

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204751 (CHEMBL3943450) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM8961 (1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204754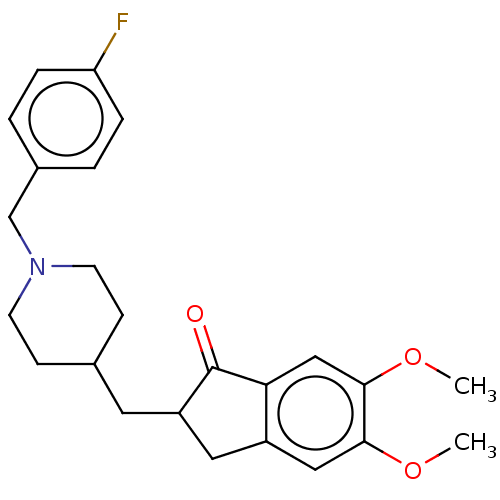 (CHEMBL3985059) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204751 (CHEMBL3943450) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 43 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM8961 (1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 46 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM8960 ((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM8960 ((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 51 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204831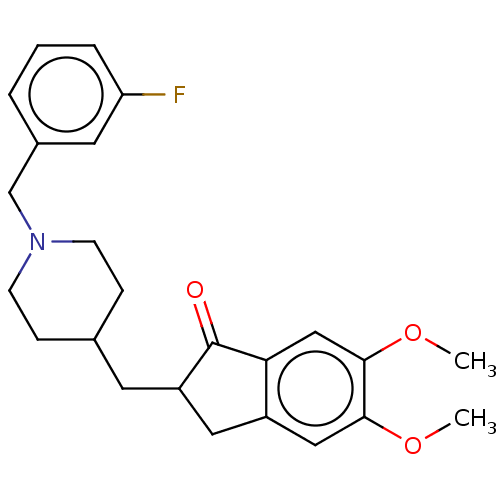 (CHEMBL3927363) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 54 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204835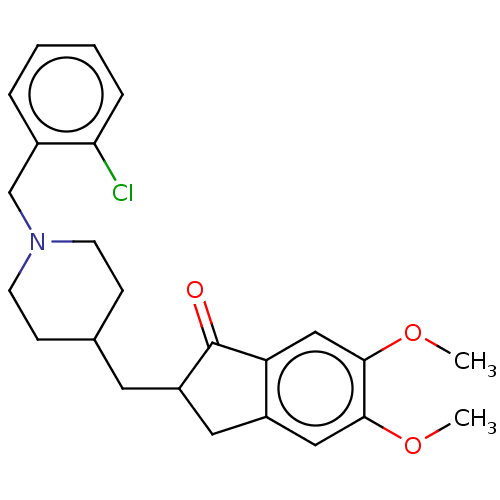 (CHEMBL3933395) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204831 (CHEMBL3927363) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 68 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204752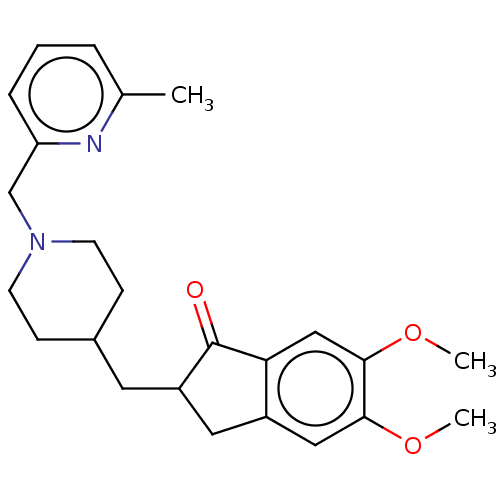 (CHEMBL3919441) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 73 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204752 (CHEMBL3919441) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 85 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50029944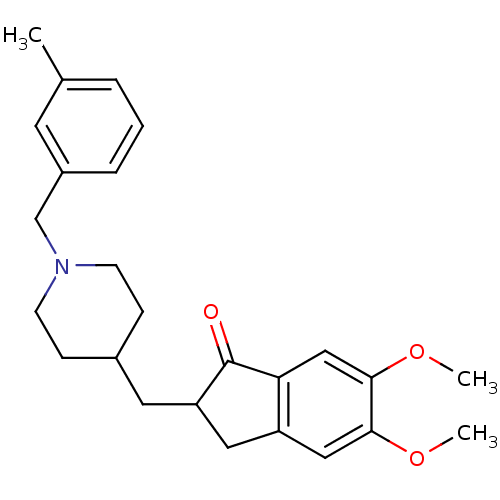 (5,6-Dimethoxy-2-[1-(3-methyl-benzyl)-piperidin-4-y...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 88 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204835 (CHEMBL3933395) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 97 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50029921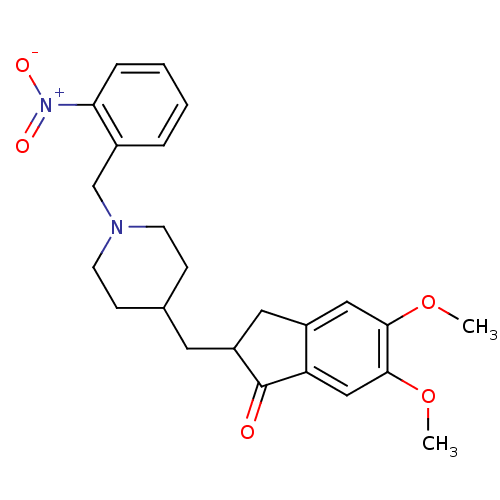 (5,6-Dimethoxy-2-[1-(2-nitro-benzyl)-piperidin-4-yl...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 106 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204758 (CHEMBL3891369) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 108 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50029944 (5,6-Dimethoxy-2-[1-(3-methyl-benzyl)-piperidin-4-y...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 108 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204758 (CHEMBL3891369) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 113 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204757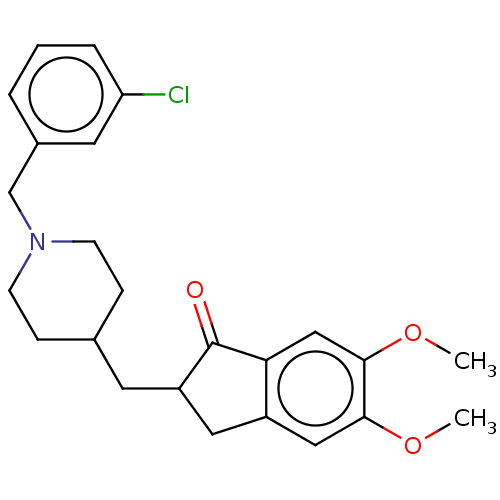 (CHEMBL3960861) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 114 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50029937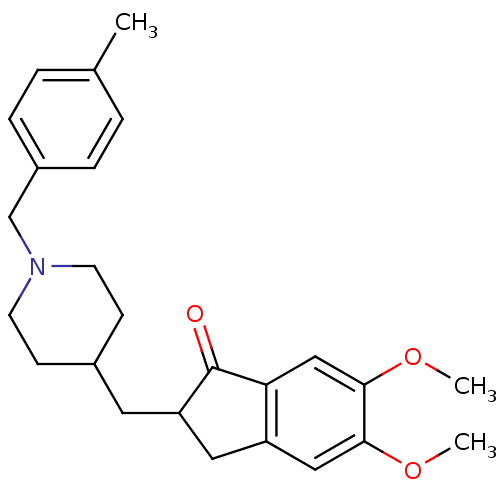 (5,6-Dimethoxy-2-[1-(4-methyl-benzyl)-piperidin-4-y...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 127 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204754 (CHEMBL3985059) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204755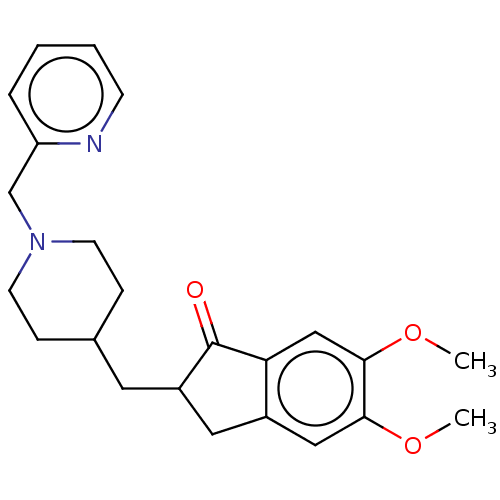 (CHEMBL3910513) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 147 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50029945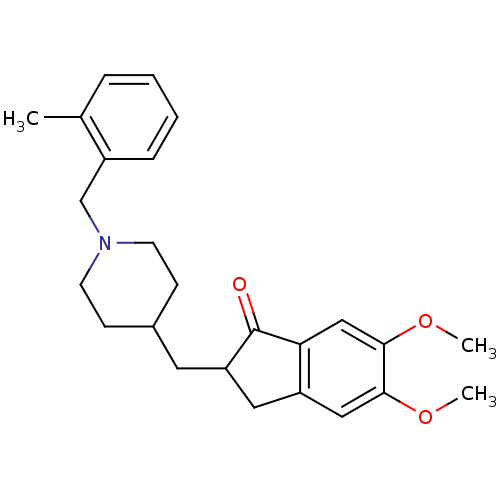 (5,6-Dimethoxy-2-[1-(2-methyl-benzyl)-piperidin-4-y...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 184 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204830 (CHEMBL3928343) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 195 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204829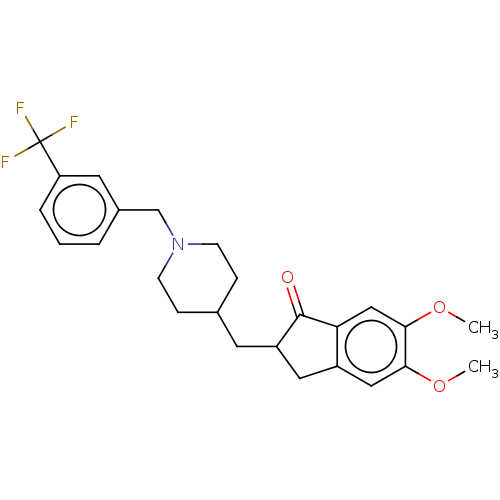 (CHEMBL3961494) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 197 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM8961 (1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 218 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50029937 (5,6-Dimethoxy-2-[1-(4-methyl-benzyl)-piperidin-4-y...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 221 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204757 (CHEMBL3960861) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 237 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50029945 (5,6-Dimethoxy-2-[1-(2-methyl-benzyl)-piperidin-4-y...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 243 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204830 (CHEMBL3928343) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 246 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204838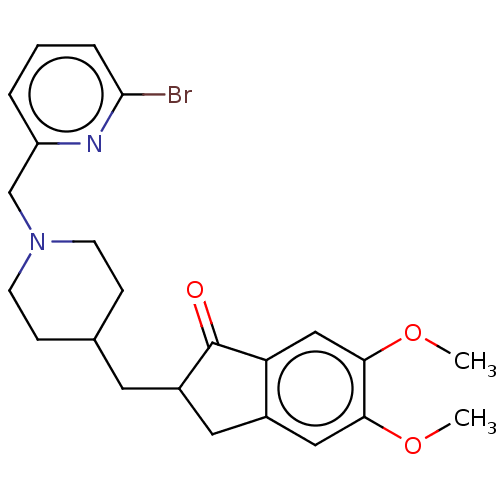 (CHEMBL3973337) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 253 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204761 (CHEMBL3944652) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 409 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50029932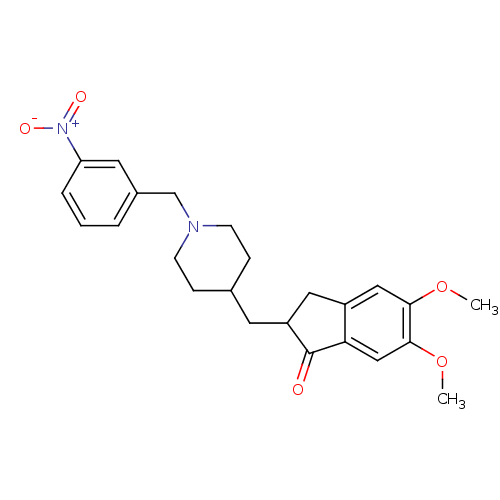 (5,6-Dimethoxy-2-[1-(3-nitro-benzyl)-piperidin-4-yl...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 414 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM8961 (1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 459 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204750 (CHEMBL3889713) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 511 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50029932 (5,6-Dimethoxy-2-[1-(3-nitro-benzyl)-piperidin-4-yl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 652 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204760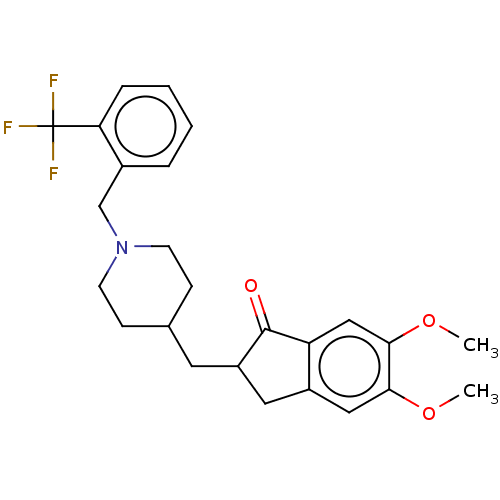 (CHEMBL3890758) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204837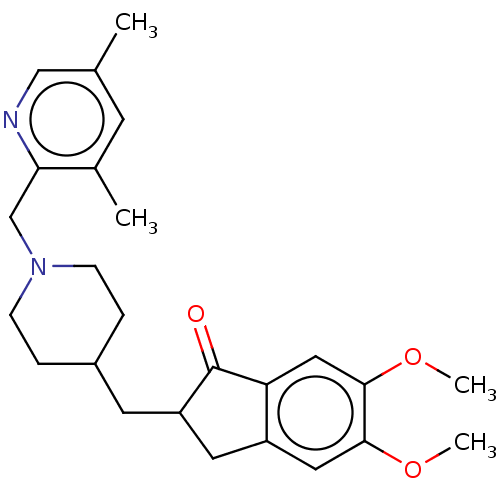 (CHEMBL3900401) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 814 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204761 (CHEMBL3944652) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 883 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204837 (CHEMBL3900401) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.01E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204755 (CHEMBL3910513) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.09E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50029934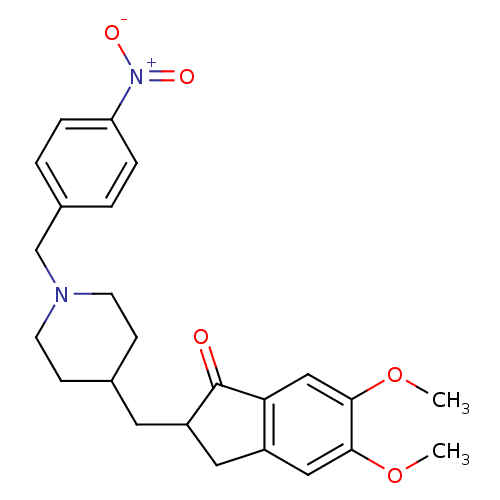 (5,6-Dimethoxy-2-[1-(4-nitro-benzyl)-piperidin-4-yl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.91E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204838 (CHEMBL3973337) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.98E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204833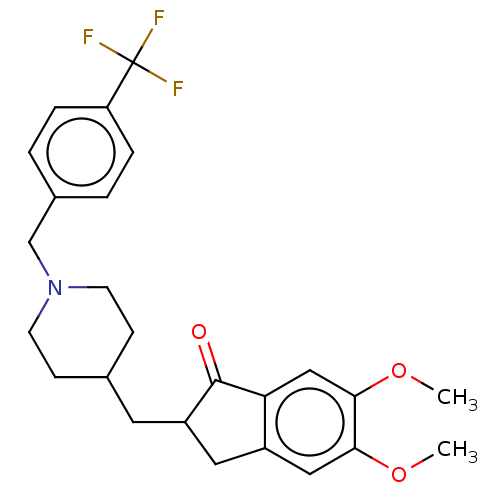 (CHEMBL3892502) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.03E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204829 (CHEMBL3961494) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.12E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204754 (CHEMBL3985059) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.31E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM8960 ((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.48E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204831 (CHEMBL3927363) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.66E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204836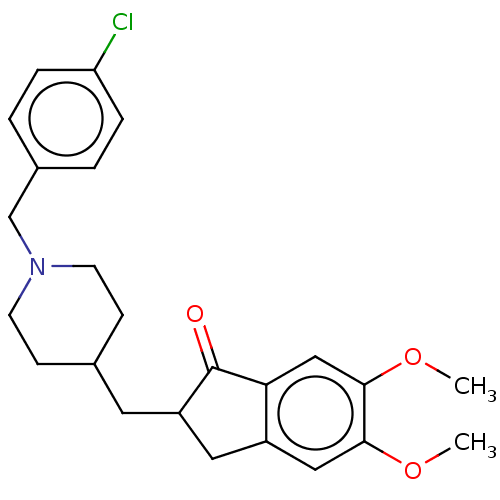 (CHEMBL3926911) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.86E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50029921 (5,6-Dimethoxy-2-[1-(2-nitro-benzyl)-piperidin-4-yl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.11E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM8960 ((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 3.17E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204835 (CHEMBL3933395) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204751 (CHEMBL3943450) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.71E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204833 (CHEMBL3892502) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3.98E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50029934 (5,6-Dimethoxy-2-[1-(4-nitro-benzyl)-piperidin-4-yl...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.02E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204756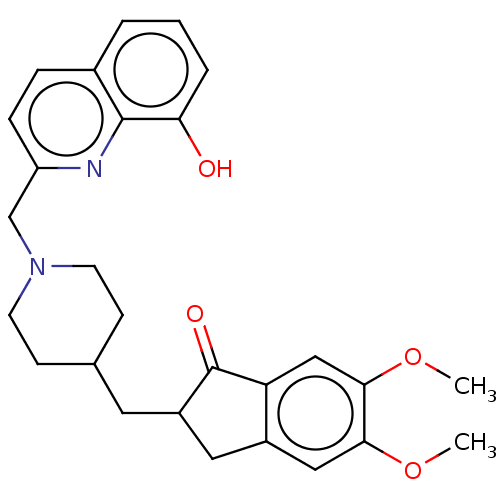 (CHEMBL3934564) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.53E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204836 (CHEMBL3926911) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204760 (CHEMBL3890758) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.87E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204833 (CHEMBL3892502) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 5.43E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204750 (CHEMBL3889713) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.78E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50029944 (5,6-Dimethoxy-2-[1-(3-methyl-benzyl)-piperidin-4-y...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.83E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204837 (CHEMBL3900401) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 6.31E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204759 (CHEMBL3889625) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.35E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204756 (CHEMBL3934564) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.42E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50029937 (5,6-Dimethoxy-2-[1-(4-methyl-benzyl)-piperidin-4-y...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.67E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204753 (CHEMBL3950063) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7.65E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204755 (CHEMBL3910513) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 7.83E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204754 (CHEMBL3985059) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 8.13E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204759 (CHEMBL3889625) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 8.43E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204837 (CHEMBL3900401) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 8.55E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204759 (CHEMBL3889625) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 8.71E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204759 (CHEMBL3889625) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 9.68E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50029934 (5,6-Dimethoxy-2-[1-(4-nitro-benzyl)-piperidin-4-yl...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.22E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204751 (CHEMBL3943450) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.28E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204758 (CHEMBL3891369) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.56E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204838 (CHEMBL3973337) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.75E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204829 (CHEMBL3961494) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.82E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204835 (CHEMBL3933395) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.88E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204752 (CHEMBL3919441) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.05E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204753 (CHEMBL3950063) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.09E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204830 (CHEMBL3928343) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.11E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204750 (CHEMBL3889713) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.54E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204831 (CHEMBL3927363) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.67E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50204753 (CHEMBL3950063) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.73E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to 3 m... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50029944 (5,6-Dimethoxy-2-[1-(3-methyl-benzyl)-piperidin-4-y...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.81E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204750 (CHEMBL3889713) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.95E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50204756 (CHEMBL3934564) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.98E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human erythrocytes AChE using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50029921 (5,6-Dimethoxy-2-[1-(2-nitro-benzyl)-piperidin-4-yl...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.11E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204761 (CHEMBL3944652) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.16E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204760 (CHEMBL3890758) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.17E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204756 (CHEMBL3934564) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.74E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50029945 (5,6-Dimethoxy-2-[1-(2-methyl-benzyl)-piperidin-4-y...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.81E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204755 (CHEMBL3910513) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.86E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204830 (CHEMBL3928343) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3.89E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204760 (CHEMBL3890758) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.14E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50029932 (5,6-Dimethoxy-2-[1-(3-nitro-benzyl)-piperidin-4-yl...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.24E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50029937 (5,6-Dimethoxy-2-[1-(4-methyl-benzyl)-piperidin-4-y...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.38E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204758 (CHEMBL3891369) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.92E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204757 (CHEMBL3960861) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.02E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204752 (CHEMBL3919441) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.57E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204757 (CHEMBL3960861) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.89E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204829 (CHEMBL3961494) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 7.69E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204761 (CHEMBL3944652) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7.87E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50204836 (CHEMBL3926911) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 8.16E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50029934 (5,6-Dimethoxy-2-[1-(4-nitro-benzyl)-piperidin-4-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 8.58E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50204836 (CHEMBL3926911) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 8.94E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50029921 (5,6-Dimethoxy-2-[1-(2-nitro-benzyl)-piperidin-4-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 9.12E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human serum BuChE using s-butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured up to ... | Eur J Med Chem 123: 282-297 (2016) Article DOI: 10.1016/j.ejmech.2016.07.052 BindingDB Entry DOI: 10.7270/Q2ZW1NW5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||