| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2B6 |
|---|
| Ligand | BDBM50250181 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_1681250 |
|---|
| IC50 | >20000±n/a nM |
|---|
| Citation |  Dos Santos Fernandes, GF; de Souza, PC; Moreno-Viguri, E; Santiva�ez-Veliz, M; Paucar, R; P�rez-Silanes, S; Chegaev, K; Guglielmo, S; Lazzarato, L; Fruttero, R; Man Chin, C; da Silva, PB; Chorilli, M; Solcia, MC; Ribeiro, CM; Silva, CSP; Marino, LB; Bosquesi, PL; Hunt, DM; de Carvalho, LPS; de Souza Costa, CA; Cho, SH; Wang, Y; Franzblau, SG; Pavan, FR; Dos Santos, JL Design, Synthesis, and Characterization of N-Oxide-Containing Heterocycles with in Vivo Sterilizing Antitubercular Activity. J Med Chem60:8647-8660 (2017) [PubMed] Article Dos Santos Fernandes, GF; de Souza, PC; Moreno-Viguri, E; Santiva�ez-Veliz, M; Paucar, R; P�rez-Silanes, S; Chegaev, K; Guglielmo, S; Lazzarato, L; Fruttero, R; Man Chin, C; da Silva, PB; Chorilli, M; Solcia, MC; Ribeiro, CM; Silva, CSP; Marino, LB; Bosquesi, PL; Hunt, DM; de Carvalho, LPS; de Souza Costa, CA; Cho, SH; Wang, Y; Franzblau, SG; Pavan, FR; Dos Santos, JL Design, Synthesis, and Characterization of N-Oxide-Containing Heterocycles with in Vivo Sterilizing Antitubercular Activity. J Med Chem60:8647-8660 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2B6 |
|---|
| Name: | Cytochrome P450 2B6 |
|---|
| Synonyms: | CP2B6_HUMAN | CYP2B6 | Cytochrome P450 2B6 (CYP2B6) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 56289.75 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P20813 |
|---|
| Residue: | 491 |
|---|
| Sequence: | MELSVLLFLALLTGLLLLLVQRHPNTHDRLPPGPRPLPLLGNLLQMDRRGLLKSFLRFRE
KYGDVFTVHLGPRPVVMLCGVEAIREALVDKAEAFSGRGKIAMVDPFFRGYGVIFANGNR
WKVLRRFSVTTMRDFGMGKRSVEERIQEEAQCLIEELRKSKGALMDPTFLFQSITANIIC
SIVFGKRFHYQDQEFLKMLNLFYQTFSLISSVFGQLFELFSGFLKYFPGAHRQVYKNLQE
INAYIGHSVEKHRETLDPSAPKDLIDTYLLHMEKEKSNAHSEFSHQNLNLNTLSLFFAGT
ETTSTTLRYGFLLMLKYPHVAERVYREIEQVIGPHRPPELHDRAKMPYTEAVIYEIQRFS
DLLPMGVPHIVTQHTSFRGYIIPKDTEVFLILSTALHDPHYFEKPDAFNPDHFLDANGAL
KKTEAFIPFSLGKRICLGEGIARAELFLFFTTILQNFSMASPVAPEDIDLTPQECGVGKI
PPTYQIRFLPR
|
|
|
|---|
| BDBM50250181 |
|---|
| n/a |
|---|
| Name | BDBM50250181 |
|---|
| Synonyms: | CHEMBL2338421 | N''-(5-Benzofuroxanylmethylidene)Isoniazide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C13H9N5O3 |
|---|
| Mol. Mass. | 283.2423 |
|---|
| SMILES | [O-][n+]1onc2ccc(\C=N\NC(=O)c3ccncc3)cc12 |
|---|
| Structure | 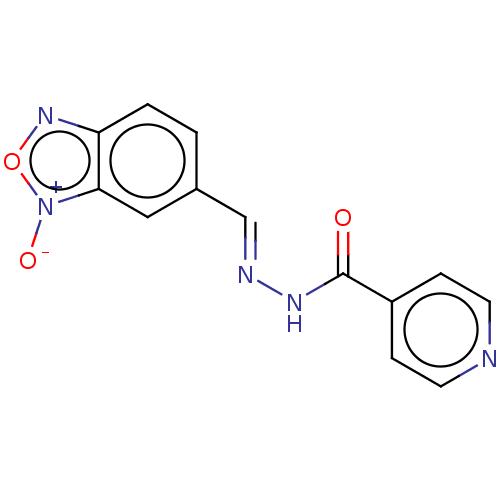
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Dos Santos Fernandes, GF; de Souza, PC; Moreno-Viguri, E; Santiva�ez-Veliz, M; Paucar, R; P�rez-Silanes, S; Chegaev, K; Guglielmo, S; Lazzarato, L; Fruttero, R; Man Chin, C; da Silva, PB; Chorilli, M; Solcia, MC; Ribeiro, CM; Silva, CSP; Marino, LB; Bosquesi, PL; Hunt, DM; de Carvalho, LPS; de Souza Costa, CA; Cho, SH; Wang, Y; Franzblau, SG; Pavan, FR; Dos Santos, JL Design, Synthesis, and Characterization of N-Oxide-Containing Heterocycles with in Vivo Sterilizing Antitubercular Activity. J Med Chem60:8647-8660 (2017) [PubMed] Article
Dos Santos Fernandes, GF; de Souza, PC; Moreno-Viguri, E; Santiva�ez-Veliz, M; Paucar, R; P�rez-Silanes, S; Chegaev, K; Guglielmo, S; Lazzarato, L; Fruttero, R; Man Chin, C; da Silva, PB; Chorilli, M; Solcia, MC; Ribeiro, CM; Silva, CSP; Marino, LB; Bosquesi, PL; Hunt, DM; de Carvalho, LPS; de Souza Costa, CA; Cho, SH; Wang, Y; Franzblau, SG; Pavan, FR; Dos Santos, JL Design, Synthesis, and Characterization of N-Oxide-Containing Heterocycles with in Vivo Sterilizing Antitubercular Activity. J Med Chem60:8647-8660 (2017) [PubMed] Article