| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2D6 |
|---|
| Ligand | BDBM249466 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1805962 (CHEMBL4305321) |
|---|
| Ki | 720±n/a nM |
|---|
| Citation |  Cioffi, CL; Racz, B; Varadi, A; Freeman, EE; Conlon, MP; Chen, P; Zhu, L; Kitchen, DB; Barnes, KD; Martin, WH; Pearson, PG; Johnson, G; Blaner, WS; Petrukhin, K Design, Synthesis, and Preclinical Efficacy of Novel Nonretinoid Antagonists of Retinol-Binding Protein 4 in the Mouse Model of Hepatic Steatosis. J Med Chem62:5470-5500 (2019) [PubMed] Article Cioffi, CL; Racz, B; Varadi, A; Freeman, EE; Conlon, MP; Chen, P; Zhu, L; Kitchen, DB; Barnes, KD; Martin, WH; Pearson, PG; Johnson, G; Blaner, WS; Petrukhin, K Design, Synthesis, and Preclinical Efficacy of Novel Nonretinoid Antagonists of Retinol-Binding Protein 4 in the Mouse Model of Hepatic Steatosis. J Med Chem62:5470-5500 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2D6 |
|---|
| Name: | Cytochrome P450 2D6 |
|---|
| Synonyms: | CP2D6_HUMAN | CYP2D6 | CYP2DL1 | CYPIID6 | Cytochrome P450 2D6 (CYP2D6) | Debrisoquine 4-hydroxylase | P450-DB1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55774.82 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P10635 |
|---|
| Residue: | 497 |
|---|
| Sequence: | MGLEALVPLAVIVAIFLLLVDLMHRRQRWAARYPPGPLPLPGLGNLLHVDFQNTPYCFDQ
LRRRFGDVFSLQLAWTPVVVLNGLAAVREALVTHGEDTADRPPVPITQILGFGPRSQGVF
LARYGPAWREQRRFSVSTLRNLGLGKKSLEQWVTEEAACLCAAFANHSGRPFRPNGLLDK
AVSNVIASLTCGRRFEYDDPRFLRLLDLAQEGLKEESGFLREVLNAVPVLLHIPALAGKV
LRFQKAFLTQLDELLTEHRMTWDPAQPPRDLTEAFLAEMEKAKGNPESSFNDENLRIVVA
DLFSAGMVTTSTTLAWGLLLMILHPDVQRRVQQEIDDVIGQVRRPEMGDQAHMPYTTAVI
HEVQRFGDIVPLGVTHMTSRDIEVQGFRIPKGTTLITNLSSVLKDEAVWEKPFRFHPEHF
LDAQGHFVKPEAFLPFSAGRRACLGEPLARMELFLFFTSLLQHFSFSVPTGQPRPSHHGV
FAFLVSPSPYELCAVPR
|
|
|
|---|
| BDBM249466 |
|---|
| n/a |
|---|
| Name | BDBM249466 |
|---|
| Synonyms: | US10072016, Compound 65 | US10407433, Compound 65 | US10913746, Compound 65 | US11649240, Compound 65 | US9434727, 65 | US9777010, Compound 65 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H15F4N5O |
|---|
| Mol. Mass. | 417.3596 |
|---|
| SMILES | Fc1cccc(C2CCN(CC2)C(=O)c2nnc3ccc(cn23)C#N)c1C(F)(F)F |
|---|
| Structure | 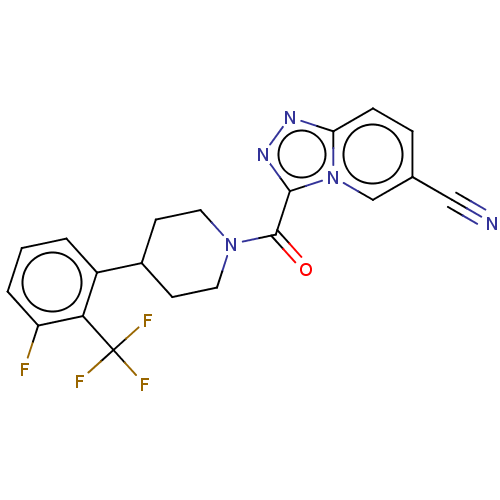
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cioffi, CL; Racz, B; Varadi, A; Freeman, EE; Conlon, MP; Chen, P; Zhu, L; Kitchen, DB; Barnes, KD; Martin, WH; Pearson, PG; Johnson, G; Blaner, WS; Petrukhin, K Design, Synthesis, and Preclinical Efficacy of Novel Nonretinoid Antagonists of Retinol-Binding Protein 4 in the Mouse Model of Hepatic Steatosis. J Med Chem62:5470-5500 (2019) [PubMed] Article
Cioffi, CL; Racz, B; Varadi, A; Freeman, EE; Conlon, MP; Chen, P; Zhu, L; Kitchen, DB; Barnes, KD; Martin, WH; Pearson, PG; Johnson, G; Blaner, WS; Petrukhin, K Design, Synthesis, and Preclinical Efficacy of Novel Nonretinoid Antagonists of Retinol-Binding Protein 4 in the Mouse Model of Hepatic Steatosis. J Med Chem62:5470-5500 (2019) [PubMed] Article