| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Plasminogen |
|---|
| Ligand | BDBM16168 |
|---|
| Substrate/Competitor | BDBM12679 |
|---|
| Meas. Tech. | Determination of Inhibitor Potency and Selectivity |
|---|
| Ki | >100000±n/a nM |
|---|
| Citation |  Fish, PV; Barber, CG; Brown, DG; Butt, R; Collis, MG; Dickinson, RP; Henry, BT; Horne, VA; Huggins, JP; King, E; O'gara, M; McCleverty, D; McIntosh, F; Phillips, C; Webster, R Selective Urokinase-Type Plasminogen Activator Inhibitors. 4. 1-(7-Sulfonamidoisoquinolinyl)guanidines. J Med Chem50:2341-51 (2007) [PubMed] Article Fish, PV; Barber, CG; Brown, DG; Butt, R; Collis, MG; Dickinson, RP; Henry, BT; Horne, VA; Huggins, JP; King, E; O'gara, M; McCleverty, D; McIntosh, F; Phillips, C; Webster, R Selective Urokinase-Type Plasminogen Activator Inhibitors. 4. 1-(7-Sulfonamidoisoquinolinyl)guanidines. J Med Chem50:2341-51 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Plasminogen |
|---|
| Name: | Plasminogen |
|---|
| Synonyms: | Activation peptide | Angiostatin | PLG | PLMN_HUMAN | Plasmin | Plasmin heavy chain A | Plasmin heavy chain A, short form | Plasmin light chain B |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 90579.18 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 810 |
|---|
| Sequence: | MEHKEVVLLLLLFLKSGQGEPLDDYVNTQGASLFSVTKKQLGAGSIEECAAKCEEDEEFT
CRAFQYHSKEQQCVIMAENRKSSIIIRMRDVVLFEKKVYLSECKTGNGKNYRGTMSKTKN
GITCQKWSSTSPHRPRFSPATHPSEGLEENYCRNPDNDPQGPWCYTTDPEKRYDYCDILE
CEEECMHCSGENYDGKISKTMSGLECQAWDSQSPHAHGYIPSKFPNKNLKKNYCRNPDRE
LRPWCFTTDPNKRWELCDIPRCTTPPPSSGPTYQCLKGTGENYRGNVAVTVSGHTCQHWS
AQTPHTHNRTPENFPCKNLDENYCRNPDGKRAPWCHTTNSQVRWEYCKIPSCDSSPVSTE
QLAPTAPPELTPVVQDCYHGDGQSYRGTSSTTTTGKKCQSWSSMTPHRHQKTPENYPNAG
LTMNYCRNPDADKGPWCFTTDPSVRWEYCNLKKCSGTEASVVAPPPVVLLPDVETPSEED
CMFGNGKGYRGKRATTVTGTPCQDWAAQEPHRHSIFTPETNPRAGLEKNYCRNPDGDVGG
PWCYTTNPRKLYDYCDVPQCAAPSFDCGKPQVEPKKCPGRVVGGCVAHPHSWPWQVSLRT
RFGMHFCGGTLISPEWVLTAAHCLEKSPRPSSYKVILGAHQEVNLEPHVQEIEVSRLFLE
PTRKDIALLKLSSPAVITDKVIPACLPSPNYVVADRTECFITGWGETQGTFGAGLLKEAQ
LPVIENKVCNRYEFLNGRVQSTELCAGHLAGGTDSCQGDSGGPLVCFEKDKYILQGVTSW
GLGCARPNKPGVYVRVSRFVTWIEGVMRNN
|
|
|
|---|
| BDBM16168 |
|---|
| BDBM12679 |
|---|
| Name | BDBM16168 |
|---|
| Synonyms: | 1-({4-chloro-1-[(diaminomethylidene)amino]isoquinoline-7-}sulfonamido)-N-[2-(dimethylamino)ethyl]cyclopentane-1-carboxamide | cycloleucine deriv. 42 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H28ClN7O3S |
|---|
| Mol. Mass. | 481.999 |
|---|
| SMILES | [#6]-[#7](-[#6])-[#6]-[#6]-[#7]-[#6](=O)C1([#6]-[#6]-[#6]-[#6]1)[#7]S(=O)(=O)c1ccc2c(Cl)cnc(\[#7]=[#6](/[#7])-[#7])c2c1 |
|---|
| Structure | 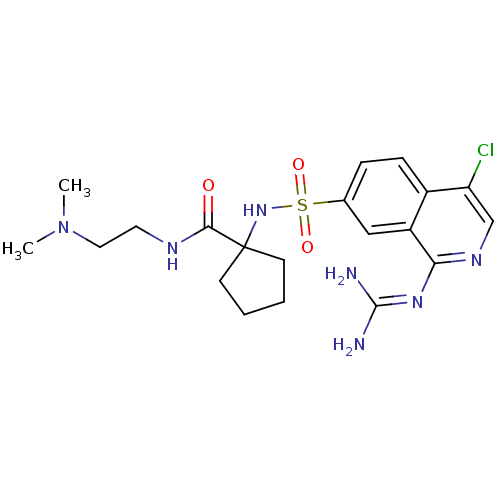
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fish, PV; Barber, CG; Brown, DG; Butt, R; Collis, MG; Dickinson, RP; Henry, BT; Horne, VA; Huggins, JP; King, E; O'gara, M; McCleverty, D; McIntosh, F; Phillips, C; Webster, R Selective Urokinase-Type Plasminogen Activator Inhibitors. 4. 1-(7-Sulfonamidoisoquinolinyl)guanidines. J Med Chem50:2341-51 (2007) [PubMed] Article
Fish, PV; Barber, CG; Brown, DG; Butt, R; Collis, MG; Dickinson, RP; Henry, BT; Horne, VA; Huggins, JP; King, E; O'gara, M; McCleverty, D; McIntosh, F; Phillips, C; Webster, R Selective Urokinase-Type Plasminogen Activator Inhibitors. 4. 1-(7-Sulfonamidoisoquinolinyl)guanidines. J Med Chem50:2341-51 (2007) [PubMed] Article