| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase ITK/TSK |
|---|
| Ligand | BDBM165209 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1778709 (CHEMBL4235701) |
|---|
| IC50 | 8.3±n/a nM |
|---|
| Citation |  Liu, Q; Batt, DG; Chaudhry, C; Lippy, JS; Pattoli, MA; Surti, N; Xu, S; Carter, PH; Burke, JR; Tino, JA Conversion of carbazole carboxamide based reversible inhibitors of Bruton's tyrosine kinase (BTK) into potent, selective irreversible inhibitors in the carbazole, tetrahydrocarbazole, and a new 2,3-dimethylindole series. Bioorg Med Chem Lett28:3080-3084 (2018) [PubMed] Article Liu, Q; Batt, DG; Chaudhry, C; Lippy, JS; Pattoli, MA; Surti, N; Xu, S; Carter, PH; Burke, JR; Tino, JA Conversion of carbazole carboxamide based reversible inhibitors of Bruton's tyrosine kinase (BTK) into potent, selective irreversible inhibitors in the carbazole, tetrahydrocarbazole, and a new 2,3-dimethylindole series. Bioorg Med Chem Lett28:3080-3084 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase ITK/TSK |
|---|
| Name: | Tyrosine-protein kinase ITK/TSK |
|---|
| Synonyms: | EMT | ITK | ITK_HUMAN | Kinase EMT | LYK | T-cell-specific kinase | Tyrosine-protein kinase ITK | Tyrosine-protein kinase ITK (ITK) | Tyrosine-protein kinase Lyk |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 71839.20 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q08881 |
|---|
| Residue: | 620 |
|---|
| Sequence: | MNNFILLEEQLIKKSQQKRRTSPSNFKVRFFVLTKASLAYFEDRHGKKRTLKGSIELSRI
KCVEIVKSDISIPCHYKYPFQVVHDNYLLYVFAPDRESRQRWVLALKEETRNNNSLVPKY
HPNFWMDGKWRCCSQLEKLATGCAQYDPTKNASKKPLPPTPEDNRRPLWEPEETVVIALY
DYQTNDPQELALRRNEEYCLLDSSEIHWWRVQDRNGHEGYVPSSYLVEKSPNNLETYEWY
NKSISRDKAEKLLLDTGKEGAFMVRDSRTAGTYTVSVFTKAVVSENNPCIKHYHIKETND
NPKRYYVAEKYVFDSIPLLINYHQHNGGGLVTRLRYPVCFGRQKAPVTAGLRYGKWVIDP
SELTFVQEIGSGQFGLVHLGYWLNKDKVAIKTIREGAMSEEDFIEEAEVMMKLSHPKLVQ
LYGVCLEQAPICLVFEFMEHGCLSDYLRTQRGLFAAETLLGMCLDVCEGMAYLEEACVIH
RDLAARNCLVGENQVIKVSDFGMTRFVLDDQYTSSTGTKFPVKWASPEVFSFSRYSSKSD
VWSFGVLMWEVFSEGKIPYENRSNSEVVEDISTGFRLYKPRLASTHVYQIMNHCWKERPE
DRPAFSRLLRQLAEIAESGL
|
|
|
|---|
| BDBM165209 |
|---|
| n/a |
|---|
| Name | BDBM165209 |
|---|
| Synonyms: | US10604504, Example 34 | US11623921, Example 34 | US9688629, 34 | US9802915, Example 34 | US9920031, Example 34 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H21N3O3S |
|---|
| Mol. Mass. | 383.464 |
|---|
| SMILES | Cc1[nH]c2c(ccc(-c3cccc(NS(=O)(=O)C=C)c3C)c2c1C)C(N)=O |
|---|
| Structure | 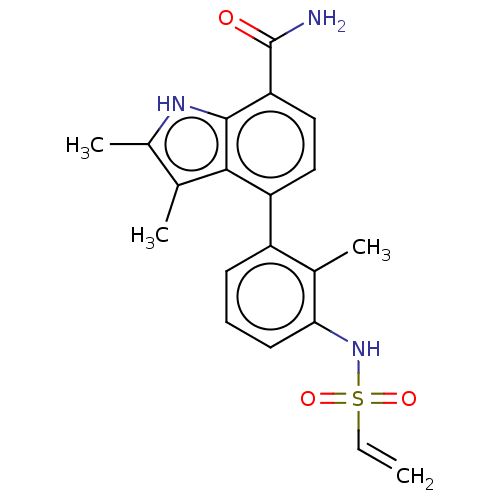
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Liu, Q; Batt, DG; Chaudhry, C; Lippy, JS; Pattoli, MA; Surti, N; Xu, S; Carter, PH; Burke, JR; Tino, JA Conversion of carbazole carboxamide based reversible inhibitors of Bruton's tyrosine kinase (BTK) into potent, selective irreversible inhibitors in the carbazole, tetrahydrocarbazole, and a new 2,3-dimethylindole series. Bioorg Med Chem Lett28:3080-3084 (2018) [PubMed] Article
Liu, Q; Batt, DG; Chaudhry, C; Lippy, JS; Pattoli, MA; Surti, N; Xu, S; Carter, PH; Burke, JR; Tino, JA Conversion of carbazole carboxamide based reversible inhibitors of Bruton's tyrosine kinase (BTK) into potent, selective irreversible inhibitors in the carbazole, tetrahydrocarbazole, and a new 2,3-dimethylindole series. Bioorg Med Chem Lett28:3080-3084 (2018) [PubMed] Article