 Found 10 hits of Enzyme Inhibition Constant Data
Found 10 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50500145

(CHEMBL1235987)Show SMILES NC(=O)[C@@H](CS)NC(=O)CCCCCNC(=O)[C@H](Cc1ccc(cc1)C(F)(F)P(O)(O)=O)NC(=O)[C@@H](CC(O)=O)NC(=O)Cc1ccc(cc1)C(F)(F)P(O)(O)=O |r| Show InChI InChI=1S/C32H41F4N5O13P2S/c33-31(34,55(49,50)51)20-9-5-18(6-10-20)14-22(29(47)38-13-3-1-2-4-25(42)40-24(17-57)28(37)46)41-30(48)23(16-27(44)45)39-26(43)15-19-7-11-21(12-8-19)32(35,36)56(52,53)54/h5-12,22-24,57H,1-4,13-17H2,(H2,37,46)(H,38,47)(H,39,43)(H,40,42)(H,41,48)(H,44,45)(H2,49,50,51)(H2,52,53,54)/t22-,23+,24+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Stony Brook University
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) |
J Med Chem 58: 9063-88 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00586
BindingDB Entry DOI: 10.7270/Q24T6NC6 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219566
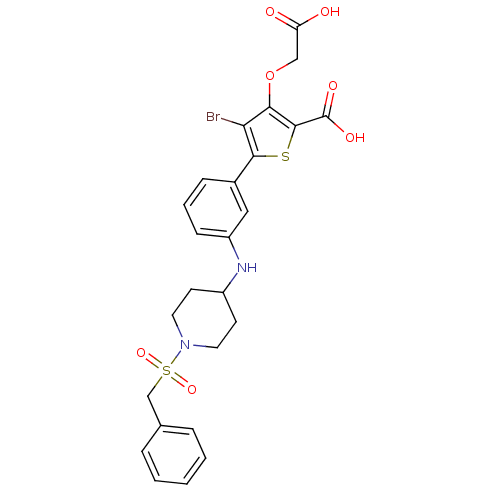
(4-bromo-3-carboxymethoxy-5-[3-(1-phenylmethanesulf...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1cccc(NC2CCN(CC2)S(=O)(=O)Cc2ccccc2)c1 Show InChI InChI=1S/C25H25BrN2O7S2/c26-21-22(35-14-20(29)30)24(25(31)32)36-23(21)17-7-4-8-19(13-17)27-18-9-11-28(12-10-18)37(33,34)15-16-5-2-1-3-6-16/h1-8,13,18,27H,9-12,14-15H2,(H,29,30)(H,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Stony Brook University
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) |
J Med Chem 58: 9063-88 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00586
BindingDB Entry DOI: 10.7270/Q24T6NC6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glucose-1-phosphate cytidylyltransferase
(Salmonella typhi) | BDBM50454133

(CHEBI:17677 | Cystetine Triphosphate)Show SMILES Nc1ccn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)n1 |r| Show InChI InChI=1S/C9H16N3O14P3/c10-5-1-2-12(9(15)11-5)8-7(14)6(13)4(24-8)3-23-28(19,20)26-29(21,22)25-27(16,17)18/h1-2,4,6-8,13-14H,3H2,(H,19,20)(H,21,22)(H2,10,11,15)(H2,16,17,18)/t4-,6-,7-,8-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| 3.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Stony Brook University
Curated by ChEMBL
| Assay Description
Binding affinity to Salmonella typhi glucose-1-phosphate cytidylyl-transferase assessed as dissociation constant by spectrophotometry |
J Med Chem 58: 9063-88 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00586
BindingDB Entry DOI: 10.7270/Q24T6NC6 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50500147
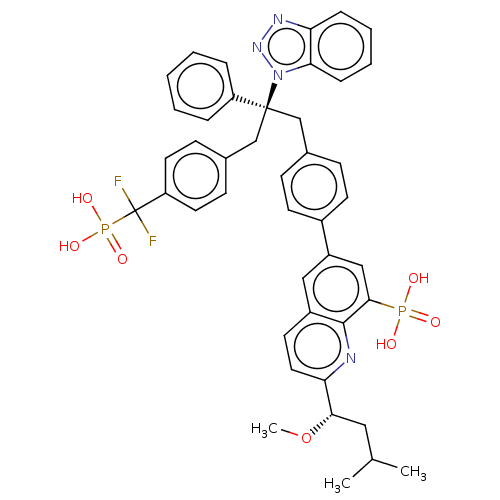
(CHEMBL3746639)Show SMILES CO[C@@H](CC(C)C)c1ccc2cc(cc(c2n1)P(O)(O)=O)-c1ccc(C[C@](Cc2ccc(cc2)C(F)(F)P(O)(O)=O)(c2ccccc2)n2nnc3ccccc23)cc1 |r| Show InChI InChI=1S/C43H42F2N4O7P2/c1-28(2)23-39(56-3)37-22-19-32-24-33(25-40(41(32)46-37)57(50,51)52)31-17-13-29(14-18-31)26-42(34-9-5-4-6-10-34,49-38-12-8-7-11-36(38)47-48-49)27-30-15-20-35(21-16-30)43(44,45)58(53,54)55/h4-22,24-25,28,39H,23,26-27H2,1-3H3,(H2,50,51,52)(H2,53,54,55)/t39-,42-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Stony Brook University
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) |
J Med Chem 58: 9063-88 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00586
BindingDB Entry DOI: 10.7270/Q24T6NC6 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM13604

(6-{4-[2-(1H-1,2,3-benzotriazol-1-yl)-3-{4-[difluor...)Show SMILES COC(CC(C)C)c1ccc2cc(cc(c2n1)P(O)(O)=O)-c1ccc(CC(Cc2ccc(cc2)C(F)(F)P(O)(O)=O)(c2ccccc2)n2nnc3ccccc23)cc1 Show InChI InChI=1S/C43H42F2N4O7P2/c1-28(2)23-39(56-3)37-22-19-32-24-33(25-40(41(32)46-37)57(50,51)52)31-17-13-29(14-18-31)26-42(34-9-5-4-6-10-34,49-38-12-8-7-11-36(38)47-48-49)27-30-15-20-35(21-16-30)43(44,45)58(53,54)55/h4-22,24-25,28,39H,23,26-27H2,1-3H3,(H2,50,51,52)(H2,53,54,55) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
Stony Brook University
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) |
J Med Chem 58: 9063-88 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00586
BindingDB Entry DOI: 10.7270/Q24T6NC6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50500146
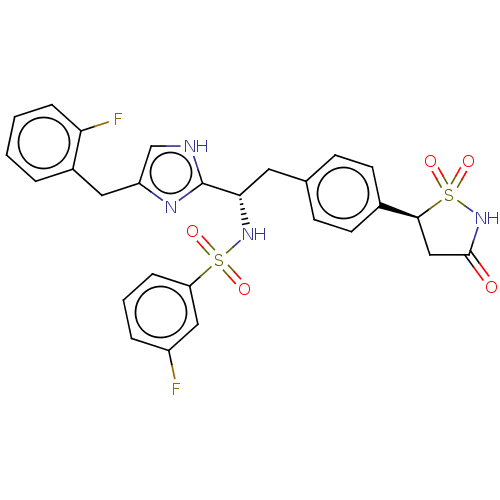
(CHEMBL3747361)Show SMILES Fc1cccc(c1)S(=O)(=O)N[C@@H](Cc1ccc(cc1)[C@@H]1CC(=O)NS1(=O)=O)c1nc(Cc2ccccc2F)c[nH]1 |r| Show InChI InChI=1S/C27H24F2N4O5S2/c28-20-5-3-6-22(14-20)39(35,36)32-24(27-30-16-21(31-27)13-19-4-1-2-7-23(19)29)12-17-8-10-18(11-9-17)25-15-26(34)33-40(25,37)38/h1-11,14,16,24-25,32H,12-13,15H2,(H,30,31)(H,33,34)/t24-,25-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 64 | n/a | n/a | n/a | n/a | n/a | n/a |
Stony Brook University
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) |
J Med Chem 58: 9063-88 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00586
BindingDB Entry DOI: 10.7270/Q24T6NC6 |
More data for this
Ligand-Target Pair | |
Integrase
(Human immunodeficiency virus 1) | BDBM50498473

(CHEMBL1230654)Show SMILES CCC[C@H](C(O)=O)c1c(C)nc2ccc(Cl)cc2c1-c1ccccc1 |r| Show InChI InChI=1S/C21H20ClNO2/c1-3-7-16(21(24)25)19-13(2)23-18-11-10-15(22)12-17(18)20(19)14-8-5-4-6-9-14/h4-6,8-12,16H,3,7H2,1-2H3,(H,24,25)/t16-/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 69 | n/a | n/a | n/a | n/a |
Stony Brook University
Curated by ChEMBL
| Assay Description
Antiviral activity against HIV1 assessed as inhibition of integrase |
J Med Chem 58: 9063-88 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00586
BindingDB Entry DOI: 10.7270/Q24T6NC6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
GTPase KRas
(Homo sapiens (Human)) | BDBM50500144
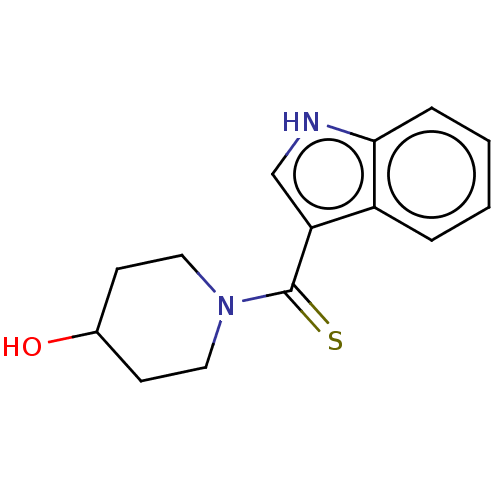
(CHEMBL2086792)Show InChI InChI=1S/C14H16N2OS/c17-10-5-7-16(8-6-10)14(18)12-9-15-13-4-2-1-3-11(12)13/h1-4,9-10,15,17H,5-8H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | n/a | 4.20E+5 | n/a | n/a | n/a | n/a | n/a |
Stony Brook University
Curated by ChEMBL
| Assay Description
Binding affinity to KRAS (unknown origin) |
J Med Chem 58: 9063-88 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00586
BindingDB Entry DOI: 10.7270/Q24T6NC6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Integrase
(Human immunodeficiency virus 1) | BDBM50500143
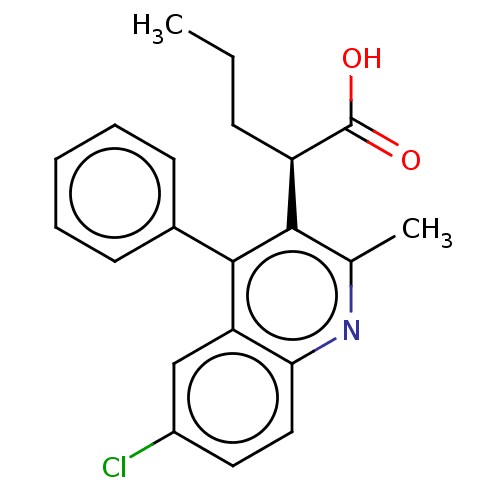
(CHEMBL3747158)Show SMILES CCC[C@@H](C(O)=O)c1c(C)nc2ccc(Cl)cc2c1-c1ccccc1 |r| Show InChI InChI=1S/C21H20ClNO2/c1-3-7-16(21(24)25)19-13(2)23-18-11-10-15(22)12-17(18)20(19)14-8-5-4-6-9-14/h4-6,8-12,16H,3,7H2,1-2H3,(H,24,25)/t16-/m1/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 69 | n/a | n/a | n/a | n/a |
Stony Brook University
Curated by ChEMBL
| Assay Description
Antiviral activity against HIV1 assessed as inhibition of integrase |
J Med Chem 58: 9063-88 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00586
BindingDB Entry DOI: 10.7270/Q24T6NC6 |
More data for this
Ligand-Target Pair | |
GTPase KRas
(Homo sapiens (Human)) | BDBM50088363
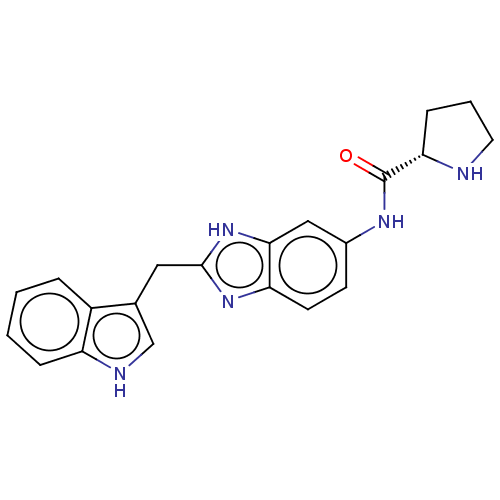
(CHEMBL2086796)Show SMILES O=C(Nc1ccc2nc(Cc3c[nH]c4ccccc34)[nH]c2c1)[C@@H]1CCCN1 |r| Show InChI InChI=1S/C21H21N5O/c27-21(18-6-3-9-22-18)24-14-7-8-17-19(11-14)26-20(25-17)10-13-12-23-16-5-2-1-4-15(13)16/h1-2,4-5,7-8,11-12,18,22-23H,3,6,9-10H2,(H,24,27)(H,25,26)/t18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.90E+5 | n/a | n/a | n/a | n/a | n/a |
Stony Brook University
Curated by ChEMBL
| Assay Description
Binding affinity to KRAS (unknown origin) |
J Med Chem 58: 9063-88 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00586
BindingDB Entry DOI: 10.7270/Q24T6NC6 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data