| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Myeloblastin |
|---|
| Ligand | BDBM50263166 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_1697682 |
|---|
| Ki | 5.4±n/a nM |
|---|
| Citation |  Guarino, C; Gruba, N; Grzywa, R; Dyguda-Kazimierowicz, E; Hamon, Y; ??gowska, M; Skore?ski, M; Dallet-Choisy, S; Marchand-Adam, S; Kellenberger, C; Jenne, DE; Sie?czyk, M; Lesner, A; Gauthier, F; Korkmaz, B Exploiting the S4-S5 Specificity of Human Neutrophil Proteinase 3 to Improve the Potency of Peptidyl Di(chlorophenyl)-phosphonate Ester Inhibitors: A Kinetic and Molecular Modeling Analysis. J Med Chem61:1858-1870 (2018) [PubMed] Article Guarino, C; Gruba, N; Grzywa, R; Dyguda-Kazimierowicz, E; Hamon, Y; ??gowska, M; Skore?ski, M; Dallet-Choisy, S; Marchand-Adam, S; Kellenberger, C; Jenne, DE; Sie?czyk, M; Lesner, A; Gauthier, F; Korkmaz, B Exploiting the S4-S5 Specificity of Human Neutrophil Proteinase 3 to Improve the Potency of Peptidyl Di(chlorophenyl)-phosphonate Ester Inhibitors: A Kinetic and Molecular Modeling Analysis. J Med Chem61:1858-1870 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Myeloblastin |
|---|
| Name: | Myeloblastin |
|---|
| Synonyms: | Leukocyte proteinase 3 | MBN | PRTN3 | PRTN3_HUMAN |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 27816.30 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1463425 |
|---|
| Residue: | 256 |
|---|
| Sequence: | MAHRPPSPALASVLLALLLSGAARAAEIVGGHEAQPHSRPYMASLQMRGNPGSHFCGGTL
IHPSFVLTAAHCLRDIPQRLVNVVLGAHNVRTQEPTQQHFSVAQVFLNNYDAENKLNDVL
LIQLSSPANLSASVATVQLPQQDQPVPHGTQCLAMGWGRVGAHDPPAQVLQELNVTVVTF
FCRPHNICTFVPRRKAGICFGDSGGPLICDGIIQGIDSFVIWGCATRLFPDFFTRVALYV
DWIRSTLRRVEAKGRP
|
|
|
|---|
| BDBM50263166 |
|---|
| n/a |
|---|
| Name | BDBM50263166 |
|---|
| Synonyms: | CHEMBL4071346 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C44H55Cl2N6O11PS |
|---|
| Mol. Mass. | 977.886 |
|---|
| SMILES | [H][C@]12CS[C@@H](CCCCC(=O)N[C@@H](C(C)C)C(=O)N[C@@H](Cc3ccc(O)cc3)C(=O)N[C@@H](CC(O)=O)C(=O)NC(CCC)P(=O)(Oc3ccc(Cl)cc3)Oc3ccc(Cl)cc3)[C@@]1([H])NC(=O)N2 |r| |
|---|
| Structure | 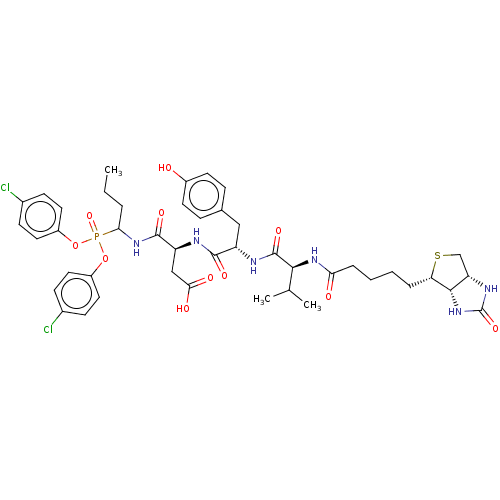
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Guarino, C; Gruba, N; Grzywa, R; Dyguda-Kazimierowicz, E; Hamon, Y; ??gowska, M; Skore?ski, M; Dallet-Choisy, S; Marchand-Adam, S; Kellenberger, C; Jenne, DE; Sie?czyk, M; Lesner, A; Gauthier, F; Korkmaz, B Exploiting the S4-S5 Specificity of Human Neutrophil Proteinase 3 to Improve the Potency of Peptidyl Di(chlorophenyl)-phosphonate Ester Inhibitors: A Kinetic and Molecular Modeling Analysis. J Med Chem61:1858-1870 (2018) [PubMed] Article
Guarino, C; Gruba, N; Grzywa, R; Dyguda-Kazimierowicz, E; Hamon, Y; ??gowska, M; Skore?ski, M; Dallet-Choisy, S; Marchand-Adam, S; Kellenberger, C; Jenne, DE; Sie?czyk, M; Lesner, A; Gauthier, F; Korkmaz, B Exploiting the S4-S5 Specificity of Human Neutrophil Proteinase 3 to Improve the Potency of Peptidyl Di(chlorophenyl)-phosphonate Ester Inhibitors: A Kinetic and Molecular Modeling Analysis. J Med Chem61:1858-1870 (2018) [PubMed] Article