| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Protein-arginine deiminase type-4 |
|---|
| Ligand | BDBM646153 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | In Vitro PAD4 BAEE Biochemical Assay |
|---|
| IC50 | 27.6±n/a nM |
|---|
| Citation |  Byun, DH; Cai, ZR; Canales, EY; Debien, LP; Hansen, TR; Huang, R; Jansa, P; Lee, RA; Loyer-Drew, JA; McFadden, R; Mitchell, ML; Pyun, H; Saito, RD; Sangi, MS; Schrier, AJ; Shatskikh, ME; Taylor, JG; Van Veldhuizen, JJ; Xu, L Inhibitors of peptidylarginine deiminases US Patent US11878965 Publication Date 1/23/2024 Byun, DH; Cai, ZR; Canales, EY; Debien, LP; Hansen, TR; Huang, R; Jansa, P; Lee, RA; Loyer-Drew, JA; McFadden, R; Mitchell, ML; Pyun, H; Saito, RD; Sangi, MS; Schrier, AJ; Shatskikh, ME; Taylor, JG; Van Veldhuizen, JJ; Xu, L Inhibitors of peptidylarginine deiminases US Patent US11878965 Publication Date 1/23/2024 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Protein-arginine deiminase type-4 |
|---|
| Name: | Protein-arginine deiminase type-4 |
|---|
| Synonyms: | PAD4 | PADI4 | PADI4_HUMAN | PADI5 | PDI5 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 74078.90 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | gi_216548487 |
|---|
| Residue: | 663 |
|---|
| Sequence: | MAQGTLIRVTPEQPTHAVCVLGTLTQLDICSSAPEDCTSFSINASPGVVVDIAHGPPAKK
KSTGSSTWPLDPGVEVTLTMKVASGSTGDQKVQISYYGPKTPPVKALLYLTGVEISLCAD
ITRTGKVKPTRAVKDQRTWTWGPCGQGAILLVNCDRDNLESSAMDCEDDEVLDSEDLQDM
SLMTLSTKTPKDFFTNHTLVLHVARSEMDKVRVFQATRGKLSSKCSVVLGPKWPSHYLMV
PGGKHNMDFYVEALAFPDTDFPGLITLTISLLDTSNLELPEAVVFQDSVVFRVAPWIMTP
NTQPPQEVYACSIFENEDFLKSVTTLAMKAKCKLTICPEEENMDDQWMQDEMEIGYIQAP
HKTLPVVFDSPRNRGLKEFPIKRVMGPDFGYVTRGPQTGGISGLDSFGNLEVSPPVTVRG
KEYPLGRILFGDSCYPSNDSRQMHQALQDFLSAQQVQAPVKLYSDWLSVGHVDEFLSFVP
APDRKGFRLLLASPRSCYKLFQEQQNEGHGEALLFEGIKKKKQQKIKNILSNKTLREHNS
FVERCIDWNRELLKRELGLAESDIIDIPQLFKLKEFSKAEAFFPNMVNMLVLGKHLGIPK
PFGPVINGRCCLEEKVCSLLEPLGLQCTFINDFFTYHIRHGEVHCGTNVRRKPFSFKWWN
MVP
|
|
|
|---|
| BDBM646153 |
|---|
| n/a |
|---|
| Name | BDBM646153 |
|---|
| Synonyms: | N-((R)-1-(2-(5-((1R,2R,4S)-2- amino-7-azabicyclo[2.2.1]heptane-7- carbonyl)-7-methoxy-1-methyl-1H- benzo[d]imidazol-2-yl)-1- (cyclopropylmethyl)-1H-indol-6- yl)ethyl)-2-methyloxazole-5- carboxamide | US11878965, Example 138 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C35H39N7O4 |
|---|
| Mol. Mass. | 621.7287 |
|---|
| SMILES | COc1cc(cc2nc(-c3cc4ccc(cc4n3CC3CC3)[C@@H](C)NC(=O)c3cnc(C)o3)n(C)c12)C(=O)N1[C@H]2CC[C@@H]1[C@H](N)C2 |r,THB:36:38:43.45:41.40| |
|---|
| Structure | 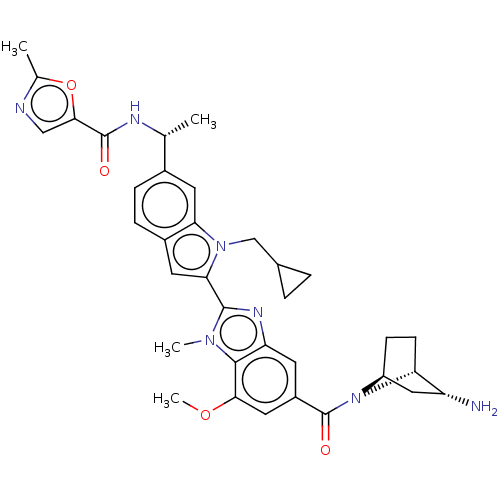
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Byun, DH; Cai, ZR; Canales, EY; Debien, LP; Hansen, TR; Huang, R; Jansa, P; Lee, RA; Loyer-Drew, JA; McFadden, R; Mitchell, ML; Pyun, H; Saito, RD; Sangi, MS; Schrier, AJ; Shatskikh, ME; Taylor, JG; Van Veldhuizen, JJ; Xu, L Inhibitors of peptidylarginine deiminases US Patent US11878965 Publication Date 1/23/2024
Byun, DH; Cai, ZR; Canales, EY; Debien, LP; Hansen, TR; Huang, R; Jansa, P; Lee, RA; Loyer-Drew, JA; McFadden, R; Mitchell, ML; Pyun, H; Saito, RD; Sangi, MS; Schrier, AJ; Shatskikh, ME; Taylor, JG; Van Veldhuizen, JJ; Xu, L Inhibitors of peptidylarginine deiminases US Patent US11878965 Publication Date 1/23/2024