 Found 37 hits of Enzyme Inhibition Constant Data
Found 37 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM50043407
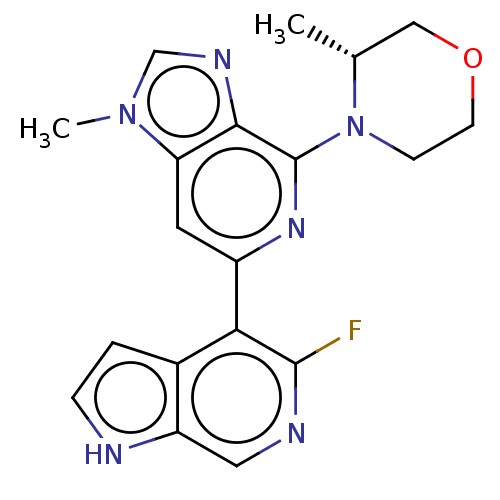
(CHEMBL3355476)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1c(F)ncc2[nH]ccc12 |r| | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ATR using ATP substrate measured after 3 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM50043409
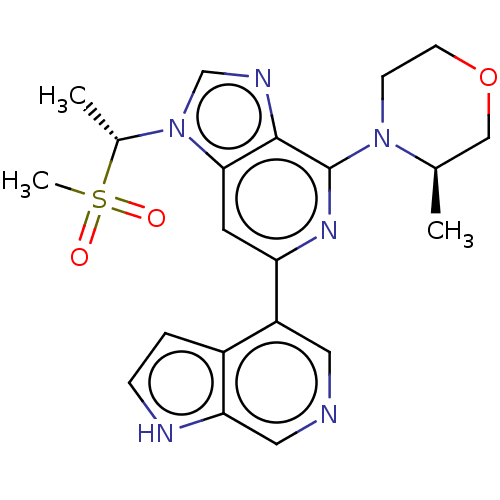
(CHEMBL3355474)Show SMILES C[C@H](n1cnc2c(nc(cc12)-c1cncc2[nH]ccc12)N1CCOC[C@H]1C)S(C)(=O)=O |r| | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ATR using ATP substrate measured after 3 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM50043408
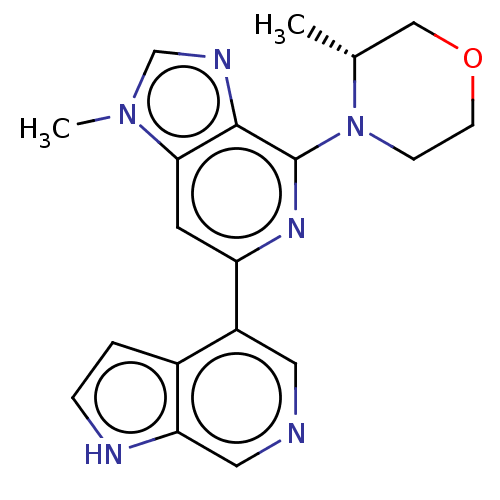
(CHEMBL3355475)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1cncc2[nH]ccc12 |r| | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ATR using ATP substrate measured after 3 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM50043411

(CHEMBL3355480)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1nncc2[nH]ccc12 |r| | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ATR using ATP substrate measured after 3 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM50043410

(CHEMBL3355473)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(CS(C)(=O)=O)cnc12)-c1cccc2[nH]ccc12 |r| | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ATR using ATP substrate measured after 3 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM50043403
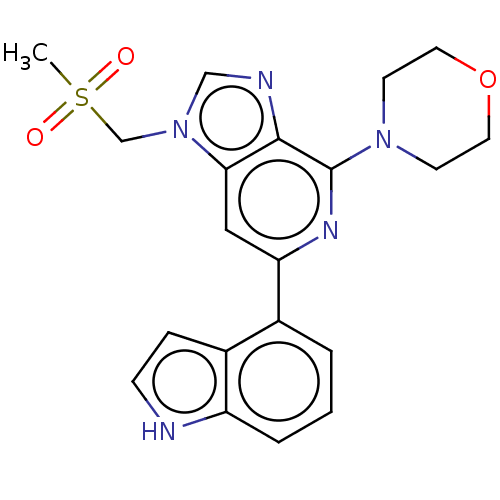
(CHEMBL3355472)Show SMILES CS(=O)(=O)Cn1cnc2c(nc(cc12)-c1cccc2[nH]ccc12)N1CCOCC1 Show InChI InChI=1S/C20H21N5O3S/c1-29(26,27)13-25-12-22-19-18(25)11-17(23-20(19)24-7-9-28-10-8-24)14-3-2-4-16-15(14)5-6-21-16/h2-6,11-12,21H,7-10,13H2,1H3 | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ATR using ATP substrate measured after 3 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM50043413

(CHEMBL3355478)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(CS(C)(=O)=O)cnc12)-c1cnc(N)nc1 |r| Show InChI InChI=1S/C17H21N7O3S/c1-11-8-27-4-3-24(11)16-15-14(23(9-21-15)10-28(2,25)26)5-13(22-16)12-6-19-17(18)20-7-12/h5-7,9,11H,3-4,8,10H2,1-2H3,(H2,18,19,20)/t11-/m1/s1 | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ATR using ATP substrate measured after 3 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM50043404
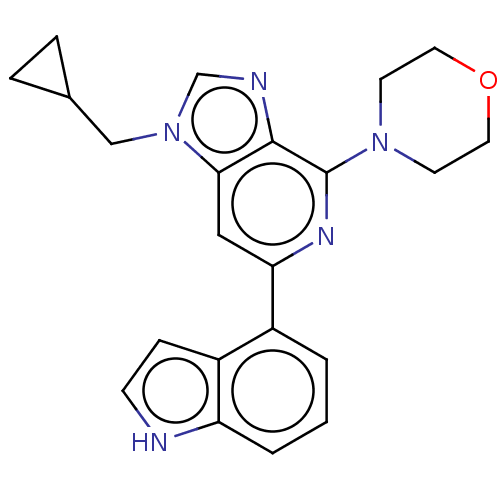
(CHEMBL3352848)Show SMILES C(C1CC1)n1cnc2c(nc(cc12)-c1cccc2[nH]ccc12)N1CCOCC1 Show InChI InChI=1S/C22H23N5O/c1-2-16(17-6-7-23-18(17)3-1)19-12-20-21(24-14-27(20)13-15-4-5-15)22(25-19)26-8-10-28-11-9-26/h1-3,6-7,12,14-15,23H,4-5,8-11,13H2 | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ATR using ATP substrate measured after 3 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM50043414
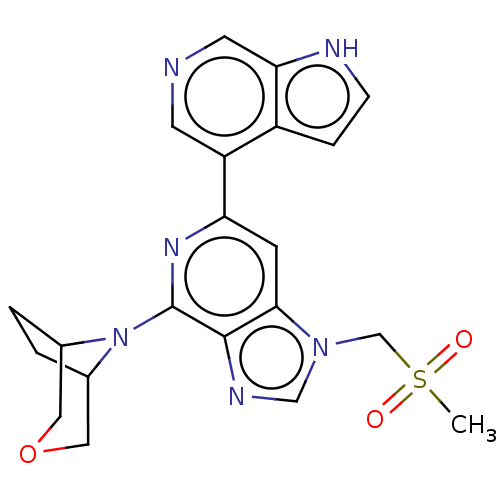
(CHEMBL3355477)Show SMILES CS(=O)(=O)Cn1cnc2c(nc(cc12)-c1cncc2[nH]ccc12)N1C2CCC1COC2 | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ATR using ATP substrate measured after 3 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM50043415

(CHEMBL3355471)Show SMILES O=C(NC1CC1)c1cn2c(nc(cc2n1)-c1cccc2[nH]ccc12)N1CCOCC1 Show InChI InChI=1S/C22H22N6O2/c29-21(24-14-4-5-14)19-13-28-20(25-19)12-18(26-22(28)27-8-10-30-11-9-27)15-2-1-3-17-16(15)6-7-23-17/h1-3,6-7,12-14,23H,4-5,8-11H2,(H,24,29) | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ATR using ATP substrate measured after 3 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase Chk1
(Homo sapiens (Human)) | BDBM50043409

(CHEMBL3355474)Show SMILES C[C@H](n1cnc2c(nc(cc12)-c1cncc2[nH]ccc12)N1CCOC[C@H]1C)S(C)(=O)=O |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of full-length Chk1 in HeLa S3 cells assessed as phosphorylation at Ser345 after 4 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase Chk1
(Homo sapiens (Human)) | BDBM50043408

(CHEMBL3355475)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1cncc2[nH]ccc12 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 84 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of full-length Chk1 in HeLa S3 cells assessed as phosphorylation at Ser345 after 4 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM50043405
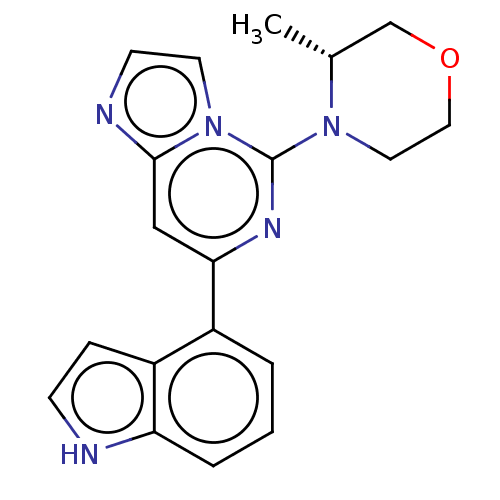
(CHEMBL3355470)Show SMILES C[C@@H]1COCCN1c1nc(cc2nccn12)-c1cccc2[nH]ccc12 |r| | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 95 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ATR using ATP substrate measured after 3 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM50043406

(CHEMBL3355469)Show InChI InChI=1S/C18H17N5O/c1-2-13(14-4-5-19-15(14)3-1)16-12-17-20-6-7-23(17)18(21-16)22-8-10-24-11-9-22/h1-7,12,19H,8-11H2 | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 96 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ATR using ATP substrate measured after 3 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase Chk1
(Homo sapiens (Human)) | BDBM50043407

(CHEMBL3355476)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1c(F)ncc2[nH]ccc12 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 96 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of full-length Chk1 in HeLa S3 cells assessed as phosphorylation at Ser345 after 4 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase ATR
(Homo sapiens (Human)) | BDBM50043412
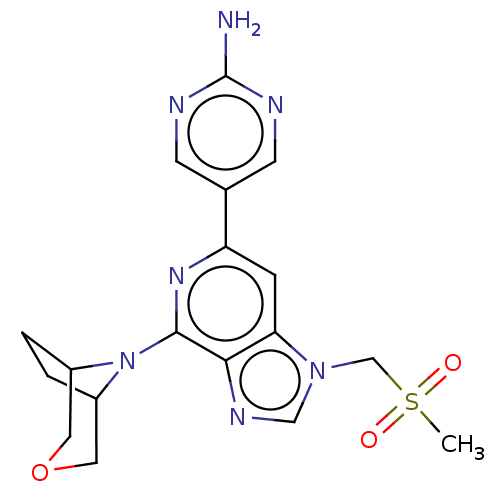
(CHEMBL3355479)Show SMILES CS(=O)(=O)Cn1cnc2c(nc(cc12)-c1cnc(N)nc1)N1C2CCC1COC2 Show InChI InChI=1S/C18H21N7O3S/c1-29(26,27)10-24-9-22-16-15(24)4-14(11-5-20-18(19)21-6-11)23-17(16)25-12-2-3-13(25)8-28-7-12/h4-6,9,12-13H,2-3,7-8,10H2,1H3,(H2,19,20,21) | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ATR using ATP substrate measured after 3 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase mTOR
(Homo sapiens (Human)) | BDBM50043409

(CHEMBL3355474)Show SMILES C[C@H](n1cnc2c(nc(cc12)-c1cncc2[nH]ccc12)N1CCOC[C@H]1C)S(C)(=O)=O |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >4.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of mTOR (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase mTOR
(Homo sapiens (Human)) | BDBM50043408

(CHEMBL3355475)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1cncc2[nH]ccc12 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of mTOR (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50043405

(CHEMBL3355470)Show SMILES C[C@@H]1COCCN1c1nc(cc2nccn12)-c1cccc2[nH]ccc12 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50043406

(CHEMBL3355469)Show InChI InChI=1S/C18H17N5O/c1-2-13(14-4-5-19-15(14)3-1)16-12-17-20-6-7-23(17)18(21-16)22-8-10-24-11-9-22/h1-7,12,19H,8-11H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50043404

(CHEMBL3352848)Show SMILES C(C1CC1)n1cnc2c(nc(cc12)-c1cccc2[nH]ccc12)N1CCOCC1 Show InChI InChI=1S/C22H23N5O/c1-2-16(17-6-7-23-18(17)3-1)19-12-20-21(24-14-27(20)13-15-4-5-15)22(25-19)26-8-10-28-11-9-26/h1-3,6-7,12,14-15,23H,4-5,8-11,13H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50043403

(CHEMBL3355472)Show SMILES CS(=O)(=O)Cn1cnc2c(nc(cc12)-c1cccc2[nH]ccc12)N1CCOCC1 Show InChI InChI=1S/C20H21N5O3S/c1-29(26,27)13-25-12-22-19-18(25)11-17(23-20(19)24-7-9-28-10-8-24)14-3-2-4-16-15(14)5-6-21-16/h2-6,11-12,21H,7-10,13H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase mTOR
(Homo sapiens (Human)) | BDBM50043411

(CHEMBL3355480)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1nncc2[nH]ccc12 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of mTOR (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50043411

(CHEMBL3355480)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1nncc2[nH]ccc12 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of ATM (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50043411

(CHEMBL3355480)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1nncc2[nH]ccc12 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50043411

(CHEMBL3355480)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1nncc2[nH]ccc12 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50043411

(CHEMBL3355480)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1nncc2[nH]ccc12 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50043411

(CHEMBL3355480)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1nncc2[nH]ccc12 |r| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
DNA-dependent protein kinase catalytic subunit
(Homo sapiens (Human)) | BDBM50043411

(CHEMBL3355480)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1nncc2[nH]ccc12 |r| | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DNAPK (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50043408

(CHEMBL3355475)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1cncc2[nH]ccc12 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ERG channel by dofetilide binding assay |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50043411

(CHEMBL3355480)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(C)cnc12)-c1nncc2[nH]ccc12 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human ERG channel by dofetilide binding assay |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50043409

(CHEMBL3355474)Show SMILES C[C@H](n1cnc2c(nc(cc12)-c1cncc2[nH]ccc12)N1CCOC[C@H]1C)S(C)(=O)=O |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50043409

(CHEMBL3355474)Show SMILES C[C@H](n1cnc2c(nc(cc12)-c1cncc2[nH]ccc12)N1CCOC[C@H]1C)S(C)(=O)=O |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50043409

(CHEMBL3355474)Show SMILES C[C@H](n1cnc2c(nc(cc12)-c1cncc2[nH]ccc12)N1CCOC[C@H]1C)S(C)(=O)=O |r| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50043409

(CHEMBL3355474)Show SMILES C[C@H](n1cnc2c(nc(cc12)-c1cncc2[nH]ccc12)N1CCOC[C@H]1C)S(C)(=O)=O |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase Chk1
(Homo sapiens (Human)) | BDBM50043410

(CHEMBL3355473)Show SMILES C[C@@H]1COCCN1c1nc(cc2n(CS(C)(=O)=O)cnc12)-c1cccc2[nH]ccc12 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 570 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of full-length Chk1 in HeLa S3 cells assessed as phosphorylation at Ser345 after 4 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase Chk1
(Homo sapiens (Human)) | BDBM50043403

(CHEMBL3355472)Show SMILES CS(=O)(=O)Cn1cnc2c(nc(cc12)-c1cccc2[nH]ccc12)N1CCOCC1 Show InChI InChI=1S/C20H21N5O3S/c1-29(26,27)13-25-12-22-19-18(25)11-17(23-20(19)24-7-9-28-10-8-24)14-3-2-4-16-15(14)5-6-21-16/h2-6,11-12,21H,7-10,13H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.30E+3 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of full-length Chk1 in HeLa S3 cells assessed as phosphorylation at Ser345 after 4 hrs |
ACS Med Chem Lett 6: 42-6 (2015)
Article DOI: 10.1021/ml500352s
BindingDB Entry DOI: 10.7270/Q2XG9SRM |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data