| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Indoleamine 2,3-dioxygenase 1 |
|---|
| Ligand | BDBM50456431 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1756912 (CHEMBL4191920) |
|---|
| IC50 | >100000±n/a nM |
|---|
| Citation |  Zou, Y; Wang, Y; Wang, F; Luo, M; Li, Y; Liu, W; Huang, Z; Zhang, Y; Guo, W; Xu, Q; Lai, Y Discovery of potent IDO1 inhibitors derived from tryptophan using scaffold-hopping and structure-based design approaches. Eur J Med Chem138:199-211 (2017) [PubMed] Article Zou, Y; Wang, Y; Wang, F; Luo, M; Li, Y; Liu, W; Huang, Z; Zhang, Y; Guo, W; Xu, Q; Lai, Y Discovery of potent IDO1 inhibitors derived from tryptophan using scaffold-hopping and structure-based design approaches. Eur J Med Chem138:199-211 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Indoleamine 2,3-dioxygenase 1 |
|---|
| Name: | Indoleamine 2,3-dioxygenase 1 |
|---|
| Synonyms: | I23O1_HUMAN | IDO | IDO-1 | IDO1 | INDO | Indoleamine 2,3-Dioxygenasae (IDO) | Indoleamine 2,3-dioxygenase | Indoleamine-pyrrole 2,3-dioxygenase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 45330.80 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P14902 |
|---|
| Residue: | 403 |
|---|
| Sequence: | MAHAMENSWTISKEYHIDEEVGFALPNPQENLPDFYNDWMFIAKHLPDLIESGQLRERVE
KLNMLSIDHLTDHKSQRLARLVLGCITMAYVWGKGHGDVRKVLPRNIAVPYCQLSKKLEL
PPILVYADCVLANWKKKDPNKPLTYENMDVLFSFRDGDCSKGFFLVSLLVEIAAASAIKV
IPTVFKAMQMQERDTLLKALLEIASCLEKALQVFHQIHDHVNPKAFFSVLRIYLSGWKGN
PQLSDGLVYEGFWEDPKEFAGGSAGQSSVFQCFDVLLGIQQTAGGGHAAQFLQDMRRYMP
PAHRNFLCSLESNPSVREFVLSKGDAGLREAYDACVKALVSLRSYHLQIVTKYILIPASQ
QPKENKTSEDPSKLEAKGTGGTDLMNFLKTVRSTTEKSLLKEG
|
|
|
|---|
| BDBM50456431 |
|---|
| n/a |
|---|
| Name | BDBM50456431 |
|---|
| Synonyms: | CHEMBL1444923 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H13NO2 |
|---|
| Mol. Mass. | 239.2692 |
|---|
| SMILES | O=c1oc2ccccc2n1CCc1ccccc1 |
|---|
| Structure | 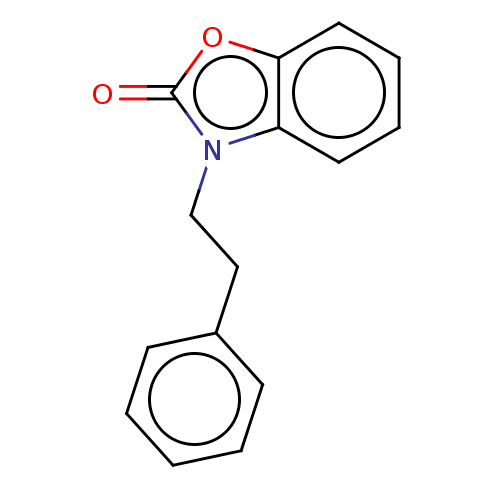
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zou, Y; Wang, Y; Wang, F; Luo, M; Li, Y; Liu, W; Huang, Z; Zhang, Y; Guo, W; Xu, Q; Lai, Y Discovery of potent IDO1 inhibitors derived from tryptophan using scaffold-hopping and structure-based design approaches. Eur J Med Chem138:199-211 (2017) [PubMed] Article
Zou, Y; Wang, Y; Wang, F; Luo, M; Li, Y; Liu, W; Huang, Z; Zhang, Y; Guo, W; Xu, Q; Lai, Y Discovery of potent IDO1 inhibitors derived from tryptophan using scaffold-hopping and structure-based design approaches. Eur J Med Chem138:199-211 (2017) [PubMed] Article