| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase BTK |
|---|
| Ligand | BDBM50466206 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1792781 (CHEMBL4264700) |
|---|
| IC50 | 52±n/a nM |
|---|
| Citation |  Caldwell, R; Liu-Bujalski, L; Qiu, H; Mochalkin, I; Jones, R; Neagu, C; Goutopoulos, A; Grenningloh, R; Johnson, T; Sherer, B; Gardberg, A; Follis, AV; Morandi, F; Head, J Discovery of a novel series of pyridine and pyrimidine carboxamides as potent and selective covalent inhibitors of Btk. Bioorg Med Chem Lett28:3419-3424 (2018) [PubMed] Article Caldwell, R; Liu-Bujalski, L; Qiu, H; Mochalkin, I; Jones, R; Neagu, C; Goutopoulos, A; Grenningloh, R; Johnson, T; Sherer, B; Gardberg, A; Follis, AV; Morandi, F; Head, J Discovery of a novel series of pyridine and pyrimidine carboxamides as potent and selective covalent inhibitors of Btk. Bioorg Med Chem Lett28:3419-3424 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase BTK |
|---|
| Name: | Tyrosine-protein kinase BTK |
|---|
| Synonyms: | AGMX1 | ATK | Agammaglobulinaemia tyrosine kinase | Agammaglobulinemia tyrosine kinase | B cell progenitor kinase | B-cell progenitor kinase | BPK | BTK | BTK_HUMAN | Bruton tyrosine kinase | Tyrosine Kinase BTK | Tyrosine-protein kinase (BTK) | Tyrosine-protein kinase BTK (BTK) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 76289.95 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q06187 |
|---|
| Residue: | 659 |
|---|
| Sequence: | MAAVILESIFLKRSQQKKKTSPLNFKKRLFLLTVHKLSYYEYDFERGRRGSKKGSIDVEK
ITCVETVVPEKNPPPERQIPRRGEESSEMEQISIIERFPYPFQVVYDEGPLYVFSPTEEL
RKRWIHQLKNVIRYNSDLVQKYHPCFWIDGQYLCCSQTAKNAMGCQILENRNGSLKPGSS
HRKTKKPLPPTPEEDQILKKPLPPEPAAAPVSTSELKKVVALYDYMPMNANDLQLRKGDE
YFILEESNLPWWRARDKNGQEGYIPSNYVTEAEDSIEMYEWYSKHMTRSQAEQLLKQEGK
EGGFIVRDSSKAGKYTVSVFAKSTGDPQGVIRHYVVCSTPQSQYYLAEKHLFSTIPELIN
YHQHNSAGLISRLKYPVSQQNKNAPSTAGLGYGSWEIDPKDLTFLKELGTGQFGVVKYGK
WRGQYDVAIKMIKEGSMSEDEFIEEAKVMMNLSHEKLVQLYGVCTKQRPIFIITEYMANG
CLLNYLREMRHRFQTQQLLEMCKDVCEAMEYLESKQFLHRDLAARNCLVNDQGVVKVSDF
GLSRYVLDDEYTSSVGSKFPVRWSPPEVLMYSKFSSKSDIWAFGVLMWEIYSLGKMPYER
FTNSETAEHIAQGLRLYRPHLASEKVYTIMYSCWHEKADERPTFKILLSNILDVMDEES
|
|
|
|---|
| BDBM50466206 |
|---|
| n/a |
|---|
| Name | BDBM50466206 |
|---|
| Synonyms: | CHEMBL4281335 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H32N6O3 |
|---|
| Mol. Mass. | 464.56 |
|---|
| SMILES | C[C@H]1CN(C[C@@H](C)O1)c1ccc(Nc2nc(ccc2C(N)=O)N2CC[C@@H](C2)NC(=O)C=C)cc1 |r| |
|---|
| Structure | 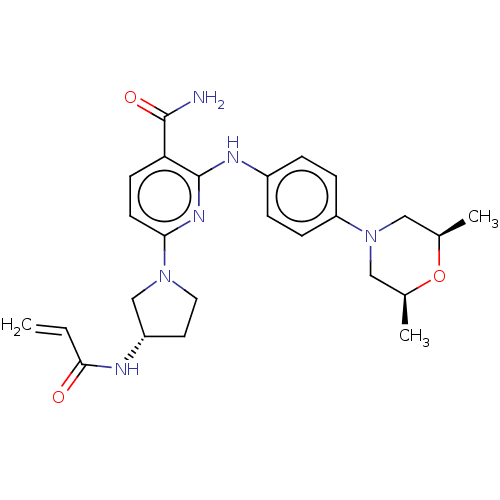
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Caldwell, R; Liu-Bujalski, L; Qiu, H; Mochalkin, I; Jones, R; Neagu, C; Goutopoulos, A; Grenningloh, R; Johnson, T; Sherer, B; Gardberg, A; Follis, AV; Morandi, F; Head, J Discovery of a novel series of pyridine and pyrimidine carboxamides as potent and selective covalent inhibitors of Btk. Bioorg Med Chem Lett28:3419-3424 (2018) [PubMed] Article
Caldwell, R; Liu-Bujalski, L; Qiu, H; Mochalkin, I; Jones, R; Neagu, C; Goutopoulos, A; Grenningloh, R; Johnson, T; Sherer, B; Gardberg, A; Follis, AV; Morandi, F; Head, J Discovery of a novel series of pyridine and pyrimidine carboxamides as potent and selective covalent inhibitors of Btk. Bioorg Med Chem Lett28:3419-3424 (2018) [PubMed] Article