| Reaction Details |
|---|
 | Report a problem with these data |
| Target | DNA gyrase subunit B |
|---|
| Ligand | BDBM50006565 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1802725 (CHEMBL4275017) |
|---|
| IC50 | 6±n/a nM |
|---|
| Citation |  Ho, SY; Wang, W; Ng, FM; Wong, YX; Poh, ZY; Tan, SWE; Ang, SH; Liew, SS; Joyner Wong, YS; Tan, Y; Poulsen, A; Pendharkar, V; Sangthongpitag, K; Manchester, J; Basarab, G; Hill, J; Keller, TH; Cherian, J Discovery of dual GyrB/ParE inhibitors active against Gram-negative bacteria. Eur J Med Chem157:610-621 (2018) [PubMed] Article Ho, SY; Wang, W; Ng, FM; Wong, YX; Poh, ZY; Tan, SWE; Ang, SH; Liew, SS; Joyner Wong, YS; Tan, Y; Poulsen, A; Pendharkar, V; Sangthongpitag, K; Manchester, J; Basarab, G; Hill, J; Keller, TH; Cherian, J Discovery of dual GyrB/ParE inhibitors active against Gram-negative bacteria. Eur J Med Chem157:610-621 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| DNA gyrase subunit B |
|---|
| Name: | DNA gyrase subunit B |
|---|
| Synonyms: | DNA gyrase subunit B | DNA gyrase subunit B (gyrB) | GYRB_ECOLI | Type IIA topoisomerase subunit GyrB | acrB | cou | gyrB | himB | hisU | nalC | parA | pcbA |
|---|
| Type: | Enzyme Subunit |
|---|
| Mol. Mass.: | 89941.28 |
|---|
| Organism: | Escherichia coli (strain K12) |
|---|
| Description: | P0AES6 |
|---|
| Residue: | 804 |
|---|
| Sequence: | MSNSYDSSSIKVLKGLDAVRKRPGMYIGDTDDGTGLHHMVFEVVDNAIDEALAGHCKEII
VTIHADNSVSVQDDGRGIPTGIHPEEGVSAAEVIMTVLHAGGKFDDNSYKVSGGLHGVGV
SVVNALSQKLELVIQREGKIHRQIYEHGVPQAPLAVTGETEKTGTMVRFWPSLETFTNVT
EFEYEILAKRLRELSFLNSGVSIRLRDKRDGKEDHFHYEGGIKAFVEYLNKNKTPIHPNI
FYFSTEKDGIGVEVALQWNDGFQENIYCFTNNIPQRDGGTHLAGFRAAMTRTLNAYMDKE
GYSKKAKVSATGDDAREGLIAVVSVKVPDPKFSSQTKDKLVSSEVKSAVEQQMNELLAEY
LLENPTDAKIVVGKIIDAARAREAARRAREMTRRKGALDLAGLPGKLADCQERDPALSEL
YLVEGDSAGGSAKQGRNRKNQAILPLKGKILNVEKARFDKMLSSQEVATLITALGCGIGR
DEYNPDKLRYHSIIIMTDADVDGSHIRTLLLTFFYRQMPEIVERGHVYIAQPPLYKVKKG
KQEQYIKDDEAMDQYQISIALDGATLHTNASAPALAGEALEKLVSEYNATQKMINRMERR
YPKAMLKELIYQPTLTEADLSDEQTVTRWVNALVSELNDKEQHGSQWKFDVHTNAEQNLF
EPIVRVRTHGVDTDYPLDHEFITGGEYRRICTLGEKLRGLLEEDAFIERGERRQPVASFE
QALDWLVKESRRGLSIQRYKGLGEMNPEQLWETTMDPESRRMLRVTVKDAIAADQLFTTL
MGDAVEPRRAFIEENALKAANIDI
|
|
|
|---|
| BDBM50006565 |
|---|
| n/a |
|---|
| Name | BDBM50006565 |
|---|
| Synonyms: | CHEMBL3235085 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H14F3N5O3S |
|---|
| Mol. Mass. | 437.396 |
|---|
| SMILES | CCNC(=O)Nc1cc(-c2nc(cs2)C(F)(F)F)c(cn1)-c1cncc(c1)C(O)=O |
|---|
| Structure | 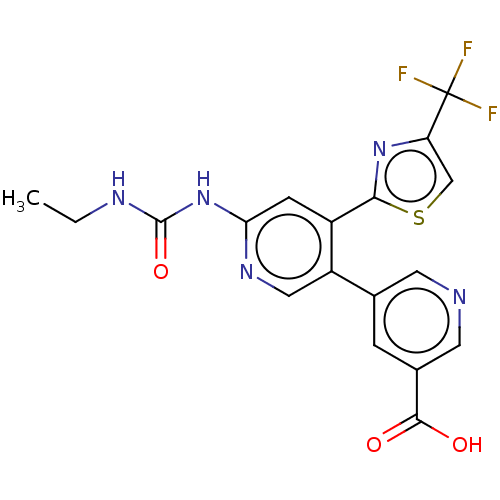
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ho, SY; Wang, W; Ng, FM; Wong, YX; Poh, ZY; Tan, SWE; Ang, SH; Liew, SS; Joyner Wong, YS; Tan, Y; Poulsen, A; Pendharkar, V; Sangthongpitag, K; Manchester, J; Basarab, G; Hill, J; Keller, TH; Cherian, J Discovery of dual GyrB/ParE inhibitors active against Gram-negative bacteria. Eur J Med Chem157:610-621 (2018) [PubMed] Article
Ho, SY; Wang, W; Ng, FM; Wong, YX; Poh, ZY; Tan, SWE; Ang, SH; Liew, SS; Joyner Wong, YS; Tan, Y; Poulsen, A; Pendharkar, V; Sangthongpitag, K; Manchester, J; Basarab, G; Hill, J; Keller, TH; Cherian, J Discovery of dual GyrB/ParE inhibitors active against Gram-negative bacteria. Eur J Med Chem157:610-621 (2018) [PubMed] Article