| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2B |
|---|
| Ligand | BDBM50456162 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1755208 (CHEMBL4189968) |
|---|
| Ki | 2780±n/a nM |
|---|
| Citation |  Yu, J; Ciancetta, A; Dudas, S; Duca, S; Lottermoser, J; Jacobson, KA Structure-Guided Modification of Heterocyclic Antagonists of the P2Y J Med Chem61:4860-4882 (2018) [PubMed] Article Yu, J; Ciancetta, A; Dudas, S; Duca, S; Lottermoser, J; Jacobson, KA Structure-Guided Modification of Heterocyclic Antagonists of the P2Y J Med Chem61:4860-4882 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2B |
|---|
| Name: | 5-hydroxytryptamine receptor 2B |
|---|
| Synonyms: | 5-HT-2B | 5-HT2B | 5-hydroxytryptamine (serotonin) receptor 2B [Homo sapiens] | 5-hydroxytryptamine receptor 2B (5-HT2B) | 5-hydroxytryptamine receptor 2C (5HT2C) | 5HT2B_HUMAN | HTR2B | Serotonin (5-HT3) receptor | Serotonin 2b (5-HT2b) receptor | Serotonin Receptor 2B |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 54312.47 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Receptor binding assays were performed using human clone stably expressed in CHO cells. |
|---|
| Residue: | 481 |
|---|
| Sequence: | MALSYRVSELQSTIPEHILQSTFVHVISSNWSGLQTESIPEEMKQIVEEQGNKLHWAALL
ILMVIIPTIGGNTLVILAVSLEKKLQYATNYFLMSLAVADLLVGLFVMPIALLTIMFEAM
WPLPLVLCPAWLFLDVLFSTASIMHLCAISVDRYIAIKKPIQANQYNSRATAFIKITVVW
LISIGIAIPVPIKGIETDVDNPNNITCVLTKERFGDFMLFGSLAAFFTPLAIMIVTYFLT
IHALQKKAYLVKNKPPQRLTWLTVSTVFQRDETPCSSPEKVAMLDGSRKDKALPNSGDET
LMRRTSTIGKKSVQTISNEQRASKVLGIVFFLFLLMWCPFFITNITLVLCDSCNQTTLQM
LLEIFVWIGYVSSGVNPLVYTLFNKTFRDAFGRYITCNYRATKSVKTLRKRSSKIYFRNP
MAENSKFFKKHGIRNGINPAMYQSPMRLRSSTIQSSSIILLDTLLLTENEGDKTEEQVSY
V
|
|
|
|---|
| BDBM50456162 |
|---|
| n/a |
|---|
| Name | BDBM50456162 |
|---|
| Synonyms: | CHEMBL4217398 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C46H54F3N5O4 |
|---|
| Mol. Mass. | 797.9473 |
|---|
| SMILES | CC(C)(C)OC(=O)NCCCCCCn1cc(CCCCN2CCC(CC2)c2ccc(cc2)-c2cc(cc3cc(ccc23)-c2ccc(cc2)C(F)(F)F)C(O)=O)nn1 |
|---|
| Structure | 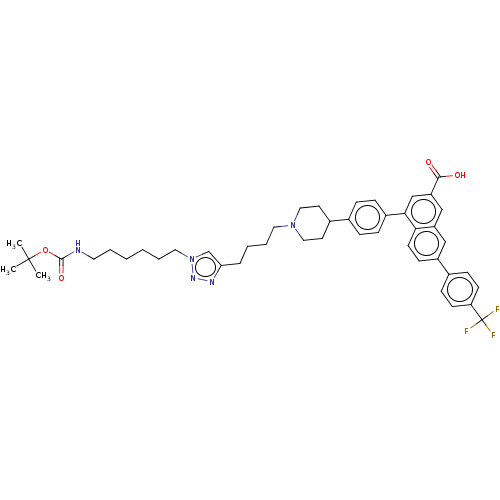
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yu, J; Ciancetta, A; Dudas, S; Duca, S; Lottermoser, J; Jacobson, KA Structure-Guided Modification of Heterocyclic Antagonists of the P2Y J Med Chem61:4860-4882 (2018) [PubMed] Article
Yu, J; Ciancetta, A; Dudas, S; Duca, S; Lottermoser, J; Jacobson, KA Structure-Guided Modification of Heterocyclic Antagonists of the P2Y J Med Chem61:4860-4882 (2018) [PubMed] Article