| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM50308002 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_975876 (CHEMBL2415525) |
|---|
| Ki | 6910±n/a nM |
|---|
| Citation |  Pompeu, TE; Alves, FR; Figueiredo, CD; Antonio, CB; Herzfeldt, V; Moura, BC; Rates, SM; Barreiro, EJ; Fraga, CA; No�l, F Synthesis and pharmacological evaluation of new N-phenylpiperazine derivatives designed as homologues of the antipsychotic lead compound LASSBio-579. Eur J Med Chem66:122-34 (2013) [PubMed] Article Pompeu, TE; Alves, FR; Figueiredo, CD; Antonio, CB; Herzfeldt, V; Moura, BC; Rates, SM; Barreiro, EJ; Fraga, CA; No�l, F Synthesis and pharmacological evaluation of new N-phenylpiperazine derivatives designed as homologues of the antipsychotic lead compound LASSBio-579. Eur J Med Chem66:122-34 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2A | 5-HT2 | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_RAT | Htr2 | Htr2a | Serotonin Receptor 2A |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52852.05 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Rat cortex membranes 5-HT2A receptors. |
|---|
| Residue: | 471 |
|---|
| Sequence: | MEILCEDNISLSSIPNSLMQLGDGPRLYHNDFNSRDANTSEASNWTIDAENRTNLSCEGY
LPPTCLSILHLQEKNWSALLTTVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAIWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VAFFIPLTIMVITYFLTIKSLQKEATLCVSDLSTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYAGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNENVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENRKPLQLILVNTIPALAYK
SSQLQVGQKKNSQEDAEQTVDDCSMVTLGKQQSEENCTDNIETVNEKVSCV
|
|
|
|---|
| BDBM50308002 |
|---|
| n/a |
|---|
| Name | BDBM50308002 |
|---|
| Synonyms: | 1-[(1-(4-Chlorophenyl)-1H-pyrazol-4-yl)methyl]-4-phenylpiperazine | CHEMBL597601 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H21ClN4 |
|---|
| Mol. Mass. | 352.861 |
|---|
| SMILES | Clc1ccc(cc1)-n1cc(CN2CCN(CC2)c2ccccc2)cn1 |
|---|
| Structure | 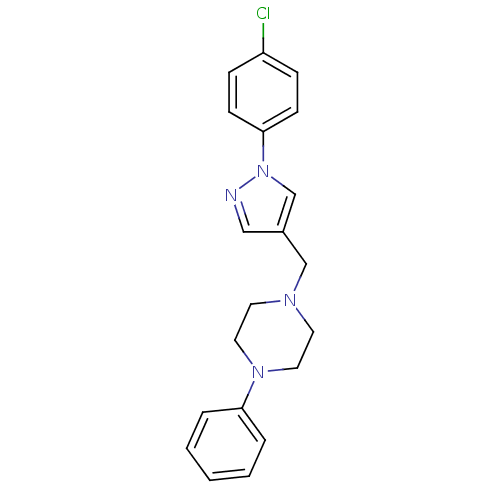
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Pompeu, TE; Alves, FR; Figueiredo, CD; Antonio, CB; Herzfeldt, V; Moura, BC; Rates, SM; Barreiro, EJ; Fraga, CA; No�l, F Synthesis and pharmacological evaluation of new N-phenylpiperazine derivatives designed as homologues of the antipsychotic lead compound LASSBio-579. Eur J Med Chem66:122-34 (2013) [PubMed] Article
Pompeu, TE; Alves, FR; Figueiredo, CD; Antonio, CB; Herzfeldt, V; Moura, BC; Rates, SM; Barreiro, EJ; Fraga, CA; No�l, F Synthesis and pharmacological evaluation of new N-phenylpiperazine derivatives designed as homologues of the antipsychotic lead compound LASSBio-579. Eur J Med Chem66:122-34 (2013) [PubMed] Article