 Found 142 hits of Enzyme Inhibition Constant Data
Found 142 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50146155
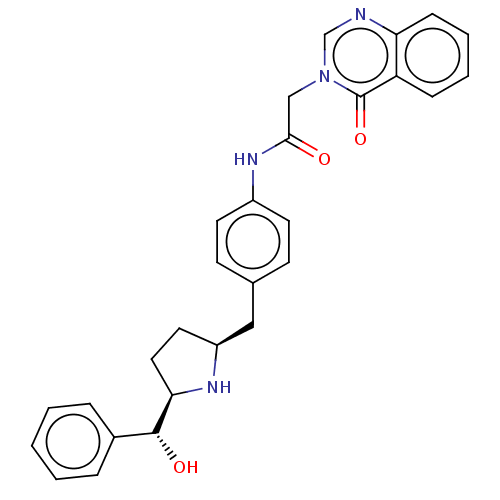
(CHEMBL3764774)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnc4ccccc4c3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C28H28N4O3/c33-26(17-32-18-29-24-9-5-4-8-23(24)28(32)35)31-21-12-10-19(11-13-21)16-22-14-15-25(30-22)27(34)20-6-2-1-3-7-20/h1-13,18,22,25,27,30,34H,14-17H2,(H,31,33)/t22-,25+,27+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human SERT expressed in HEK293 cells preincubated for 30 mins followed by fluorescent substrate addition measured after 30 mins by plat... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50092645
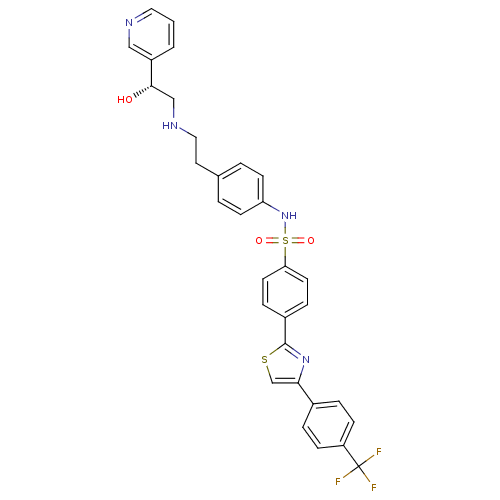
((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...)Show SMILES O[C@@H](CNCCc1ccc(NS(=O)(=O)c2ccc(cc2)-c2nc(cs2)-c2ccc(cc2)C(F)(F)F)cc1)c1cccnc1 Show InChI InChI=1S/C31H27F3N4O3S2/c32-31(33,34)25-9-5-22(6-10-25)28-20-42-30(37-28)23-7-13-27(14-8-23)43(40,41)38-26-11-3-21(4-12-26)15-17-36-19-29(39)24-2-1-16-35-18-24/h1-14,16,18,20,29,36,38-39H,15,17,19H2/t29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 220 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERG channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50448787

(CHEMBL3128178)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4scnc34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H26N4O2S/c29-23(16-2-1-11-25-13-16)20-9-7-18(27-20)12-15-3-5-17(6-4-15)28-24(30)19-8-10-21-22(19)26-14-31-21/h1-6,11,13-14,18-20,23,27,29H,7-10,12H2,(H,28,30)/t18-,19+,20+,23+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 420 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human SERT expressed in HEK293 cells preincubated for 30 mins followed by fluorescent substrate addition measured after 30 mins by plat... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50092645

((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...)Show SMILES O[C@@H](CNCCc1ccc(NS(=O)(=O)c2ccc(cc2)-c2nc(cs2)-c2ccc(cc2)C(F)(F)F)cc1)c1cccnc1 Show InChI InChI=1S/C31H27F3N4O3S2/c32-31(33,34)25-9-5-22(6-10-25)28-20-42-30(37-28)23-7-13-27(14-8-23)43(40,41)38-26-11-3-21(4-12-26)15-17-36-19-29(39)24-2-1-16-35-18-24/h1-14,16,18,20,29,36,38-39H,15,17,19H2/t29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 670 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from human recombinant beta2 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50146157
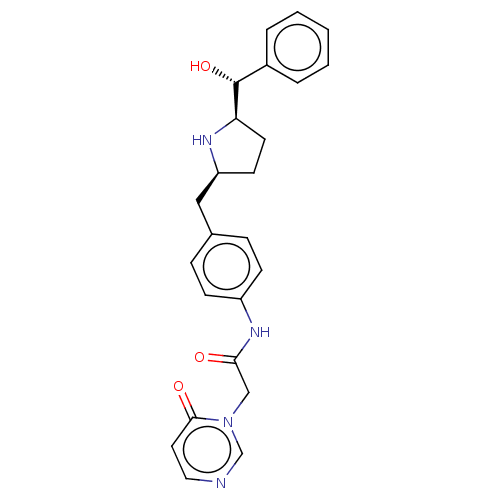
(CHEMBL3764950)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(15-28-16-25-13-12-23(28)30)27-19-8-6-17(7-9-19)14-20-10-11-21(26-20)24(31)18-4-2-1-3-5-18/h1-9,12-13,16,20-21,24,26,31H,10-11,14-15H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human SERT expressed in HEK293 cells preincubated for 30 mins followed by fluorescent substrate addition measured after 30 mins by plat... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50146161
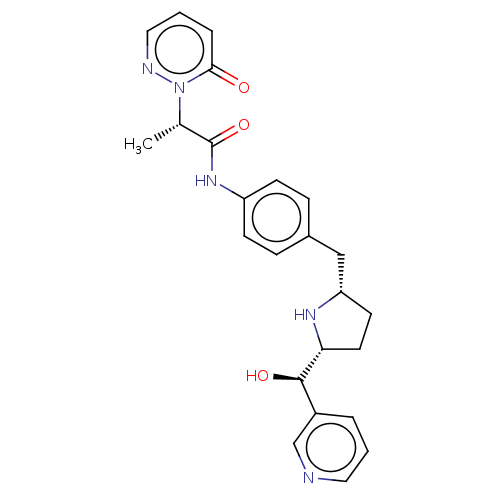
(CHEMBL3763998)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O3/c1-16(29-22(30)5-3-13-26-29)24(32)28-19-8-6-17(7-9-19)14-20-10-11-21(27-20)23(31)18-4-2-12-25-15-18/h2-9,12-13,15-16,20-21,23,27,31H,10-11,14H2,1H3,(H,28,32)/t16-,20-,21+,23+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human SERT expressed in HEK293 cells preincubated for 30 mins followed by fluorescent substrate addition measured after 30 mins by plat... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50448789

(CHEMBL3128188)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4ccnn34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O2/c30-23(17-2-1-12-25-15-17)21-9-7-19(27-21)14-16-3-5-18(6-4-16)28-24(31)22-10-8-20-11-13-26-29(20)22/h1-6,11-13,15,19,21-23,27,30H,7-10,14H2,(H,28,31)/t19-,21+,22-,23+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human SERT expressed in HEK293 cells preincubated for 30 mins followed by fluorescent substrate addition measured after 30 mins by plat... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50092645

((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...)Show SMILES O[C@@H](CNCCc1ccc(NS(=O)(=O)c2ccc(cc2)-c2nc(cs2)-c2ccc(cc2)C(F)(F)F)cc1)c1cccnc1 Show InChI InChI=1S/C31H27F3N4O3S2/c32-31(33,34)25-9-5-22(6-10-25)28-20-42-30(37-28)23-7-13-27(14-8-23)43(40,41)38-26-11-3-21(4-12-26)15-17-36-19-29(39)24-2-1-16-35-18-24/h1-14,16,18,20,29,36,38-39H,15,17,19H2/t29-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from recombinant human beta1 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50146159
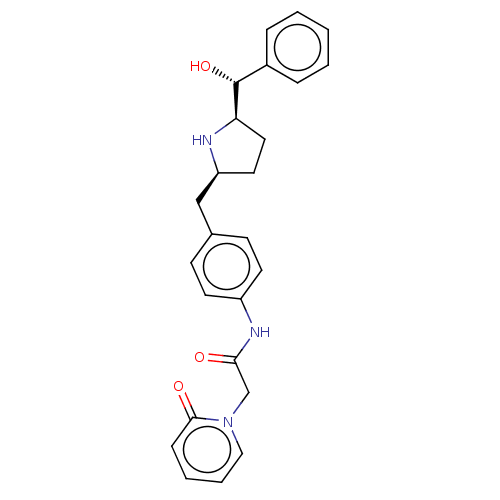
(CHEMBL3764592)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ccccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H27N3O3/c29-23(17-28-15-5-4-8-24(28)30)27-20-11-9-18(10-12-20)16-21-13-14-22(26-21)25(31)19-6-2-1-3-7-19/h1-12,15,21-22,25-26,31H,13-14,16-17H2,(H,27,29)/t21-,22+,25+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human SERT expressed in HEK293 cells preincubated for 30 mins followed by fluorescent substrate addition measured after 30 mins by plat... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50338821
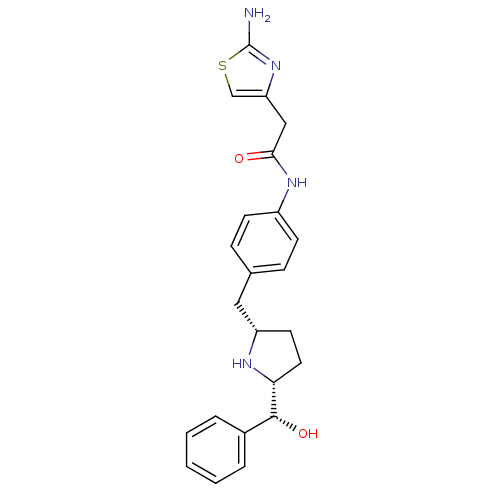
(2-(2-aminothiazol-4-yl)-N-(4-(((2S,5R)-5-((R)-hydr...)Show SMILES Nc1nc(CC(=O)Nc2ccc(C[C@@H]3CC[C@@H](N3)[C@H](O)c3ccccc3)cc2)cs1 |r| Show InChI InChI=1S/C23H26N4O2S/c24-23-27-19(14-30-23)13-21(28)26-17-8-6-15(7-9-17)12-18-10-11-20(25-18)22(29)16-4-2-1-3-5-16/h1-9,14,18,20,22,25,29H,10-13H2,(H2,24,27)(H,26,28)/t18-,20+,22+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human SERT expressed in HEK293 cells preincubated for 30 mins followed by fluorescent substrate addition measured after 30 mins by plat... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM50092645

((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...)Show SMILES O[C@@H](CNCCc1ccc(NS(=O)(=O)c2ccc(cc2)-c2nc(cs2)-c2ccc(cc2)C(F)(F)F)cc1)c1cccnc1 Show InChI InChI=1S/C31H27F3N4O3S2/c32-31(33,34)25-9-5-22(6-10-25)28-20-42-30(37-28)23-7-13-27(14-8-23)43(40,41)38-26-11-3-21(4-12-26)15-17-36-19-29(39)24-2-1-16-35-18-24/h1-14,16,18,20,29,36,38-39H,15,17,19H2/t29-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-diltiazem from human Cav1.2 channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50146155

(CHEMBL3764774)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnc4ccccc4c3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C28H28N4O3/c33-26(17-32-18-29-24-9-5-4-8-23(24)28(32)35)31-21-12-10-19(11-13-21)16-22-14-15-25(30-22)27(34)20-6-2-1-3-7-20/h1-13,18,22,25,27,30,34H,14-17H2,(H,31,33)/t22-,25+,27+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERG channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146155

(CHEMBL3764774)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnc4ccccc4c3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C28H28N4O3/c33-26(17-32-18-29-24-9-5-4-8-23(24)28(32)35)31-21-12-10-19(11-13-21)16-22-14-15-25(30-22)27(34)20-6-2-1-3-7-20/h1-13,18,22,25,27,30,34H,14-17H2,(H,31,33)/t22-,25+,27+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from human recombinant beta2 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50338821

(2-(2-aminothiazol-4-yl)-N-(4-(((2S,5R)-5-((R)-hydr...)Show SMILES Nc1nc(CC(=O)Nc2ccc(C[C@@H]3CC[C@@H](N3)[C@H](O)c3ccccc3)cc2)cs1 |r| Show InChI InChI=1S/C23H26N4O2S/c24-23-27-19(14-30-23)13-21(28)26-17-8-6-15(7-9-17)12-18-10-11-20(25-18)22(29)16-4-2-1-3-5-16/h1-9,14,18,20,22,25,29H,10-13H2,(H2,24,27)(H,26,28)/t18-,20+,22+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes assessed as dextraomethorphan O-demethylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50146158
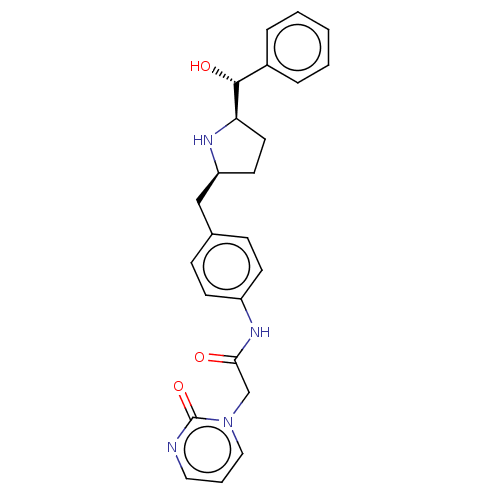
(CHEMBL3763594)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cccnc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-14-4-13-25-24(28)31)27-19-9-7-17(8-10-19)15-20-11-12-21(26-20)23(30)18-5-2-1-3-6-18/h1-10,13-14,20-21,23,26,30H,11-12,15-16H2,(H,27,29)/t20-,21+,23+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human SERT expressed in HEK293 cells preincubated for 30 mins followed by fluorescent substrate addition measured after 30 mins by plat... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50146156
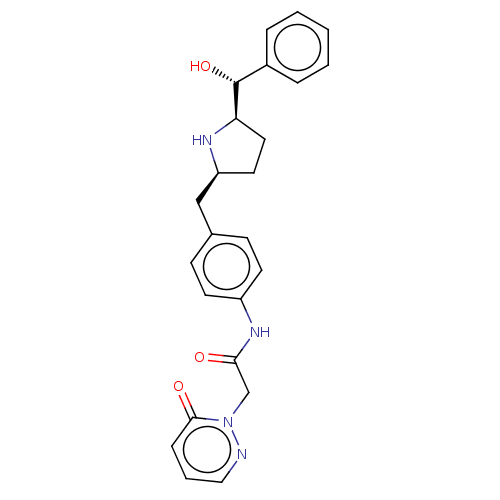
(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human SERT expressed in HEK293 cells preincubated for 30 mins followed by fluorescent substrate addition measured after 30 mins by plat... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium-dependent dopamine transporter
(Homo sapiens (Human)) | BDBM50146162

(CHEMBL3763934)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4cccc(=O)n34)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C27H29N3O3/c31-25-8-4-7-22-14-16-24(30(22)25)27(33)29-20-11-9-18(10-12-20)17-21-13-15-23(28-21)26(32)19-5-2-1-3-6-19/h1-12,21,23-24,26,28,32H,13-17H2,(H,29,33)/t21-,23+,24-,26+/m0/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human dopamine transporter |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50092645

((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...)Show SMILES O[C@@H](CNCCc1ccc(NS(=O)(=O)c2ccc(cc2)-c2nc(cs2)-c2ccc(cc2)C(F)(F)F)cc1)c1cccnc1 Show InChI InChI=1S/C31H27F3N4O3S2/c32-31(33,34)25-9-5-22(6-10-25)28-20-42-30(37-28)23-7-13-27(14-8-23)43(40,41)38-26-11-3-21(4-12-26)15-17-36-19-29(39)24-2-1-16-35-18-24/h1-14,16,18,20,29,36,38-39H,15,17,19H2/t29-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human SERT expressed in HEK293 cells preincubated for 30 mins followed by fluorescent substrate addition measured after 30 mins by plat... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50448787

(CHEMBL3128178)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4scnc34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H26N4O2S/c29-23(16-2-1-11-25-13-16)20-9-7-18(27-20)12-15-3-5-17(6-4-15)28-24(30)19-8-10-21-22(19)26-14-31-21/h1-6,11,13-14,18-20,23,27,29H,7-10,12H2,(H,28,30)/t18-,19+,20+,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERG channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50146162

(CHEMBL3763934)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4cccc(=O)n34)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C27H29N3O3/c31-25-8-4-7-22-14-16-24(30(22)25)27(33)29-20-11-9-18(10-12-20)17-21-13-15-23(28-21)26(32)19-5-2-1-3-6-19/h1-12,21,23-24,26,28,32H,13-17H2,(H,29,33)/t21-,23+,24-,26+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human SERT expressed in HEK293 cells preincubated for 30 mins followed by fluorescent substrate addition measured after 30 mins by plat... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM50338821

(2-(2-aminothiazol-4-yl)-N-(4-(((2S,5R)-5-((R)-hydr...)Show SMILES Nc1nc(CC(=O)Nc2ccc(C[C@@H]3CC[C@@H](N3)[C@H](O)c3ccccc3)cc2)cs1 |r| Show InChI InChI=1S/C23H26N4O2S/c24-23-27-19(14-30-23)13-21(28)26-17-8-6-15(7-9-17)12-18-10-11-20(25-18)22(29)16-4-2-1-3-5-16/h1-9,14,18,20,22,25,29H,10-13H2,(H2,24,27)(H,26,28)/t18-,20+,22+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 8.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-diltiazem from human Cav1.2 channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50092645

((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...)Show SMILES O[C@@H](CNCCc1ccc(NS(=O)(=O)c2ccc(cc2)-c2nc(cs2)-c2ccc(cc2)C(F)(F)F)cc1)c1cccnc1 Show InChI InChI=1S/C31H27F3N4O3S2/c32-31(33,34)25-9-5-22(6-10-25)28-20-42-30(37-28)23-7-13-27(14-8-23)43(40,41)38-26-11-3-21(4-12-26)15-17-36-19-29(39)24-2-1-16-35-18-24/h1-14,16,18,20,29,36,38-39H,15,17,19H2/t29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as testosterone 6beta-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human SERT expressed in HEK293 cells preincubated for 30 mins followed by fluorescent substrate addition measured after 30 mins by plat... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50146160
(CHEMBL3765335)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H28N4O3/c1-17(29-23(30)8-5-15-26-29)25(32)28-20-11-9-18(10-12-20)16-21-13-14-22(27-21)24(31)19-6-3-2-4-7-19/h2-12,15,17,21-22,24,27,31H,13-14,16H2,1H3,(H,28,32)/t17-,21-,22+,24+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human SERT expressed in HEK293 cells preincubated for 30 mins followed by fluorescent substrate addition measured after 30 mins by plat... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50092645

((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...)Show SMILES O[C@@H](CNCCc1ccc(NS(=O)(=O)c2ccc(cc2)-c2nc(cs2)-c2ccc(cc2)C(F)(F)F)cc1)c1cccnc1 Show InChI InChI=1S/C31H27F3N4O3S2/c32-31(33,34)25-9-5-22(6-10-25)28-20-42-30(37-28)23-7-13-27(14-8-23)43(40,41)38-26-11-3-21(4-12-26)15-17-36-19-29(39)24-2-1-16-35-18-24/h1-14,16,18,20,29,36,38-39H,15,17,19H2/t29-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes assessed as dextraomethorphan O-demethylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM50146155

(CHEMBL3764774)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnc4ccccc4c3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C28H28N4O3/c33-26(17-32-18-29-24-9-5-4-8-23(24)28(32)35)31-21-12-10-19(11-13-21)16-22-14-15-25(30-22)27(34)20-6-2-1-3-7-20/h1-13,18,22,25,27,30,34H,14-17H2,(H,31,33)/t22-,25+,27+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-diltiazem from human Cav1.2 channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50338821

(2-(2-aminothiazol-4-yl)-N-(4-(((2S,5R)-5-((R)-hydr...)Show SMILES Nc1nc(CC(=O)Nc2ccc(C[C@@H]3CC[C@@H](N3)[C@H](O)c3ccccc3)cc2)cs1 |r| Show InChI InChI=1S/C23H26N4O2S/c24-23-27-19(14-30-23)13-21(28)26-17-8-6-15(7-9-17)12-18-10-11-20(25-18)22(29)16-4-2-1-3-5-16/h1-9,14,18,20,22,25,29H,10-13H2,(H2,24,27)(H,26,28)/t18-,20+,22+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 1.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERG channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146161

(CHEMBL3763998)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O3/c1-16(29-22(30)5-3-13-26-29)24(32)28-19-8-6-17(7-9-19)14-20-10-11-21(27-20)23(31)18-4-2-12-25-15-18/h2-9,12-13,15-16,20-21,23,27,31H,10-11,14H2,1H3,(H,28,32)/t16-,20-,21+,23+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from recombinant human beta1 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50448787

(CHEMBL3128178)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4scnc34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H26N4O2S/c29-23(16-2-1-11-25-13-16)20-9-7-18(27-20)12-15-3-5-17(6-4-15)28-24(30)19-8-10-21-22(19)26-14-31-21/h1-6,11,13-14,18-20,23,27,29H,7-10,12H2,(H,28,30)/t18-,19+,20+,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from human recombinant beta2 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146157

(CHEMBL3764950)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(15-28-16-25-13-12-23(28)30)27-19-8-6-17(7-9-19)14-20-10-11-21(26-20)24(31)18-4-2-1-3-5-18/h1-9,12-13,16,20-21,24,26,31H,10-11,14-15H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from recombinant human beta1 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from human recombinant beta2 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50448789

(CHEMBL3128188)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4ccnn34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O2/c30-23(17-2-1-12-25-15-17)21-9-7-19(27-21)14-16-3-5-18(6-4-16)28-24(31)22-10-8-20-11-13-26-29(20)22/h1-6,11-13,15,19,21-23,27,30H,7-10,14H2,(H,28,31)/t19-,21+,22-,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from human recombinant beta2 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from recombinant human beta1 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146155

(CHEMBL3764774)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnc4ccccc4c3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C28H28N4O3/c33-26(17-32-18-29-24-9-5-4-8-23(24)28(32)35)31-21-12-10-19(11-13-21)16-22-14-15-25(30-22)27(34)20-6-2-1-3-7-20/h1-13,18,22,25,27,30,34H,14-17H2,(H,31,33)/t22-,25+,27+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from recombinant human beta1 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146156

(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from recombinant human beta1 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146161

(CHEMBL3763998)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O3/c1-16(29-22(30)5-3-13-26-29)24(32)28-19-8-6-17(7-9-19)14-20-10-11-21(27-20)23(31)18-4-2-12-25-15-18/h2-9,12-13,15-16,20-21,23,27,31H,10-11,14H2,1H3,(H,28,32)/t16-,20-,21+,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from human recombinant beta2 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146162

(CHEMBL3763934)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4cccc(=O)n34)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C27H29N3O3/c31-25-8-4-7-22-14-16-24(30(22)25)27(33)29-20-11-9-18(10-12-20)17-21-13-15-23(28-21)26(32)19-5-2-1-3-6-19/h1-12,21,23-24,26,28,32H,13-17H2,(H,29,33)/t21-,23+,24-,26+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from recombinant human beta1 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146158

(CHEMBL3763594)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cccnc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-14-4-13-25-24(28)31)27-19-9-7-17(8-10-19)15-20-11-12-21(26-20)23(30)18-5-2-1-3-6-18/h1-10,13-14,20-21,23,26,30H,11-12,15-16H2,(H,27,29)/t20-,21+,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from human recombinant beta2 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146157

(CHEMBL3764950)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(15-28-16-25-13-12-23(28)30)27-19-8-6-17(7-9-19)14-20-10-11-21(26-20)24(31)18-4-2-1-3-5-18/h1-9,12-13,16,20-21,24,26,31H,10-11,14-15H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from human recombinant beta2 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50338821

(2-(2-aminothiazol-4-yl)-N-(4-(((2S,5R)-5-((R)-hydr...)Show SMILES Nc1nc(CC(=O)Nc2ccc(C[C@@H]3CC[C@@H](N3)[C@H](O)c3ccccc3)cc2)cs1 |r| Show InChI InChI=1S/C23H26N4O2S/c24-23-27-19(14-30-23)13-21(28)26-17-8-6-15(7-9-17)12-18-10-11-20(25-18)22(29)16-4-2-1-3-5-16/h1-9,14,18,20,22,25,29H,10-13H2,(H2,24,27)(H,26,28)/t18-,20+,22+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from human recombinant beta2 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50448789

(CHEMBL3128188)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4ccnn34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O2/c30-23(17-2-1-12-25-15-17)21-9-7-19(27-21)14-16-3-5-18(6-4-16)28-24(31)22-10-8-20-11-13-26-29(20)22/h1-6,11-13,15,19,21-23,27,30H,7-10,14H2,(H,28,31)/t19-,21+,22-,23+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from recombinant human beta1 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50448787

(CHEMBL3128178)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4scnc34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H26N4O2S/c29-23(16-2-1-11-25-13-16)20-9-7-18(27-20)12-15-3-5-17(6-4-15)28-24(30)19-8-10-21-22(19)26-14-31-21/h1-6,11,13-14,18-20,23,27,29H,7-10,12H2,(H,28,30)/t18-,19+,20+,23+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from recombinant human beta1 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50338821

(2-(2-aminothiazol-4-yl)-N-(4-(((2S,5R)-5-((R)-hydr...)Show SMILES Nc1nc(CC(=O)Nc2ccc(C[C@@H]3CC[C@@H](N3)[C@H](O)c3ccccc3)cc2)cs1 |r| Show InChI InChI=1S/C23H26N4O2S/c24-23-27-19(14-30-23)13-21(28)26-17-8-6-15(7-9-17)12-18-10-11-20(25-18)22(29)16-4-2-1-3-5-16/h1-9,14,18,20,22,25,29H,10-13H2,(H2,24,27)(H,26,28)/t18-,20+,22+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from recombinant human beta1 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146156

(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from human recombinant beta2 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146158

(CHEMBL3763594)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cccnc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-14-4-13-25-24(28)31)27-19-9-7-17(8-10-19)15-20-11-12-21(26-20)23(30)18-5-2-1-3-6-18/h1-10,13-14,20-21,23,26,30H,11-12,15-16H2,(H,27,29)/t20-,21+,23+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from recombinant human beta1 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146162

(CHEMBL3763934)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4cccc(=O)n34)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C27H29N3O3/c31-25-8-4-7-22-14-16-24(30(22)25)27(33)29-20-11-9-18(10-12-20)17-21-13-15-23(28-21)26(32)19-5-2-1-3-6-19/h1-12,21,23-24,26,28,32H,13-17H2,(H,29,33)/t21-,23+,24-,26+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from human recombinant beta2 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146159

(CHEMBL3764592)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ccccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H27N3O3/c29-23(17-28-15-5-4-8-24(28)30)27-20-11-9-18(10-12-20)16-21-13-14-22(26-21)25(31)19-6-2-1-3-7-19/h1-12,15,21-22,25-26,31H,13-14,16-17H2,(H,27,29)/t21-,22+,25+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from recombinant human beta1 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146160

(CHEMBL3765335)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H28N4O3/c1-17(29-23(30)8-5-15-26-29)25(32)28-20-11-9-18(10-12-20)16-21-13-14-22(27-21)24(31)19-6-3-2-4-7-19/h2-12,15,17,21-22,24,27,31H,13-14,16H2,1H3,(H,28,32)/t17-,21-,22+,24+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from human recombinant beta2 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146159

(CHEMBL3764592)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ccccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H27N3O3/c29-23(17-28-15-5-4-8-24(28)30)27-20-11-9-18(10-12-20)16-21-13-14-22(26-21)25(31)19-6-2-1-3-7-19/h1-12,15,21-22,25-26,31H,13-14,16-17H2,(H,27,29)/t21-,22+,25+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from human recombinant beta2 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146160

(CHEMBL3765335)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H28N4O3/c1-17(29-23(30)8-5-15-26-29)25(32)28-20-11-9-18(10-12-20)16-21-13-14-22(27-21)24(31)19-6-3-2-4-7-19/h2-12,15,17,21-22,24,27,31H,13-14,16H2,1H3,(H,28,32)/t17-,21-,22+,24+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125]I-cyanopindolol from recombinant human beta1 adrenergic receptor after 1 hr by scintillation counting method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50092645

((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...)Show SMILES O[C@@H](CNCCc1ccc(NS(=O)(=O)c2ccc(cc2)-c2nc(cs2)-c2ccc(cc2)C(F)(F)F)cc1)c1cccnc1 Show InChI InChI=1S/C31H27F3N4O3S2/c32-31(33,34)25-9-5-22(6-10-25)28-20-42-30(37-28)23-7-13-27(14-8-23)43(40,41)38-26-11-3-21(4-12-26)15-17-36-19-29(39)24-2-1-16-35-18-24/h1-14,16,18,20,29,36,38-39H,15,17,19H2/t29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes assessed as diclofenac alpha'-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50146157

(CHEMBL3764950)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(15-28-16-25-13-12-23(28)30)27-19-8-6-17(7-9-19)14-20-10-11-21(26-20)24(31)18-4-2-1-3-5-18/h1-9,12-13,16,20-21,24,26,31H,10-11,14-15H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERG channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 5 subunit alpha
(Homo sapiens (Human)) | BDBM50146159

(CHEMBL3764592)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ccccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H27N3O3/c29-23(17-28-15-5-4-8-24(28)30)27-20-11-9-18(10-12-20)16-21-13-14-22(26-21)25(31)19-6-2-1-3-7-19/h1-12,15,21-22,25-26,31H,13-14,16-17H2,(H,27,29)/t21-,22+,25+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.2 channel expressed in Xenopus oocytes by two-intracellular microelectrode voltage clamp method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 5 subunit alpha
(Homo sapiens (Human)) | BDBM50146160

(CHEMBL3765335)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H28N4O3/c1-17(29-23(30)8-5-15-26-29)25(32)28-20-11-9-18(10-12-20)16-21-13-14-22(27-21)24(31)19-6-3-2-4-7-19/h2-12,15,17,21-22,24,27,31H,13-14,16H2,1H3,(H,28,32)/t17-,21-,22+,24+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.2 channel expressed in Xenopus oocytes by two-intracellular microelectrode voltage clamp method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERG channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM50448787

(CHEMBL3128178)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4scnc34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H26N4O2S/c29-23(16-2-1-11-25-13-16)20-9-7-18(27-20)12-15-3-5-17(6-4-15)28-24(30)19-8-10-21-22(19)26-14-31-21/h1-6,11,13-14,18-20,23,27,29H,7-10,12H2,(H,28,30)/t18-,19+,20+,23+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-diltiazem from human Cav1.2 channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM50146162

(CHEMBL3763934)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4cccc(=O)n34)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C27H29N3O3/c31-25-8-4-7-22-14-16-24(30(22)25)27(33)29-20-11-9-18(10-12-20)17-21-13-15-23(28-21)26(32)19-5-2-1-3-6-19/h1-12,21,23-24,26,28,32H,13-17H2,(H,29,33)/t21-,23+,24-,26+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-diltiazem from human Cav1.2 channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-diltiazem from human Cav1.2 channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 5 subunit alpha
(Homo sapiens (Human)) | BDBM50146155

(CHEMBL3764774)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnc4ccccc4c3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C28H28N4O3/c33-26(17-32-18-29-24-9-5-4-8-23(24)28(32)35)31-21-12-10-19(11-13-21)16-22-14-15-25(30-22)27(34)20-6-2-1-3-7-20/h1-13,18,22,25,27,30,34H,14-17H2,(H,31,33)/t22-,25+,27+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.2 channel expressed in Xenopus oocytes by two-intracellular microelectrode voltage clamp method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50448789

(CHEMBL3128188)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4ccnn34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O2/c30-23(17-2-1-12-25-15-17)21-9-7-19(27-21)14-16-3-5-18(6-4-16)28-24(31)22-10-8-20-11-13-26-29(20)22/h1-6,11-13,15,19,21-23,27,30H,7-10,14H2,(H,28,31)/t19-,21+,22-,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERG channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM50146157

(CHEMBL3764950)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(15-28-16-25-13-12-23(28)30)27-19-8-6-17(7-9-19)14-20-10-11-21(26-20)24(31)18-4-2-1-3-5-18/h1-9,12-13,16,20-21,24,26,31H,10-11,14-15H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-diltiazem from human Cav1.2 channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM50146159

(CHEMBL3764592)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ccccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H27N3O3/c29-23(17-28-15-5-4-8-24(28)30)27-20-11-9-18(10-12-20)16-21-13-14-22(26-21)25(31)19-6-2-1-3-7-19/h1-12,15,21-22,25-26,31H,13-14,16-17H2,(H,27,29)/t21-,22+,25+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-diltiazem from human Cav1.2 channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM50146161

(CHEMBL3763998)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O3/c1-16(29-22(30)5-3-13-26-29)24(32)28-19-8-6-17(7-9-19)14-20-10-11-21(27-20)23(31)18-4-2-12-25-15-18/h2-9,12-13,15-16,20-21,23,27,31H,10-11,14H2,1H3,(H,28,32)/t16-,20-,21+,23+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-diltiazem from human Cav1.2 channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 5 subunit alpha
(Homo sapiens (Human)) | BDBM50146161

(CHEMBL3763998)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O3/c1-16(29-22(30)5-3-13-26-29)24(32)28-19-8-6-17(7-9-19)14-20-10-11-21(27-20)23(31)18-4-2-12-25-15-18/h2-9,12-13,15-16,20-21,23,27,31H,10-11,14H2,1H3,(H,28,32)/t16-,20-,21+,23+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.2 channel expressed in Xenopus oocytes by two-intracellular microelectrode voltage clamp method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 5 subunit alpha
(Homo sapiens (Human)) | BDBM50146158

(CHEMBL3763594)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cccnc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-14-4-13-25-24(28)31)27-19-9-7-17(8-10-19)15-20-11-12-21(26-20)23(30)18-5-2-1-3-6-18/h1-10,13-14,20-21,23,26,30H,11-12,15-16H2,(H,27,29)/t20-,21+,23+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.2 channel expressed in Xenopus oocytes by two-intracellular microelectrode voltage clamp method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50146161

(CHEMBL3763998)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O3/c1-16(29-22(30)5-3-13-26-29)24(32)28-19-8-6-17(7-9-19)14-20-10-11-21(27-20)23(31)18-4-2-12-25-15-18/h2-9,12-13,15-16,20-21,23,27,31H,10-11,14H2,1H3,(H,28,32)/t16-,20-,21+,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERG channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50146162

(CHEMBL3763934)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4cccc(=O)n34)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C27H29N3O3/c31-25-8-4-7-22-14-16-24(30(22)25)27(33)29-20-11-9-18(10-12-20)17-21-13-15-23(28-21)26(32)19-5-2-1-3-6-19/h1-12,21,23-24,26,28,32H,13-17H2,(H,29,33)/t21-,23+,24-,26+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERG channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 5 subunit alpha
(Homo sapiens (Human)) | BDBM50146156

(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.2 channel expressed in Xenopus oocytes by two-intracellular microelectrode voltage clamp method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM50146156

(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-diltiazem from human Cav1.2 channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM50448789

(CHEMBL3128188)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4ccnn34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O2/c30-23(17-2-1-12-25-15-17)21-9-7-19(27-21)14-16-3-5-18(6-4-16)28-24(31)22-10-8-20-11-13-26-29(20)22/h1-6,11-13,15,19,21-23,27,30H,7-10,14H2,(H,28,31)/t19-,21+,22-,23+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-diltiazem from human Cav1.2 channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50146156

(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERG channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 5 subunit alpha
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.2 channel expressed in Xenopus oocytes by two-intracellular microelectrode voltage clamp method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 5 subunit alpha
(Homo sapiens (Human)) | BDBM50146162

(CHEMBL3763934)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4cccc(=O)n34)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C27H29N3O3/c31-25-8-4-7-22-14-16-24(30(22)25)27(33)29-20-11-9-18(10-12-20)17-21-13-15-23(28-21)26(32)19-5-2-1-3-6-19/h1-12,21,23-24,26,28,32H,13-17H2,(H,29,33)/t21-,23+,24-,26+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.2 channel expressed in Xenopus oocytes by two-intracellular microelectrode voltage clamp method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 5 subunit alpha
(Homo sapiens (Human)) | BDBM50146157

(CHEMBL3764950)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(15-28-16-25-13-12-23(28)30)27-19-8-6-17(7-9-19)14-20-10-11-21(26-20)24(31)18-4-2-1-3-5-18/h1-9,12-13,16,20-21,24,26,31H,10-11,14-15H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.2 channel expressed in Xenopus oocytes by two-intracellular microelectrode voltage clamp method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 5 subunit alpha
(Homo sapiens (Human)) | BDBM50448789

(CHEMBL3128188)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4ccnn34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O2/c30-23(17-2-1-12-25-15-17)21-9-7-19(27-21)14-16-3-5-18(6-4-16)28-24(31)22-10-8-20-11-13-26-29(20)22/h1-6,11-13,15,19,21-23,27,30H,7-10,14H2,(H,28,31)/t19-,21+,22-,23+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.2 channel expressed in Xenopus oocytes by two-intracellular microelectrode voltage clamp method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 5 subunit alpha
(Homo sapiens (Human)) | BDBM50338821

(2-(2-aminothiazol-4-yl)-N-(4-(((2S,5R)-5-((R)-hydr...)Show SMILES Nc1nc(CC(=O)Nc2ccc(C[C@@H]3CC[C@@H](N3)[C@H](O)c3ccccc3)cc2)cs1 |r| Show InChI InChI=1S/C23H26N4O2S/c24-23-27-19(14-30-23)13-21(28)26-17-8-6-15(7-9-17)12-18-10-11-20(25-18)22(29)16-4-2-1-3-5-16/h1-9,14,18,20,22,25,29H,10-13H2,(H2,24,27)(H,26,28)/t18-,20+,22+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.2 channel expressed in Xenopus oocytes by two-intracellular microelectrode voltage clamp method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50146160

(CHEMBL3765335)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H28N4O3/c1-17(29-23(30)8-5-15-26-29)25(32)28-20-11-9-18(10-12-20)16-21-13-14-22(27-21)24(31)19-6-3-2-4-7-19/h2-12,15,17,21-22,24,27,31H,13-14,16H2,1H3,(H,28,32)/t17-,21-,22+,24+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERG channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50146158

(CHEMBL3763594)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cccnc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-14-4-13-25-24(28)31)27-19-9-7-17(8-10-19)15-20-11-12-21(26-20)23(30)18-5-2-1-3-6-18/h1-10,13-14,20-21,23,26,30H,11-12,15-16H2,(H,27,29)/t20-,21+,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERG channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM50146160

(CHEMBL3765335)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H28N4O3/c1-17(29-23(30)8-5-15-26-29)25(32)28-20-11-9-18(10-12-20)16-21-13-14-22(27-21)24(31)19-6-3-2-4-7-19/h2-12,15,17,21-22,24,27,31H,13-14,16H2,1H3,(H,28,32)/t17-,21-,22+,24+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-diltiazem from human Cav1.2 channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM50146158

(CHEMBL3763594)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cccnc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-14-4-13-25-24(28)31)27-19-9-7-17(8-10-19)15-20-11-12-21(26-20)23(30)18-5-2-1-3-6-18/h1-10,13-14,20-21,23,26,30H,11-12,15-16H2,(H,27,29)/t20-,21+,23+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-diltiazem from human Cav1.2 channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50146159

(CHEMBL3764592)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ccccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H27N3O3/c29-23(17-28-15-5-4-8-24(28)30)27-20-11-9-18(10-12-20)16-21-13-14-22(26-21)25(31)19-6-2-1-3-7-19/h1-12,15,21-22,25-26,31H,13-14,16-17H2,(H,27,29)/t21-,22+,25+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERG channel |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 5 subunit alpha
(Homo sapiens (Human)) | BDBM50448787

(CHEMBL3128178)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4scnc34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H26N4O2S/c29-23(16-2-1-11-25-13-16)20-9-7-18(27-20)12-15-3-5-17(6-4-15)28-24(30)19-8-10-21-22(19)26-14-31-21/h1-6,11,13-14,18-20,23,27,29H,7-10,12H2,(H,28,30)/t18-,19+,20+,23+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.2 channel expressed in Xenopus oocytes by two-intracellular microelectrode voltage clamp method |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50146155

(CHEMBL3764774)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnc4ccccc4c3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C28H28N4O3/c33-26(17-32-18-29-24-9-5-4-8-23(24)28(32)35)31-21-12-10-19(11-13-21)16-22-14-15-25(30-22)27(34)20-6-2-1-3-7-20/h1-13,18,22,25,27,30,34H,14-17H2,(H,31,33)/t22-,25+,27+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes assessed as dextraomethorphan O-demethylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50146158

(CHEMBL3763594)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cccnc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-14-4-13-25-24(28)31)27-19-9-7-17(8-10-19)15-20-11-12-21(26-20)23(30)18-5-2-1-3-6-18/h1-10,13-14,20-21,23,26,30H,11-12,15-16H2,(H,27,29)/t20-,21+,23+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes assessed as dextraomethorphan O-demethylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50448787

(CHEMBL3128178)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4scnc34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H26N4O2S/c29-23(16-2-1-11-25-13-16)20-9-7-18(27-20)12-15-3-5-17(6-4-15)28-24(30)19-8-10-21-22(19)26-14-31-21/h1-6,11,13-14,18-20,23,27,29H,7-10,12H2,(H,28,30)/t18-,19+,20+,23+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes assessed as dextraomethorphan O-demethylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50146155

(CHEMBL3764774)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnc4ccccc4c3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C28H28N4O3/c33-26(17-32-18-29-24-9-5-4-8-23(24)28(32)35)31-21-12-10-19(11-13-21)16-22-14-15-25(30-22)27(34)20-6-2-1-3-7-20/h1-13,18,22,25,27,30,34H,14-17H2,(H,31,33)/t22-,25+,27+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as testosterone 6beta-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50448789

(CHEMBL3128188)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4ccnn34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O2/c30-23(17-2-1-12-25-15-17)21-9-7-19(27-21)14-16-3-5-18(6-4-16)28-24(31)22-10-8-20-11-13-26-29(20)22/h1-6,11-13,15,19,21-23,27,30H,7-10,14H2,(H,28,31)/t19-,21+,22-,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes assessed as diclofenac alpha'-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50448787

(CHEMBL3128178)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4scnc34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H26N4O2S/c29-23(16-2-1-11-25-13-16)20-9-7-18(27-20)12-15-3-5-17(6-4-15)28-24(30)19-8-10-21-22(19)26-14-31-21/h1-6,11,13-14,18-20,23,27,29H,7-10,12H2,(H,28,30)/t18-,19+,20+,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as testosterone 6beta-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50448787

(CHEMBL3128178)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4scnc34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H26N4O2S/c29-23(16-2-1-11-25-13-16)20-9-7-18(27-20)12-15-3-5-17(6-4-15)28-24(30)19-8-10-21-22(19)26-14-31-21/h1-6,11,13-14,18-20,23,27,29H,7-10,12H2,(H,28,30)/t18-,19+,20+,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes assessed as diclofenac alpha'-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes assessed as dextraomethorphan O-demethylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as testosterone 6beta-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50146156

(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes assessed as diclofenac alpha'-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50448789

(CHEMBL3128188)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4ccnn34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O2/c30-23(17-2-1-12-25-15-17)21-9-7-19(27-21)14-16-3-5-18(6-4-16)28-24(31)22-10-8-20-11-13-26-29(20)22/h1-6,11-13,15,19,21-23,27,30H,7-10,14H2,(H,28,31)/t19-,21+,22-,23+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes assessed as dextraomethorphan O-demethylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C8 in human liver microsomes |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50448789

(CHEMBL3128188)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4ccnn34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O2/c30-23(17-2-1-12-25-15-17)21-9-7-19(27-21)14-16-3-5-18(6-4-16)28-24(31)22-10-8-20-11-13-26-29(20)22/h1-6,11-13,15,19,21-23,27,30H,7-10,14H2,(H,28,31)/t19-,21+,22-,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as testosterone 6beta-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 in human liver microsomes |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 in human liver microsomes |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50146155

(CHEMBL3764774)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnc4ccccc4c3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C28H28N4O3/c33-26(17-32-18-29-24-9-5-4-8-23(24)28(32)35)31-21-12-10-19(11-13-21)16-22-14-15-25(30-22)27(34)20-6-2-1-3-7-20/h1-13,18,22,25,27,30,34H,14-17H2,(H,31,33)/t22-,25+,27+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes assessed as diclofenac alpha'-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50146156

(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes assessed as dextraomethorphan O-demethylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50338821

(2-(2-aminothiazol-4-yl)-N-(4-(((2S,5R)-5-((R)-hydr...)Show SMILES Nc1nc(CC(=O)Nc2ccc(C[C@@H]3CC[C@@H](N3)[C@H](O)c3ccccc3)cc2)cs1 |r| Show InChI InChI=1S/C23H26N4O2S/c24-23-27-19(14-30-23)13-21(28)26-17-8-6-15(7-9-17)12-18-10-11-20(25-18)22(29)16-4-2-1-3-5-16/h1-9,14,18,20,22,25,29H,10-13H2,(H2,24,27)(H,26,28)/t18-,20+,22+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes assessed as diclofenac alpha'-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50146158

(CHEMBL3763594)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cccnc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-14-4-13-25-24(28)31)27-19-9-7-17(8-10-19)15-20-11-12-21(26-20)23(30)18-5-2-1-3-6-18/h1-10,13-14,20-21,23,26,30H,11-12,15-16H2,(H,27,29)/t20-,21+,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes assessed as diclofenac alpha'-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes assessed as diclofenac alpha'-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50338821

(2-(2-aminothiazol-4-yl)-N-(4-(((2S,5R)-5-((R)-hydr...)Show SMILES Nc1nc(CC(=O)Nc2ccc(C[C@@H]3CC[C@@H](N3)[C@H](O)c3ccccc3)cc2)cs1 |r| Show InChI InChI=1S/C23H26N4O2S/c24-23-27-19(14-30-23)13-21(28)26-17-8-6-15(7-9-17)12-18-10-11-20(25-18)22(29)16-4-2-1-3-5-16/h1-9,14,18,20,22,25,29H,10-13H2,(H2,24,27)(H,26,28)/t18-,20+,22+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as testosterone 6beta-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50146158

(CHEMBL3763594)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cccnc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-14-4-13-25-24(28)31)27-19-9-7-17(8-10-19)15-20-11-12-21(26-20)23(30)18-5-2-1-3-6-18/h1-10,13-14,20-21,23,26,30H,11-12,15-16H2,(H,27,29)/t20-,21+,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as testosterone 6beta-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50146156

(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as testosterone 6beta-hydroxylation |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2B6
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of CYP2B6 in human liver microsomes |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146157

(CHEMBL3764950)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(15-28-16-25-13-12-23(28)30)27-19-8-6-17(7-9-19)14-20-10-11-21(26-20)24(31)18-4-2-1-3-5-18/h1-9,12-13,16,20-21,24,26,31H,10-11,14-15H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.90 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Macaca mulatta) | BDBM50092645

((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...)Show SMILES O[C@@H](CNCCc1ccc(NS(=O)(=O)c2ccc(cc2)-c2nc(cs2)-c2ccc(cc2)C(F)(F)F)cc1)c1cccnc1 Show InChI InChI=1S/C31H27F3N4O3S2/c32-31(33,34)25-9-5-22(6-10-25)28-20-42-30(37-28)23-7-13-27(14-8-23)43(40,41)38-26-11-3-21(4-12-26)15-17-36-19-29(39)24-2-1-16-35-18-24/h1-14,16,18,20,29,36,38-39H,15,17,19H2/t29-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.430 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at rhesus monkey beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.60 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human adrenergic beta3 receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay in presenc... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Canis familiaris) | BDBM50146160

(CHEMBL3765335)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H28N4O3/c1-17(29-23(30)8-5-15-26-29)25(32)28-20-11-9-18(10-12-20)16-21-13-14-22(27-21)24(31)19-6-3-2-4-7-19/h2-12,15,17,21-22,24,27,31H,13-14,16H2,1H3,(H,28,32)/t17-,21-,22+,24+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 39 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at dog beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146161

(CHEMBL3763998)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O3/c1-16(29-22(30)5-3-13-26-29)24(32)28-19-8-6-17(7-9-19)14-20-10-11-21(27-20)23(31)18-4-2-12-25-15-18/h2-9,12-13,15-16,20-21,23,27,31H,10-11,14H2,1H3,(H,28,32)/t16-,20-,21+,23+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.60 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Macaca mulatta) | BDBM50146160

(CHEMBL3765335)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H28N4O3/c1-17(29-23(30)8-5-15-26-29)25(32)28-20-11-9-18(10-12-20)16-21-13-14-22(27-21)24(31)19-6-3-2-4-7-19/h2-12,15,17,21-22,24,27,31H,13-14,16H2,1H3,(H,28,32)/t17-,21-,22+,24+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.5 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at rhesus monkey beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Rattus norvegicus) | BDBM50146160

(CHEMBL3765335)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H28N4O3/c1-17(29-23(30)8-5-15-26-29)25(32)28-20-11-9-18(10-12-20)16-21-13-14-22(27-21)24(31)19-6-3-2-4-7-19/h2-12,15,17,21-22,24,27,31H,13-14,16H2,1H3,(H,28,32)/t17-,21-,22+,24+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 245 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at rat beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay in presence ... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146156

(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 5.90 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human adrenergic beta3 receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay in presenc... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50448787

(CHEMBL3128178)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4scnc34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H26N4O2S/c29-23(16-2-1-11-25-13-16)20-9-7-18(27-20)12-15-3-5-17(6-4-15)28-24(30)19-8-10-21-22(19)26-14-31-21/h1-6,11,13-14,18-20,23,27,29H,7-10,12H2,(H,28,30)/t18-,19+,20+,23+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50448789

(CHEMBL3128188)Show SMILES O[C@@H]([C@H]1CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4ccnn34)cc2)N1)c1cccnc1 |r| Show InChI InChI=1S/C24H27N5O2/c30-23(17-2-1-12-25-15-17)21-9-7-19(27-21)14-16-3-5-18(6-4-16)28-24(31)22-10-8-20-11-13-26-29(20)22/h1-6,11-13,15,19,21-23,27,30H,7-10,14H2,(H,28,31)/t19-,21+,22-,23+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.70 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146156

(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 4.80 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146158

(CHEMBL3763594)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cccnc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-14-4-13-25-24(28)31)27-19-9-7-17(8-10-19)15-20-11-12-21(26-20)23(30)18-5-2-1-3-6-18/h1-10,13-14,20-21,23,26,30H,11-12,15-16H2,(H,27,29)/t20-,21+,23+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.20 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Rattus norvegicus) | BDBM50092645

((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...)Show SMILES O[C@@H](CNCCc1ccc(NS(=O)(=O)c2ccc(cc2)-c2nc(cs2)-c2ccc(cc2)C(F)(F)F)cc1)c1cccnc1 Show InChI InChI=1S/C31H27F3N4O3S2/c32-31(33,34)25-9-5-22(6-10-25)28-20-42-30(37-28)23-7-13-27(14-8-23)43(40,41)38-26-11-3-21(4-12-26)15-17-36-19-29(39)24-2-1-16-35-18-24/h1-14,16,18,20,29,36,38-39H,15,17,19H2/t29-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.40E+3 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at rat beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Macaca mulatta) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 7.10 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at rhesus monkey beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay in... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50092645

((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...)Show SMILES O[C@@H](CNCCc1ccc(NS(=O)(=O)c2ccc(cc2)-c2nc(cs2)-c2ccc(cc2)C(F)(F)F)cc1)c1cccnc1 Show InChI InChI=1S/C31H27F3N4O3S2/c32-31(33,34)25-9-5-22(6-10-25)28-20-42-30(37-28)23-7-13-27(14-8-23)43(40,41)38-26-11-3-21(4-12-26)15-17-36-19-29(39)24-2-1-16-35-18-24/h1-14,16,18,20,29,36,38-39H,15,17,19H2/t29-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 7.90 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human adrenergic beta3 receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay in presenc... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50092645

((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...)Show SMILES O[C@@H](CNCCc1ccc(NS(=O)(=O)c2ccc(cc2)-c2nc(cs2)-c2ccc(cc2)C(F)(F)F)cc1)c1cccnc1 Show InChI InChI=1S/C31H27F3N4O3S2/c32-31(33,34)25-9-5-22(6-10-25)28-20-42-30(37-28)23-7-13-27(14-8-23)43(40,41)38-26-11-3-21(4-12-26)15-17-36-19-29(39)24-2-1-16-35-18-24/h1-14,16,18,20,29,36,38-39H,15,17,19H2/t29-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Macaca mulatta) | BDBM50146156

(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.5 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at rhesus monkey beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Nuclear receptor subfamily 1 group I member 2
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Activation of human PX receptor |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Macaca mulatta) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.570 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at rhesus monkey beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Rattus norvegicus) | BDBM50146156

(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.06E+3 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at rat beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay in presence ... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146160

(CHEMBL3765335)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H28N4O3/c1-17(29-23(30)8-5-15-26-29)25(32)28-20-11-9-18(10-12-20)16-21-13-14-22(27-21)24(31)19-6-3-2-4-7-19/h2-12,15,17,21-22,24,27,31H,13-14,16H2,1H3,(H,28,32)/t17-,21-,22+,24+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.10 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human adrenergic beta3 receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay in presenc... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Canis familiaris) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 11 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at dog beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146162

(CHEMBL3763934)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@@H]3CCc4cccc(=O)n34)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C27H29N3O3/c31-25-8-4-7-22-14-16-24(30(22)25)27(33)29-20-11-9-18(10-12-20)17-21-13-15-23(28-21)26(32)19-5-2-1-3-6-19/h1-12,21,23-24,26,28,32H,13-17H2,(H,29,33)/t21-,23+,24-,26+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.620 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Macaca mulatta) | BDBM50146160

(CHEMBL3765335)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H28N4O3/c1-17(29-23(30)8-5-15-26-29)25(32)28-20-11-9-18(10-12-20)16-21-13-14-22(27-21)24(31)19-6-3-2-4-7-19/h2-12,15,17,21-22,24,27,31H,13-14,16H2,1H3,(H,28,32)/t17-,21-,22+,24+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 12 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at rhesus monkey beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay in... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Canis familiaris) | BDBM50092645

((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...)Show SMILES O[C@@H](CNCCc1ccc(NS(=O)(=O)c2ccc(cc2)-c2nc(cs2)-c2ccc(cc2)C(F)(F)F)cc1)c1cccnc1 Show InChI InChI=1S/C31H27F3N4O3S2/c32-31(33,34)25-9-5-22(6-10-25)28-20-42-30(37-28)23-7-13-27(14-8-23)43(40,41)38-26-11-3-21(4-12-26)15-17-36-19-29(39)24-2-1-16-35-18-24/h1-14,16,18,20,29,36,38-39H,15,17,19H2/t29-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 25 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at dog beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146160

(CHEMBL3765335)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H28N4O3/c1-17(29-23(30)8-5-15-26-29)25(32)28-20-11-9-18(10-12-20)16-21-13-14-22(27-21)24(31)19-6-3-2-4-7-19/h2-12,15,17,21-22,24,27,31H,13-14,16H2,1H3,(H,28,32)/t17-,21-,22+,24+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.70 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Rattus norvegicus) | BDBM50146156

(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 615 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at rat beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Rattus norvegicus) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 86 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at rat beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Rattus norvegicus) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 122 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at rat beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay in presence ... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Macaca mulatta) | BDBM50092645

((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...)Show SMILES O[C@@H](CNCCc1ccc(NS(=O)(=O)c2ccc(cc2)-c2nc(cs2)-c2ccc(cc2)C(F)(F)F)cc1)c1cccnc1 Show InChI InChI=1S/C31H27F3N4O3S2/c32-31(33,34)25-9-5-22(6-10-25)28-20-42-30(37-28)23-7-13-27(14-8-23)43(40,41)38-26-11-3-21(4-12-26)15-17-36-19-29(39)24-2-1-16-35-18-24/h1-14,16,18,20,29,36,38-39H,15,17,19H2/t29-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 17 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at rhesus monkey beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay in... |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Canis familiaris) | BDBM50146156

(CHEMBL3764088)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C24H26N4O3/c29-22(16-28-23(30)7-4-14-25-28)27-19-10-8-17(9-11-19)15-20-12-13-21(26-20)24(31)18-5-2-1-3-6-18/h1-11,14,20-21,24,26,31H,12-13,15-16H2,(H,27,29)/t20-,21+,24+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 47 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at dog beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50338821

(2-(2-aminothiazol-4-yl)-N-(4-(((2S,5R)-5-((R)-hydr...)Show SMILES Nc1nc(CC(=O)Nc2ccc(C[C@@H]3CC[C@@H](N3)[C@H](O)c3ccccc3)cc2)cs1 |r| Show InChI InChI=1S/C23H26N4O2S/c24-23-27-19(14-30-23)13-21(28)26-17-8-6-15(7-9-17)12-18-10-11-20(25-18)22(29)16-4-2-1-3-5-16/h1-9,14,18,20,22,25,29H,10-13H2,(H2,24,27)(H,26,28)/t18-,20+,22+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.900 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146155

(CHEMBL3764774)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3cnc4ccccc4c3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C28H28N4O3/c33-26(17-32-18-29-24-9-5-4-8-23(24)28(32)35)31-21-12-10-19(11-13-21)16-22-14-15-25(30-22)27(34)20-6-2-1-3-7-20/h1-13,18,22,25,27,30,34H,14-17H2,(H,31,33)/t22-,25+,27+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.70 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146154

(KRP-114V | MK-4618 | Vibegron)Show SMILES [H][C@@](O)(c1ccccc1)[C@@]1([H])CC[C@@]([H])(Cc2ccc(NC(=O)[C@]3([H])CCc4nccc(=O)n34)cc2)N1 |r| Show InChI InChI=1S/C26H28N4O3/c31-24-14-15-27-23-13-12-22(30(23)24)26(33)29-19-8-6-17(7-9-19)16-20-10-11-21(28-20)25(32)18-4-2-1-3-5-18/h1-9,14-15,20-22,25,28,32H,10-13,16H2,(H,29,33)/t20-,21+,22-,25+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Rattus norvegicus) | BDBM50146160

(CHEMBL3765335)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)[C@H](C)n3ncccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H28N4O3/c1-17(29-23(30)8-5-15-26-29)25(32)28-20-11-9-18(10-12-20)16-21-13-14-22(27-21)24(31)19-6-3-2-4-7-19/h2-12,15,17,21-22,24,27,31H,13-14,16H2,1H3,(H,28,32)/t17-,21-,22+,24+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 531 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at rat beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50146159

(CHEMBL3764592)Show SMILES [H][C@@]1(CC[C@@H](Cc2ccc(NC(=O)Cn3ccccc3=O)cc2)N1)[C@H](O)c1ccccc1 |r| Show InChI InChI=1S/C25H27N3O3/c29-23(17-28-15-5-4-8-24(28)30)27-20-11-9-18(10-12-20)16-21-13-14-22(26-21)25(31)19-6-2-1-3-7-19/h1-12,15,21-22,25-26,31H,13-14,16-17H2,(H,27,29)/t21-,22+,25+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 4.30 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay |
J Med Chem 59: 609-23 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01372
BindingDB Entry DOI: 10.7270/Q2M047B3 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data