| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Lysine-specific demethylase 5B |
|---|
| Ligand | BDBM191600 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1771179 (CHEMBL4223291) |
|---|
| IC50 | 2.0±n/a nM |
|---|
| Citation |  Nie, Z; Shi, L; Lai, C; O'Connell, SM; Xu, J; Stansfield, RK; Hosfield, DJ; Veal, JM; Stafford, JA Structure-based design and discovery of potent and selective KDM5 inhibitors. Bioorg Med Chem Lett28:1490-1494 (2018) [PubMed] Article Nie, Z; Shi, L; Lai, C; O'Connell, SM; Xu, J; Stansfield, RK; Hosfield, DJ; Veal, JM; Stafford, JA Structure-based design and discovery of potent and selective KDM5 inhibitors. Bioorg Med Chem Lett28:1490-1494 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Lysine-specific demethylase 5B |
|---|
| Name: | Lysine-specific demethylase 5B |
|---|
| Synonyms: | CT31 | Cancer/testis antigen 31 | Histone demethylase JARID1B | JARID1B | Jumonji/ARID domain-containing protein 1B | KDM5B | KDM5B_HUMAN | Lysine-specific demethylase 5B (KDM5B) | Lysine-specific demethylase 5B (KDM5Flag-5B-FL) | PLU-1 | PLU1 | RBBP2H1 | RBP2-H1 | Retinoblastoma-binding protein 2 homolog 1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 175659.67 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q9UGL1 |
|---|
| Residue: | 1544 |
|---|
| Sequence: | MEAATTLHPGPRPALPLGGPGPLGEFLPPPECPVFEPSWEEFADPFAFIHKIRPIAEQTG
ICKVRPPPDWQPPFACDVDKLHFTPRIQRLNELEAQTRVKLNFLDQIAKYWELQGSTLKI
PHVERKILDLFQLNKLVAEEGGFAVVCKDRKWTKIATKMGFAPGKAVGSHIRGHYERILN
PYNLFLSGDSLRCLQKPNLTTDTKDKEYKPHDIPQRQSVQPSETCPPARRAKRMRAEAMN
IKIEPEETTEARTHNLRRRMGCPTPKCENEKEMKSSIKQEPIERKDYIVENEKEKPKSRS
KKATNAVDLYVCLLCGSGNDEDRLLLCDGCDDSYHTFCLIPPLHDVPKGDWRCPKCLAQE
CSKPQEAFGFEQAARDYTLRTFGEMADAFKSDYFNMPVHMVPTELVEKEFWRLVSTIEED
VTVEYGADIASKEFGSGFPVRDGKIKLSPEEEEYLDSGWNLNNMPVMEQSVLAHITADIC
GMKLPWLYVGMCFSSFCWHIEDHWSYSINYLHWGEPKTWYGVPGYAAEQLENVMKKLAPE
LFVSQPDLLHQLVTIMNPNTLMTHEVPVYRTNQCAGEFVITFPRAYHSGFNQGFNFAEAV
NFCTVDWLPLGRQCVEHYRLLHRYCVFSHDEMICKMASKADVLDVVVASTVQKDMAIMIE
DEKALRETVRKLGVIDSERMDFELLPDDERQCVKCKTTCFMSAISCSCKPGLLVCLHHVK
ELCSCPPYKYKLRYRYTLDDLYPMMNALKLRAESYNEWALNVNEALEAKINKKKSLVSFK
ALIEESEMKKFPDNDLLRHLRLVTQDAEKCASVAQQLLNGKRQTRYRSGGGKSQNQLTVN
ELRQFVTQLYALPCVLSQTPLLKDLLNRVEDFQQHSQKLLSEETPSAAELQDLLDVSFEF
DVELPQLAEMRIRLEQARWLEEVQQACLDPSSLTLDDMRRLIDLGVGLAPYSAVEKAMAR
LQELLTVSEHWDDKAKSLLKARPRHSLNSLATAVKEIEEIPAYLPNGAALKDSVQRARDW
LQDVEGLQAGGRVPVLDTLIELVTRGRSIPVHLNSLPRLETLVAEVQAWKECAVNTFLTE
NSPYSLLEVLCPRCDIGLLGLKRKQRKLKEPLPNGKKKSTKLESLSDLERALTESKETAS
AMATLGEARLREMEALQSLRLANEGKLLSPLQDVDIKICLCQKAPAAPMIQCELCRDAFH
TSCVAVPSISQGLRIWLCPHCRRSEKPPLEKILPLLASLQRIRVRLPEGDALRYMIERTV
NWQHRAQQLLSSGNLKFVQDRVGSGLLYSRWQASAGQVSDTNKVSQPPGTTSFSLPDDWD
NRTSYLHSPFSTGRSCIPLHGVSPEVNELLMEAQLLQVSLPEIQELYQTLLAKPSPAQQT
DRSSPVRPSSEKNDCCRGKRDGINSLERKLKRRLEREGLSSERWERVKKMRTPKKKKIKL
SHPKDMNNFKLERERSYELVRSAETHSLPSDTSYSEQEDSEDEDAICPAVSCLQPEGDEV
DWVQCDGSCNQWFHQVCVGVSPEMAEKEDYICVRCTVKDAPSRK
|
|
|
|---|
| BDBM191600 |
|---|
| n/a |
|---|
| Name | BDBM191600 |
|---|
| Synonyms: | 2-(5-((4-chloro-2-methylbenzyl)oxy)-1Hpyrazol-1-yl)isonicotinic acid (N19) | US10173996, Example 89 | US9604961, Example 89 | US9714230, 89 | US9908865, Example 89 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H14ClN3O3 |
|---|
| Mol. Mass. | 343.764 |
|---|
| SMILES | Cc1cc(Cl)ccc1COc1ccnn1-c1cc(ccn1)C(O)=O |
|---|
| Structure | 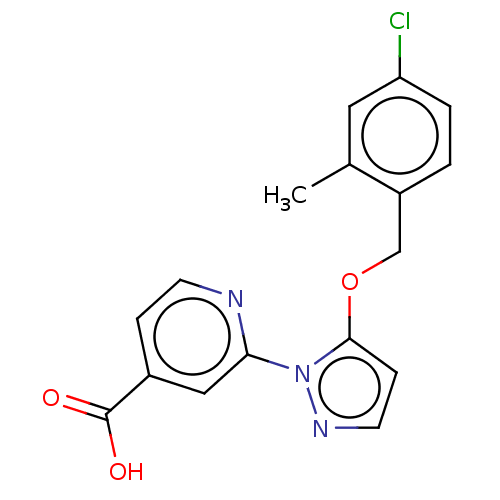
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nie, Z; Shi, L; Lai, C; O'Connell, SM; Xu, J; Stansfield, RK; Hosfield, DJ; Veal, JM; Stafford, JA Structure-based design and discovery of potent and selective KDM5 inhibitors. Bioorg Med Chem Lett28:1490-1494 (2018) [PubMed] Article
Nie, Z; Shi, L; Lai, C; O'Connell, SM; Xu, J; Stansfield, RK; Hosfield, DJ; Veal, JM; Stafford, JA Structure-based design and discovery of potent and selective KDM5 inhibitors. Bioorg Med Chem Lett28:1490-1494 (2018) [PubMed] Article