| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuropeptide Y receptor type 1 |
|---|
| Ligand | BDBM50500159 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1543512 (CHEMBL3750359) |
|---|
| Ki | 3.1±n/a nM |
|---|
| Citation |  Keller, M; Weiss, S; Hutzler, C; Kuhn, KK; Mollereau, C; Dukorn, S; Schindler, L; Bernhardt, G; K�nig, B; Buschauer, A N(?)-Carbamoylation of the Argininamide Moiety: An Avenue to Insurmountable NPY Y1 Receptor Antagonists and a Radiolabeled Selective High-Affinity Molecular Tool ([(3)H]UR-MK299) with Extended Residence Time. J Med Chem58:8834-49 (2015) [PubMed] Article Keller, M; Weiss, S; Hutzler, C; Kuhn, KK; Mollereau, C; Dukorn, S; Schindler, L; Bernhardt, G; K�nig, B; Buschauer, A N(?)-Carbamoylation of the Argininamide Moiety: An Avenue to Insurmountable NPY Y1 Receptor Antagonists and a Radiolabeled Selective High-Affinity Molecular Tool ([(3)H]UR-MK299) with Extended Residence Time. J Med Chem58:8834-49 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neuropeptide Y receptor type 1 |
|---|
| Name: | Neuropeptide Y receptor type 1 |
|---|
| Synonyms: | NPY-Y1 | NPY1-R | NPY1R | NPY1R_HUMAN | NPYR | NPYY1 | neuropeptide Y receptor Y1 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 44399.07 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | NPY-Y1 NPY1R HUMAN::P25929 |
|---|
| Residue: | 384 |
|---|
| Sequence: | MNSTLFSQVENHSVHSNFSEKNAQLLAFENDDCHLPLAMIFTLALAYGAVIILGVSGNLA
LIIIILKQKEMRNVTNILIVNLSFSDLLVAIMCLPFTFVYTLMDHWVFGEAMCKLNPFVQ
CVSITVSIFSLVLIAVERHQLIINPRGWRPNNRHAYVGIAVIWVLAVASSLPFLIYQVMT
DEPFQNVTLDAYKDKYVCFDQFPSDSHRLSYTTLLLVLQYFGPLCFIFICYFKIYIRLKR
RNNMMDKMRDNKYRSSETKRINIMLLSIVVAFAVCWLPLTIFNTVFDWNHQIIATCNHNL
LFLLCHLTAMISTCVNPIFYGFLNKNFQRDLQFFFNFCDFRSRDDDYETIAMSTMHTDVS
KTSLKQASPVAFKKINNNDDNEKI
|
|
|
|---|
| BDBM50500159 |
|---|
| n/a |
|---|
| Name | BDBM50500159 |
|---|
| Synonyms: | CHEMBL3746893 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C39H39F3N12O6 |
|---|
| Mol. Mass. | 828.799 |
|---|
| SMILES | OC(=O)C(F)(F)F.N\C(NCCC[C@@H](NC(=O)C(c1ccccc1)c1ccccc1)C(=O)NCc1ccc(O)cc1)=N\C(=O)NCc1cn(nn1)-c1cccc(c1)N=[N+]=[N-] |r| |
|---|
| Structure | 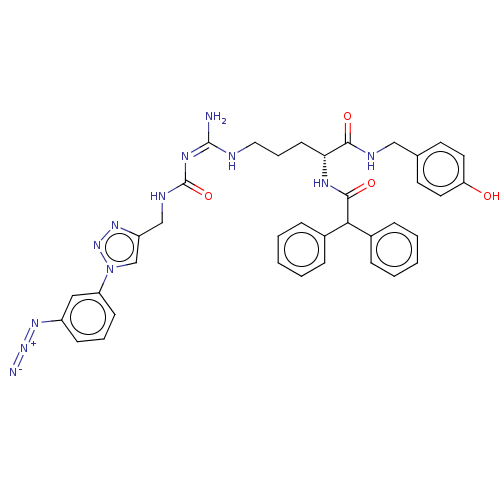
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Keller, M; Weiss, S; Hutzler, C; Kuhn, KK; Mollereau, C; Dukorn, S; Schindler, L; Bernhardt, G; K�nig, B; Buschauer, A N(?)-Carbamoylation of the Argininamide Moiety: An Avenue to Insurmountable NPY Y1 Receptor Antagonists and a Radiolabeled Selective High-Affinity Molecular Tool ([(3)H]UR-MK299) with Extended Residence Time. J Med Chem58:8834-49 (2015) [PubMed] Article
Keller, M; Weiss, S; Hutzler, C; Kuhn, KK; Mollereau, C; Dukorn, S; Schindler, L; Bernhardt, G; K�nig, B; Buschauer, A N(?)-Carbamoylation of the Argininamide Moiety: An Avenue to Insurmountable NPY Y1 Receptor Antagonists and a Radiolabeled Selective High-Affinity Molecular Tool ([(3)H]UR-MK299) with Extended Residence Time. J Med Chem58:8834-49 (2015) [PubMed] Article