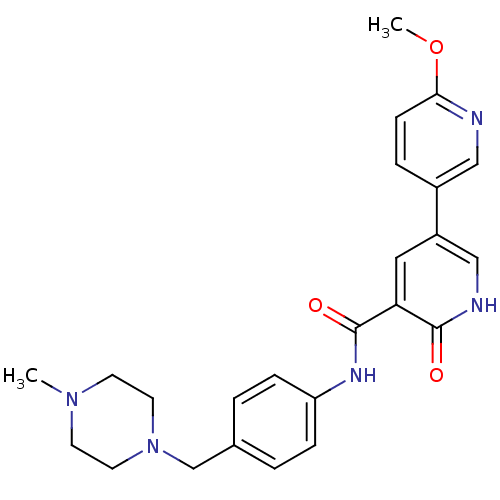 (6'-methoxy-6-oxo-1,6-dihydro-[3,3']bipyridinyl-5-c...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181672 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor/Nucleophosmin (Homo sapiens (Human)) | BDBM50181670 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Antiproliferative activity against human NPM-ALK positive anaplastic large cell lymphoma karpas299 cell line | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181671 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Breakpoint cluster region protein/Tyrosine-protein kinase ABL1 (Homo sapiens (Human)) | BDBM50181670 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Antiproliferative activity against human BCR-ABL positive chronic myeloid leukemia K562 cell line | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181679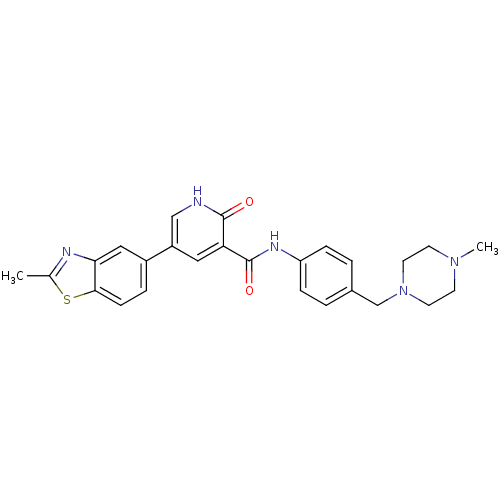 (5-(2-methyl-benzothiazol-5-yl)-2-oxo-1,2-dihydro-p...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor-type tyrosine-protein kinase FLT3 (Homo sapiens (Human)) | BDBM50181672 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against Flt3 | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor-type tyrosine-protein kinase FLT3 (Homo sapiens (Human)) | BDBM50181670 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against Flt3 | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181682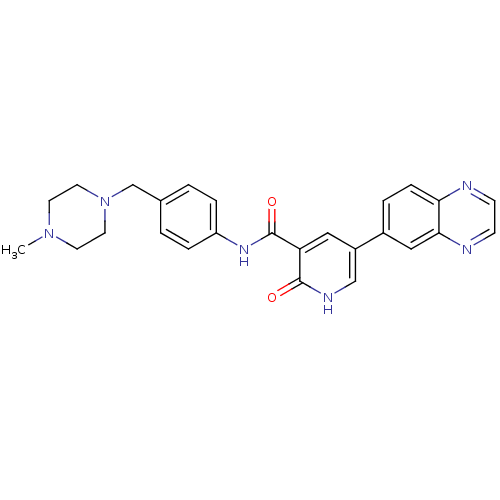 (2-oxo-5-quinoxalin-6-yl-1,2-dihydro-pyridine-3-car...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor/Nucleophosmin (Homo sapiens (Human)) | BDBM50181670 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Antiproliferative activity against murine BaF3 cell line expressing NPM-ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181682 (2-oxo-5-quinoxalin-6-yl-1,2-dihydro-pyridine-3-car...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin-like growth factor 1 receptor (Homo sapiens (Human)) | BDBM50181672 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IGF1R | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181673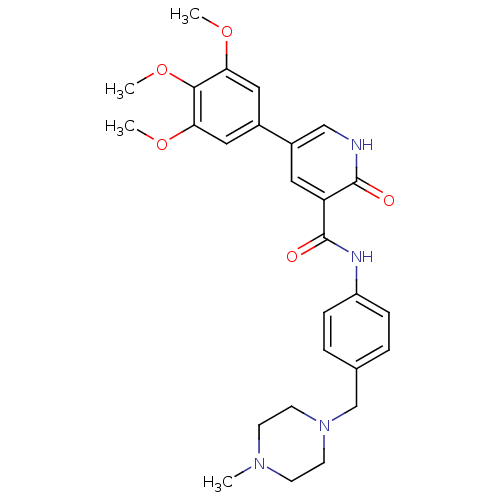 (5-(3,4,5-trimethoxyphenyl)-2-oxo-1,2-dihydro-pyrid...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor/Nucleophosmin (Homo sapiens (Human)) | BDBM50181672 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Antiproliferative activity against human NPM-ALK positive anaplastic large cell lymphoma karpas299 cell line | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor/Nucleophosmin (Homo sapiens (Human)) | BDBM50181675 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 9.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Antiproliferative activity against human NPM-ALK positive anaplastic large cell lymphoma karpas299 cell line | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Breakpoint cluster region protein/Tyrosine-protein kinase ABL1 (Homo sapiens (Human)) | BDBM50181672 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 9.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Antiproliferative activity against human BCR-ABL positive chronic myeloid leukemia K562 cell line | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor/Nucleophosmin (Homo sapiens (Human)) | BDBM50181669 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.12E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Antiproliferative activity against human NPM-ALK positive anaplastic large cell lymphoma karpas299 cell line | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Breakpoint cluster region protein/Tyrosine-protein kinase ABL1 (Homo sapiens (Human)) | BDBM50181669 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.29E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Antiproliferative activity against human BCR-ABL positive chronic myeloid leukemia K562 cell line | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor/Nucleophosmin (Homo sapiens (Human)) | BDBM50181675 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.37E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Antiproliferative activity against murine BaF3 cell line expressing NPM-ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181674 (6'-methoxy-6-oxo-1,6-dihydro-[3,3']bipyridinyl-5-c...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.38E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181683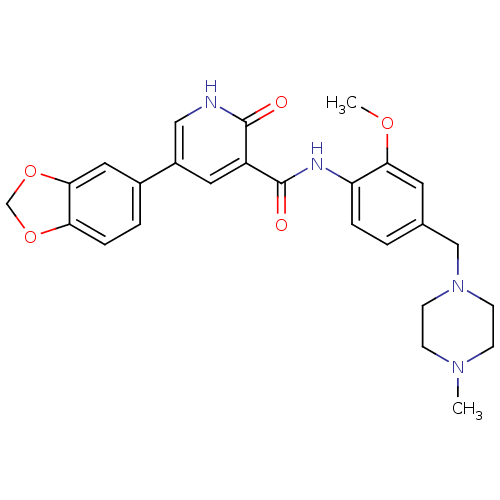 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.38E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181676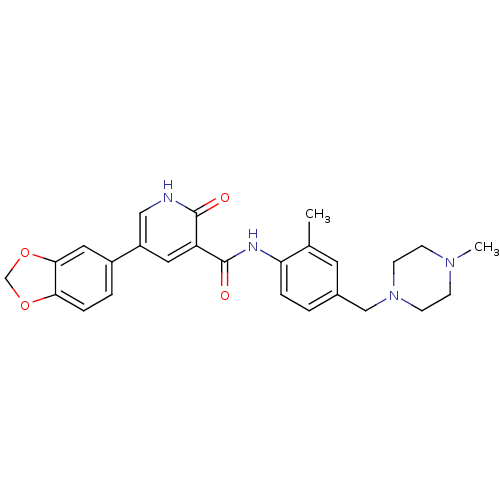 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.45E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Breakpoint cluster region protein/Tyrosine-protein kinase ABL1 (Homo sapiens (Human)) | BDBM50181675 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.46E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Antiproliferative activity against human BCR-ABL positive chronic myeloid leukemia K562 cell line | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin-like growth factor 1 receptor (Homo sapiens (Human)) | BDBM50181670 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IGF1R | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181679 (5-(2-methyl-benzothiazol-5-yl)-2-oxo-1,2-dihydro-p...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor/Nucleophosmin (Homo sapiens (Human)) | BDBM50181672 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.74E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Antiproliferative activity against murine BaF3 cell line expressing NPM-ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181680 (6-oxo-1,6-dihydro-[3,4']bipyridinyl-5-carboxylic a...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.95E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin-like growth factor 1 receptor (Homo sapiens (Human)) | BDBM50181669 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IGF1R | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein kinase ABL1 (Homo sapiens (Human)) | BDBM50181675 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against Abl | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Homo sapiens (Human)) | BDBM50181672 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against Src | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein kinase ABL1 (Homo sapiens (Human)) | BDBM50181670 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against Abl | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor-type tyrosine-protein kinase FLT3 (Homo sapiens (Human)) | BDBM50181675 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against Flt3 | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Homo sapiens (Human)) | BDBM50181669 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against Src | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein kinase ABL1 (Homo sapiens (Human)) | BDBM50181669 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against Abl | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Homo sapiens (Human)) | BDBM50181670 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against Src | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Homo sapiens (Human)) | BDBM50181675 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against Src | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein kinase ABL1 (Homo sapiens (Human)) | BDBM50181672 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against Abl | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin-like growth factor 1 receptor (Homo sapiens (Human)) | BDBM50181675 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IGF1R | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor-type tyrosine-protein kinase FLT3 (Homo sapiens (Human)) | BDBM50181669 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against Flt3 | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181677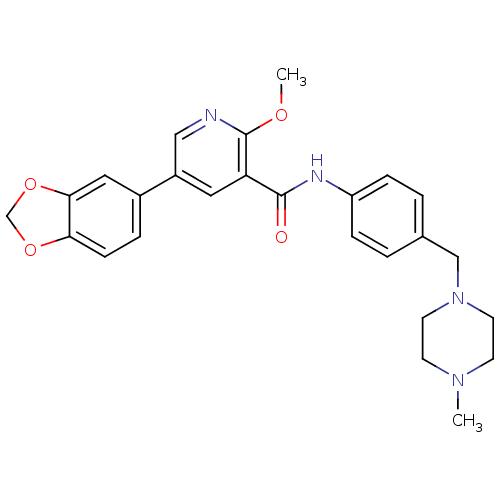 (5-benzo[1,3]dioxol-5-yl-2-methoxy-N-[4-(4-methyl-p...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.03E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor/Nucleophosmin (Homo sapiens (Human)) | BDBM50181669 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Antiproliferative activity against murine BaF3 cell line expressing NPM-ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181678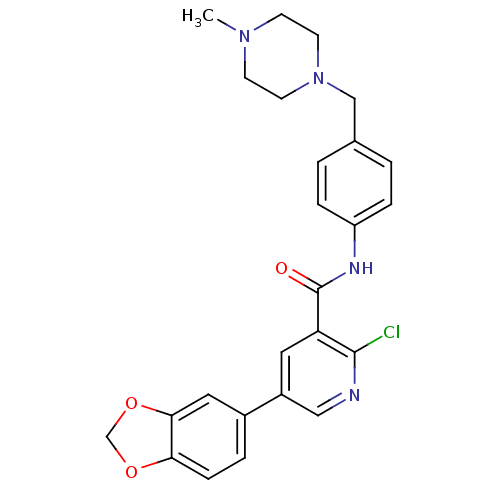 (5-benzo[1,3]dioxol-5-yl-2-chloro-N-[4-(4-methyl-pi...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.68E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181678 (5-benzo[1,3]dioxol-5-yl-2-chloro-N-[4-(4-methyl-pi...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.73E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181671 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.25E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181673 (5-(3,4,5-trimethoxyphenyl)-2-oxo-1,2-dihydro-pyrid...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.35E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181676 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181683 (5-benzo[1,3]dioxol-5-yl-2-oxo-1,2-dihydro-pyridine...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181680 (6-oxo-1,6-dihydro-[3,4']bipyridinyl-5-carboxylic a...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ALK tyrosine kinase receptor (Homo sapiens (Human)) | BDBM50181681 (5-benzo[1,3]dioxol-5-yl-1-methyl-2-oxo-1,2-dihydro...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against ALK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181681 (5-benzo[1,3]dioxol-5-yl-1-methyl-2-oxo-1,2-dihydro...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Insulin receptor (Homo sapiens (Human)) | BDBM50181677 (5-benzo[1,3]dioxol-5-yl-2-methoxy-N-[4-(4-methyl-p...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
ChemBridge Research Laboratories and ChemBridge Corporation Curated by ChEMBL | Assay Description Inhibitory activity against IRK | J Med Chem 49: 1006-15 (2006) Article DOI: 10.1021/jm050824x BindingDB Entry DOI: 10.7270/Q2H994T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||